Introduction to Dupixent and Cancer: What You Need to Know
- Dupixent and Cancer: Recent scientific investigations have raised important questions about Dupixent and its potential cancer risks, particularly regarding the development of cutaneous T-cell lymphoma (CTCL). Studies published in 2024 and 2025 have reported higher rates of this rare skin cancer among patients receiving dupilumab treatment. In response to these concerns, the FDA acknowledged the issue in October 2024 by placing Dupixent on its “Potential Signals of Serious Risks” list, which means they will be investigating the reported cases of CTCL. As a result of these developments, individual are filing a Dupixent Cancer Lawsuit against the manufacturers, with plaintiffs claiming that there were insufficient warnings about the potential risks of malignancy.
- Dupixent (dupilumab): Is a significant advancement in biologic therapy for patients suffering from chronic inflammatory conditions. It has been approved by the FDA for various uses and works by targeting specific pathways in the immune system to treat conditions such as atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyps, and eosinophilic esophagitis. Dupixent is produced through a collaboration between Sanofi and Regeneron and has experienced great commercial success since its initial approval in March 2017.
- Comprehensive Patient Guide: This article explores the connection between Dupixent and cancer, looking at available clinical data, regulatory actions, and legal proceedings. It is crucial for patients who have been prescribed this biologic therapy to have access to comprehensive information about Dupixent side effects and any emerging safety signals. This knowledge will empower them to make informed decisions about their treatment in collaboration with their healthcare providers.
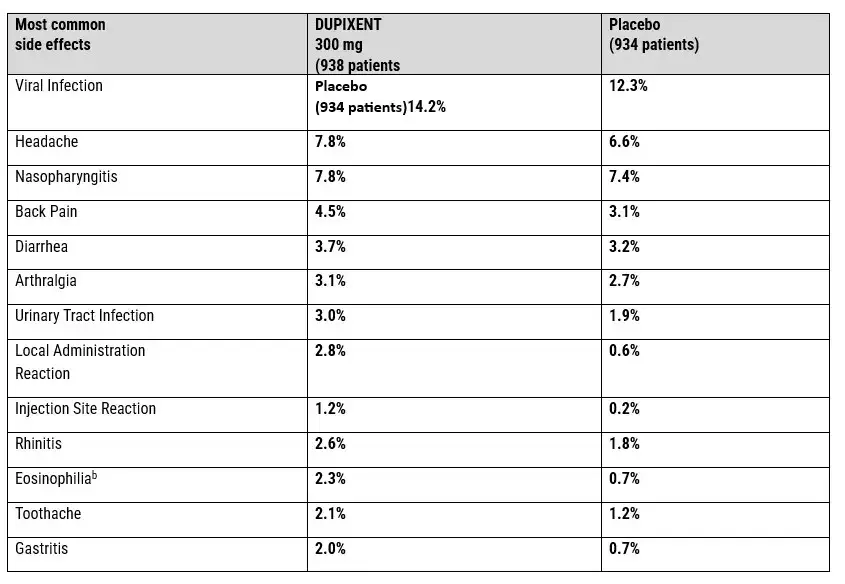
DUPIXENT COMMON SIDE EFFECTS PROFILE
Understanding Cutaneous T-Cell Lymphoma (CTCL)
- Cutaneous T-cell Lymphoma: Is a rare type of non-Hodgkin lymphoma that occurs when T-lymphocytes, which are white blood cells responsible for immune defense, become cancerous and accumulate in the skin.
- Slow Progressing Cancer: Unlike more aggressive lymphomas that develop quickly, this slow-growing cancer usually progresses over months or years.
- Laborious Diagnosis: The rarity of CTCL and its varying symptoms make it difficult to diagnose and may lead to delays in starting the right treatment.
Common Cutaneous T-Cell Lymphoma Symptoms
The signs of CTCL often resemble harmless skin conditions, making it challenging for healthcare providers to diagnose. Patients usually present with:
- Persistent itchy, scaly rashes that do not respond to standard dermatological treatments
- Red patches or plaques on the skin, especially in areas protected from the sun such as the buttocks, hips, and lower abdomen
- Raised lesions that may develop into tumors in advanced stages
- Erythroderma, characterized by widespread redness affecting large areas of the body
- Thickened skin with an unusual texture or appearance
Types of CTCL
There are several different subtypes of cutaneous T-cell lymphoma, each with its own clinical features and prognoses:
- Mycosis fungoides is the most common form of CTCL, making up about 50% of all cases. This subtype usually appears as patches and plaques that slowly progress over years, potentially leading to tumor formation in later stages.
- Sézary syndrome is an aggressive variant characterized by widespread erythroderma (redness), swollen lymph nodes (lymphadenopathy), and the presence of cancerous T-cells (Sézary cells) in the bloodstream. This leukemic phase of CTCL causes severe itching and affects the entire body.
