Introduction to Does Dupixent Cause Cancer?
If you are reasearcing “does Dupixent Cause Cancer” you have come the right place. Dupixent (dupilumab) is widely prescribed for chronic inflammatory conditions that can significantly affect quality of life, including moderate to severe atopic dermatitis (eczema), asthma with an eosinophilic phenotype, chronic rhinosinusitis with nasal polyps, eosinophilic esophagitis, prurigo nodularis, and certain pediatric indications. Because Dupixent is a long-term therapy for many patients, a practical and reasonable question follows: does Dupixent cause cancer?
The most accurate answer, based on current evidence, is this: Dupixent is not considered a cancer-causing medication, and clinical trials and post-marketing data have not established a causal link between dupilumab and cancer. However, as with any immune-modulating therapy, the topic deserves a precise explanation, careful definitions, and a forward-looking approach to risk management.
This article explains what Dupixent does, what the evidence shows regarding malignancy, where uncertainty remains, and what patients and clinicians can do to monitor risk responsibly.
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected]. (24/7/365).

What Dupixent Is, and Why the Cancer Question Comes Up
Dupixent (dupilumab) is a monoclonal antibody that targets the interleukin-4 receptor alpha (IL-4Rα) subunit. By binding IL-4Rα, Dupixent inhibits signaling of both IL-4 and IL-13, two key cytokines driving type 2 inflammation.
That mechanism matters because the immune system plays a dual role:
- Inflammation contributes to disease (eczema, asthma, nasal polyps).
- Immune surveillance helps detect and eliminate abnormal cells, including potentially malignant cells.
When a medication changes immune signaling, patients often worry about whether immune surveillance could be weakened in a way that increases cancer risk. This is a common concern with some immunosuppressive therapies used in dermatology and rheumatology. It is also a reason physicians take careful histories regarding malignancy when selecting systemic treatments.
Dupixent is generally described as an immune-modulating therapy, not a broad immunosuppressant. That distinction does not eliminate all theoretical questions about its safety profile. In fact, there have been instances where patients have pursued legal action due to alleged links between Dupixent and cancer. For example, some individuals have sought compensation in a Dupixent cancer lawsuit or have tried to qualify for such lawsuits. These cases often stem from concerns about potential malignancy risks associated with the drug.
However, it’s essential to note that while these lawsuits exist, they do not provide definitive evidence of causation between Dupixent and cancer. The legal landscape surrounding these claims is complex and evolving. If you or someone you know is considering legal action due to health concerns related to Dupixent use or if you want more information on the Dupixent cancer lawsuit process, it’s advisable to seek professional legal counsel.
In conclusion, while there are ongoing discussions and investigations into the potential risks associated with Dupixent use, it remains an important treatment option
Key Definitions: “Cancer Risk” Can Mean Several Different Things
When people ask whether a drug “causes cancer,” they may be asking different questions. Clinically, it helps to separate them:
- Carcinogenicity: Does the medication directly initiate or promote cancer formation?
- Association: Are cancer rates higher in treated populations compared with expected rates?
- Causality: If there is a higher rate, can we attribute it to the medication rather than confounding factors such as age, smoking, disease severity, or prior therapies?
- Cancer recurrence: In patients with a history of malignancy, does the medication increase recurrence risk?
- Detection bias: Are cancers found more often because patients are monitored more closely?
Good governance in medicine depends on being explicit about which question is being asked, and which evidence can answer it.
What the Clinical Evidence Says About Dupixent and Cancer
1) Clinical trials did not show a clear cancer signal
In pre-approval and subsequent clinical trials across multiple indications, malignancies have been reported, as they are in any large population over time, but no consistent pattern emerged indicating dupilumab causes cancer. Regulators and manufacturers monitor adverse events, including malignancies, during trials and in post-marketing surveillance.
A crucial point is that clinical trials have limitations:
- They may not be powered to detect rare events.
- They usually exclude patients with certain cancers or complex histories.
- Follow-up duration may be limited compared with real-world use.
Even with those limitations, the absence of a consistent malignancy signal is meaningful when interpreted alongside mechanistic considerations and accumulating post-marketing data.
However, if you or a loved one has experienced negative health effects after using Dupixent, you might want to explore your legal options. You can find out more about the eligibility criteria for a Dupixent cancer lawsuit and understand more about the potential link between Dupixent and cancer.