- Other subtypes: Include lymphomatoid papulosis, anaplastic large cell lymphoma, subcutaneous panniculitis-like T-cell lymphoma, and primary cutaneous peripheral T-cell lymphoma (unspecified).
Diagnostic Challenges in Lymphoma Diagnosis
- Skin Biopsy: To confirm a diagnosis of CTCL, doctors need to conduct a thorough evaluation using various methods. The main diagnostic procedure is a skin biopsy, which involves examining a small sample of tissue under a microscope to look for specific changes associated with lymphoma. Immunohistochemical staining techniques are used to identify certain markers on T-cells and determine if they are clonal (originating from a single abnormal cell).
- Blood Tests: May be done to check for circulating malignant cells, especially in cases of Sézary syndrome. Imaging studies such as CT scans or PET scans can help assess whether lymph nodes are involved or if there is any spread of disease beyond the skin.
- Multiple Biopsies: In some instances, multiple biopsies may be required to obtain sufficient tissue for accurate diagnosis. Dermatopathologists who specialize in studying lymphoproliferative disorders play a crucial role in interpreting these samples.
- Treatment Side Effects: Unfortunately, some patients may experience unexpected side effects from certain treatments, such as vision loss from medications like Mounjaro. If you or a loved one has faced such issues, it may be beneficial to explore potential legal avenues for compensation through a class action lawsuit. The Law Offices of Timothy L. Miles specializes in these types of cases and can provide assistance.
The Potential Link Between Dupixent and Cancer Risks
- Recent scientific investigations: Have revealed concerning patterns regarding Dupixent cancer risks among patients treated with dupilumab for inflammatory conditions.
- Dupixent and Cancer Risks: Multiple independent studies published between 2024 and 2025 have documented statistically significant associations between Dupixent use and the development of cutaneous T-cell lymphoma.
Emerging Evidence from Clinical Research
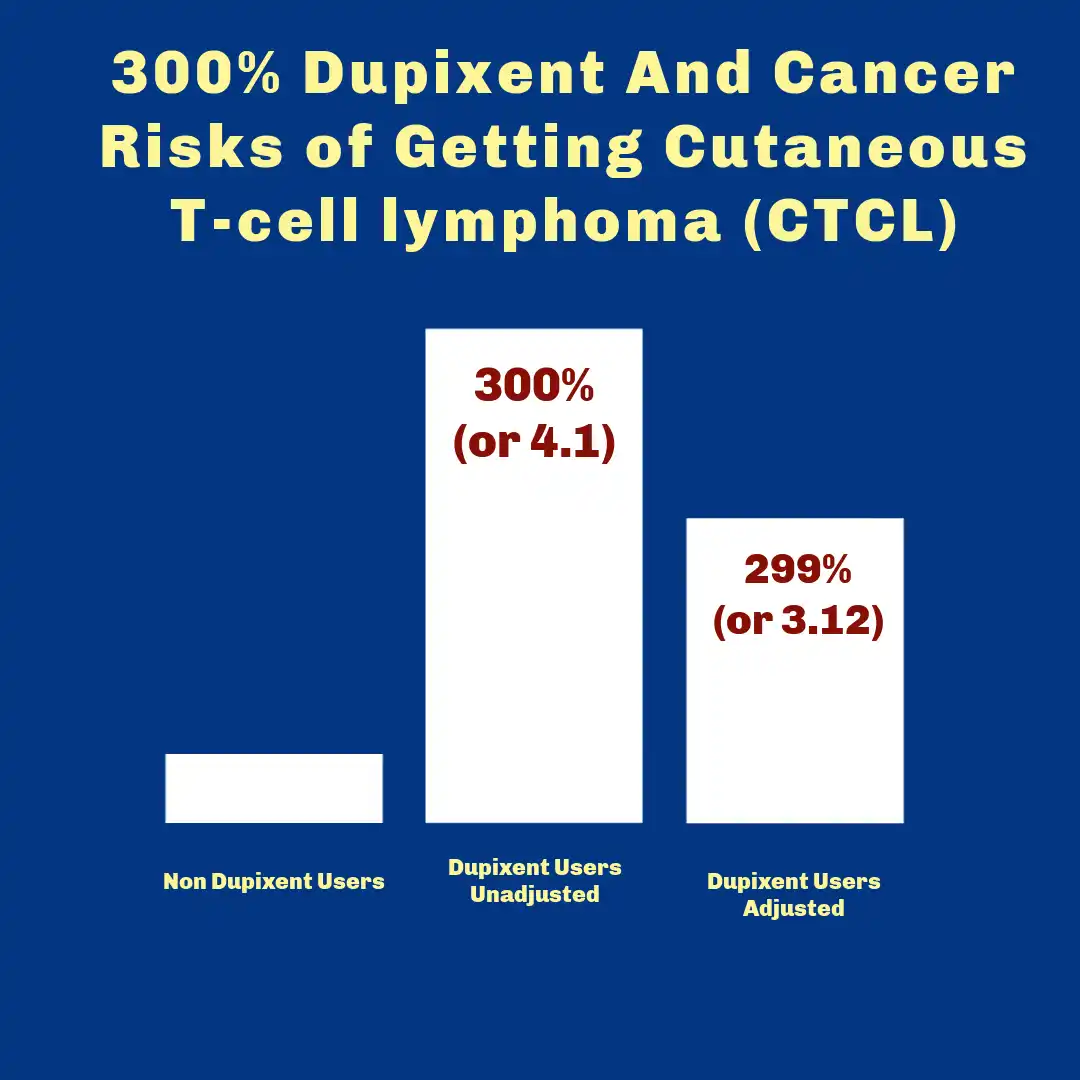
- Substantial Dupixent and Cancer Risks: An April 2024 study identified a substantial elevation in CTCL incidence among atopic dermatitis patients receiving dupilumab therapy. The adjusted analysis demonstrated that Dupixent increased CTCL risk by over 300 percent compared to patients not receiving the medication. This finding represented one of the first quantitative assessments of the potential malignancy risk associated with this biologic therapy.