2) Post-marketing surveillance has not established a causal link
After a medication is approved, adverse events are tracked through pharmacovigilance systems and real-world observational studies. To date, broad post-marketing experience with Dupixent has not established that dupilumab causes cancer. However, there have been instances where patients have experienced severe side effects leading to Dupixent lawsuits, which underscores the importance of thorough post-marketing surveillance.
However, pharmacovigilance data regarding Dupixent and Cancer must be interpreted carefully:
- Spontaneous reports do not prove causality.
- Reporting rates vary.
- Patients with severe disease may have different baseline risks.
In other words, surveillance is essential for detection, but it is not the same as proof.
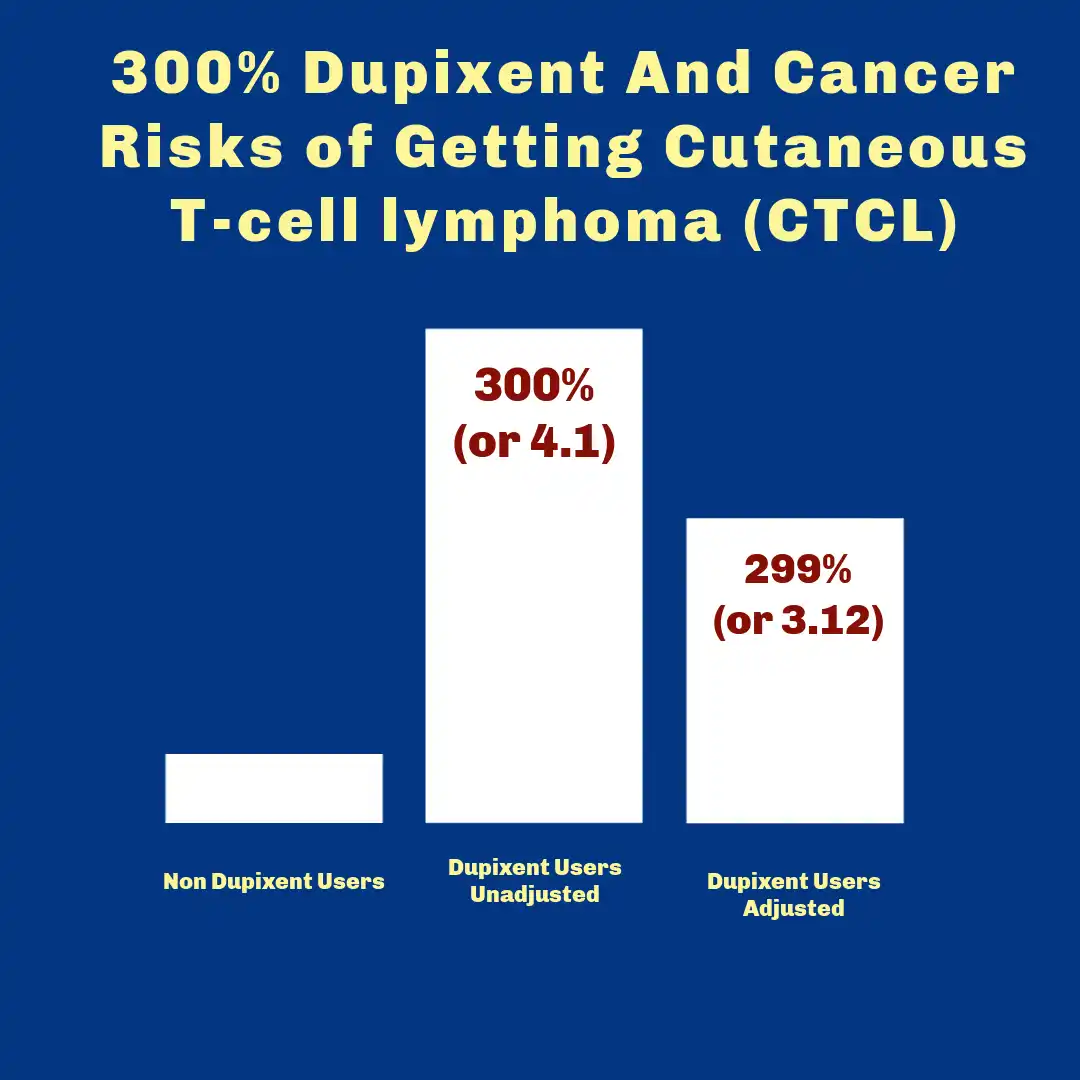
3) The special case: cutaneous T-cell lymphoma (CTCL) and diagnostic confusion
If you research Dupixent and cancer, you may come across discussions involving cutaneous T-cell lymphoma (CTCL), including mycosis fungoides, and whether some cases were “unmasked” after dupilumab initiation. This topic is nuanced and often misunderstood. The critical issue is that:
- Early CTCL can resemble eczema in both appearance and symptoms.
- Some patients treated for presumed atopic dermatitis may actually have undiagnosed CTCL.
- When a therapy improves some inflammatory pathways but not the underlying lymphoma, the clinical picture may change, leading to a diagnosis that might otherwise have been delayed.
This is not the same as saying Dupixent causes CTCL. In many reported scenarios, the more plausible explanation is misdiagnosis or delayed diagnosis, with subsequent recognition after treatment changes the disease’s presentation or triggers further evaluation.
That said, responsible clinicians treat this as a governance issue: when a patient’s “eczema” behaves atypically, is treatment-resistant, or worsens in unexpected ways, re-evaluation and biopsy should be considered. It’s crucial to note that ongoing monitoring and updates regarding potential risks associated with Dupixent are necessary. For instance, recent Dupixent lawsuit updates highlight the need for continued vigilance in understanding the full scope of this medication’s effects on patients’ health.

Mechanism: Does Blocking IL-4 and IL-13 Make Cancer More Likely?
Mechanistic reasoning can support or challenge a safety concern, but it cannot replace outcome data. Still, it matters.
Dupixent blocks IL-4 and IL-13 signaling, which are central to type 2 inflammation. This pathway is distinct from the pathways targeted by broad immunosuppressants that significantly impair host defenses.
Important considerations:
- Dupixent does not broadly deplete immune cells.
- It is not a traditional systemic immunosuppressant in the way that some corticosteroids, calcineurin inhibitors, or certain cytotoxic agents are.
- It does not appear to cause a general immunodeficiency state.
From a theoretical standpoint, altering cytokine signaling could affect tumor biology in complex ways, depending on tumor type and immune environment. However, the key question is not whether a pathway is involved in immunity in the abstract, but whether real-world outcomes show increased malignancy incidence attributable to the drug. Current evidence has not demonstrated that.
However, there have been concerns regarding the potential cancer risk associated with Dupixent, which adds another layer of complexity to this discussion.
The Baseline Risk Issue: Severe Inflammatory Disease Can Complicate Interpretation of Dupixent and Cancer
Another reason cancer questions arise is that chronic inflammatory diseases and their treatments can influence baseline risk.
For example:
- Long-standing inflammation may be associated with changes in tissue microenvironments.
- Severe eczema can lead to repeated immune activation and frequent use of systemic agents over time.
- Some patients have extensive prior exposure to therapies that may have their own risk profiles.
So when a cancer is diagnosed during Dupixent therapy, the appropriate governance question is:
- Is this temporally associated, or causally associated?
- What was the patient’s baseline risk, and what therapies preceded dupilumab?
These distinctions support better clinical decisions and more accurate risk communication.
What the Product Labeling and Safety Messaging Generally Emphasize
Medication labels and prescribing information, which are designed to reflect known risks and uncertainties based on regulatory review, are a crucial part of the healthcare system. For instance, the FDA’s labeling resources provide essential information about medication safety. While labeling evolves, the overarching reality is that Dupixent is not generally labeled as a medication known to cause cancer in the way some immunosuppressants carry boxed warnings or explicit malignancy warnings.
That does not mean “no risk exists.” It means that, at present, there is no established causal relationship that warrants that level of warning. In medical governance terms, this is the difference between:
- A known risk (established and measurable), and
- A potential risk (theoretical or unconfirmed), and
- A monitored risk (tracked over time as evidence accumulates).
Practical Risk Management: What Patients Should Monitor While on Dupixent
Even when a medication is not considered carcinogenic, proactive monitoring is still the right approach. The goal is not alarm. The goal is early detection of problems that may be unrelated to the drug but important for long-term outcomes.
Consider discussing the following with your clinician:
1) Unexpected changes in skin disease
If you use Dupixent for eczema or prurigo nodularis, inform your clinician if you notice:
- New, persistent patches or plaques that do not respond
- Worsening lesions despite adherence
- Unusual distribution (for example, new lesions in areas not previously involved)
- Persistent lymph node enlargement
- Systemic symptoms such as unexplained weight loss, drenching night sweats, or persistent fevers
These symptoms do not automatically indicate cancer, but they justify timely evaluation.
2) Age-appropriate cancer screening
While Dupixent is a widely used medication, it’s important to note that it does not replace standard preventive care. Therefore, it’s essential to stay current with screenings appropriate for your age and risk profile, such as:
- Cervical cancer screening
- Breast cancer screening
- Colorectal cancer screening
- Lung cancer screening for eligible high-risk individuals
- Prostate cancer screening when indicated
Good outcomes depend on consistency and adherence to the schedule. Screening works best when it is done regularly and on time.
However, there have been concerns about potential links between Dupixent and cancer. If you or a loved one has experienced adverse effects related to this medication, particularly concerning Dupixent and cancer, it may be advisable to seek legal counsel. A Dupixent cancer lawyer can provide guidance on how to navigate these complex issues.
3) Dermatologic surveillance for high-risk patients
If you have a history of extensive sun exposure, prior skin cancers, or significant actinic damage, periodic skin checks can be appropriate regardless of Dupixent use.
It’s crucial to be aware of the potential risks associated with Dupixent. In some cases, patients may develop skin cancers while on this medication. If you suspect that your skin condition has worsened due to Dupixent, consider consulting with a Dupixent cancer lawyer who can assist in addressing these concerns.
4) Provide a complete oncologic history
If you have a history of cancer, decisions about Dupixent should be individualized. The key variables often include:
- Cancer type and stage
- Time since remission
- Current surveillance plan
- Ongoing oncology input
Many clinicians coordinate with oncology when there is a recent cancer history or a tumor type with higher recurrence risk. This is a governance best practice because it formalizes shared decision-making and clarifies accountability.
If you have suffered from any negative consequences related to Dupixent use, particularly concerning your oncologic health, it’s crucial to seek professional help. A Dupixent cancer lawsuit may provide a pathway for addressing these serious concerns.
Who Should Be Especially Careful When Starting Dupixent?
Dupixent is generally well tolerated but certain scenarios warrant higher scrutiny and a more deliberate diagnostic approach.
Patients with “eczema” that is atypical or treatment-resistant
If dermatitis is severe, persistent, refractory, or has features that do not match classic atopic dermatitis, clinicians may consider:
- Biopsy before initiating long-term systemic therapy
- Patch testing if allergic contact dermatitis is suspected
- Evaluation for alternative dermatoses including CTCL when clinically appropriate
The purpose is not to delay care unnecessarily but rather to confirm the diagnosis before committing to long-term targeted therapy. If any complications arise during this process that could be linked back to the use of Dupixent, don’t hesitate to reach out to a [Dupixent cancer lawyer](https://classactionlawyertn.com/dupixent.
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected]. (24/7/365).
Patients with a prior malignancy
A past cancer diagnosis does not automatically exclude Dupixent, but it typically calls for:
- Documentation of remission status
- Oncology coordination where appropriate
- Clear monitoring expectations
That structure protects patients and supports clinically defensible decision-making.
Common Questions Patients Ask (With Clear Answers)
“If Dupixent is not immunosuppressive, why do people still worry?”
Because it affects immune signaling, and the immune system is involved in tumor surveillance. Concern is understandable. What matters is that current clinical evidence has not shown a causal cancer risk signal comparable to therapies that broadly suppress immunity.
“I started Dupixent and then I was diagnosed with cancer. Did Dupixent cause it?”
Not necessarily. Timing alone does not prove causation. Many cancers develop over years. The right next step is a clinician-led review of your personal risk factors, cancer type, timeline, and prior treatments.
However, there have been instances where patients have filed Dupixent cancer lawsuits due to concerns linking the medication to their cancer diagnoses. These cases are often complex and require thorough investigation.
“Can Dupixent make an existing cancer grow faster?”
There is no general evidence that Dupixent promotes cancer growth. If you have an active malignancy or are under evaluation for one, treatment decisions should be coordinated with oncology to align with your overall care plan.
“Should I stop Dupixent because I am scared of cancer?”
Do not stop a prescribed biologic abruptly without medical advice. If you have concerns, schedule a focused discussion with your prescriber. Risk should be evaluated in context, and the plan should be documented clearly.
For those worried about potential risks associated with Dupixent, it’s important to note that there have been ongoing lawsuits related to Dupixent and cancer. However, each case is unique and it’s essential to consult with healthcare professionals before making any decisions about your treatment plan.
Dupixent Safety in Context: Comparing Risk Without Creating False Equivalence
Patients often hear that “biologics can increase cancer risk.” That statement is too broad to be useful.
Biologics differ significantly by target, degree of immune suppression, and the populations studied. Some therapies have clearer malignancy warnings because they alter immune function more broadly or have shown signals in specific settings. Dupixent’s target and safety profile are different, and the current data have not established that it causes cancer.
A forward-thinking approach avoids two errors:
- Complacency: assuming no monitoring is needed.
- Catastrophizing: assuming any immune-targeting therapy is carcinogenic.
The best standard is disciplined vigilance: monitor, document, reassess, and escalate evaluation when clinical red flags appear.
When to Contact Your Doctor Urgently
Seek medical attention promptly if you experience:
- A rapidly enlarging lymph node or persistent lymph node swelling
- Unexplained weight loss or persistent fatigue that is new and progressive
- Night sweats that soak clothing or bedding
- Persistent fevers without explanation
- New skin lesions that grow, bleed, ulcerate, or fail to heal
- Significant changes in your skin condition that do not fit your usual pattern
These symptoms are not specific to cancer, but they warrant timely evaluation.
Bottom Line: Does Dupixent Cause Cancer?
Based on current clinical trial data, post-marketing experience, and mechanistic understanding, Dupixent is not considered to cause cancer, and no causal relationship has been established between dupilumab and malignancy. However, it’s essential to stay informed about the ongoing discussions surrounding Dupixent and its potential links to cancer, as some patients have raised concerns leading to Dupixent cancer lawsuits.
At the same time, long-term therapies deserve long-term governance. That means:
- Confirming the diagnosis, especially in atypical dermatitis.
- Staying current on routine cancer screening.
- Reporting unexpected changes promptly.
- Coordinating care in patients with a prior malignancy.
If you are using Dupixent or considering it and want a personalized risk assessment regarding its potential cancer risks, the most productive next step is to review your medical history, family history, prior immunomodulating treatments, and screening status with your clinician. This is how uncertainty is managed responsibly, and how long-term outcomes are protected proactively.
Frequently Asked Questions About Dupixent and Cancer
What is Dupixent (dupilumab) used for?
Dupixent (dupilumab) is a monoclonal antibody prescribed to treat chronic inflammatory conditions such as moderate to severe atopic dermatitis (eczema), asthma with an eosinophilic phenotype, chronic rhinosinusitis with nasal polyps, eosinophilic esophagitis, prurigo nodularis, and certain pediatric indications.
Does Dupixent cause cancer?
Based on current clinical trials and post-marketing data, Dupixent is not considered a cancer-causing medication. No causal link between dupilumab and cancer has been established. However, as with any immune-modulating therapy, ongoing monitoring and careful risk management are important.
How does Dupixent affect the immune system?
Dupixent targets the interleukin-4 receptor alpha (IL-4Rα) subunit and inhibits signaling of IL-4 and IL-13 cytokines that drive type 2 inflammation. It is classified as an immune-modulating therapy rather than a broad immunosuppressant, which helps reduce inflammation while maintaining immune surveillance against abnormal cells.
What does ‘cancer risk’ mean in the context of medications like Dupixent?
‘Cancer risk’ can refer to several concepts: carcinogenicity (whether the drug directly causes cancer), association (higher cancer rates in treated populations), causality (if the medication is responsible rather than other factors), cancer recurrence risk in patients with prior malignancy, and detection bias due to increased monitoring. Understanding these distinctions is crucial when evaluating safety evidence.
Have there been any legal actions related to Dupixent and cancer?
Some individuals have pursued or considered filing a Dupixent Cancer Lawsuit alleging links between Dupixent and cancer. These lawsuits reflect patient concerns but do not provide definitive scientific evidence of causation. The legal landscape is complex and evolving; those interested should seek professional legal counsel regarding eligibility and process for Dupixent cancer lawsuits.
What limitations exist in clinical trials assessing Dupixent’s cancer risk?
Clinical trials may not detect rare events due to limited sample size or duration, often exclude patients with certain cancers or complex medical histories, and follow-up periods may be shorter than real-world use. Despite these limitations, no consistent pattern indicating that dupilumab causes cancer has emerged from trials or post-marketing surveillance.