- Dupixent and Cancer Risks Corroborated: Subsequent research in August 2024 corroborated these findings, revealing that Dupixent users were more than 4.5 times more likely to receive a CTCL diagnosis during treatment. The consistency across multiple independent investigations strengthened concerns about the medication’s safety profile regarding lymphoproliferative disorders.
- Clinical Research Expanded: A June 2025 study expanded the scope of investigation to asthma patients, demonstrating that individuals using Dupixent for respiratory conditions faced elevated CTCL risk compared to those receiving alternative asthma medications. This research, published in the European Respiratory Journal, suggested the cancer risk extended beyond dermatological applications to other approved indications.
Pharmacovigilance Data and Reporting Patterns
- FAERS Database: Analysis of the FDA Adverse Event Reporting System (FAERS) database through September 2025 revealed over 300 lymphoma diagnoses reported in association with Dupixent use.
- Significantly Higher Reporting Rates: A May 2025 analysis of this data indicated that Dupixent demonstrated approximately 30 times higher reporting rates for cancer compared to the average pharmaceutical product, representing a significant deviation from expected adverse event patterns.
Immunological Mechanisms and Cancer Surveillance
- Compromised Immune System: The dupilumab safety data raises questions about how the medication’s mechanism of action may compromise immune system function. Dupilumab functions as a monoclonal antibody targeting the interleukin-4 receptor alpha (IL-4Rα), thereby blocking signaling pathways mediated by both IL-4 and IL-13. These interleukin pathways serve critical regulatory functions in immune response coordination.
- Impaired Immune System: The blockade of IL-4 and IL-13 signaling may interfere with T-cell differentiation and function, processes essential for immune surveillance against malignant cells. T-cells represent the body’s primary defense mechanism for identifying and eliminating abnormal cellular proliferation, including early-stage lymphomas. Disruption of these pathways could theoretically impair the immune system’s capacity to detect and suppress malignant T-cell populations before they progress to clinically apparent CTCL.
Regulatory Status and Safety Updates on Dupixent
- Breakthrough Therapy Designation: The regulatory trajectory of dupilumab demonstrates the FDA’s progressive expansion of approved indications across multiple patient populations. The agency granted Breakthrough Therapy designation in November 2014, recognizing the medication’s potential to address unmet medical needs in atopic dermatitis treatment. This designation accelerated the development and review process, culminating in FDA approval for adult eczema treatment in March 2017.
- Subsequent regulatory actions: Expanded the medication’s approved use across diverse age groups and conditions:
-
- October 2018: Approval as add-on asthma treatment for eosinophilic or oral corticosteroid-dependent patients aged 12 and older
- March 2019: Authorization for moderate-to-severe atopic dermatitis in adolescents aged 12-17
- May 2020: Extension to children aged 6-11 with moderate-to-severe atopic dermatitis
- June 2022: Historic approval as the first biologic medicine for moderate-to-severe atopic dermatitis in children aged 6 months to 5 years
- January 2024: Label update for uncontrolled moderate-to-severe hand and foot atopic dermatitis
- FDA Deems Serious Risk: The regulatory landscape shifted significantly in March 2025 when the FDA placed Dupixent on the “Potential Signals of Serious Risks” list following accumulating reports of cutaneous T-cell lymphoma among treated patients. This designation indicates the agency’s active evaluation of potential regulatory actions regarding FDA label updates dupilumab cancer risks. The safety signal emerged from analysis of the FDA Adverse Event Reporting System (FAERS) database, which documented over 300 lymphoma diagnoses by September 2025.
- Regulatory Scrutiny Intensifies: A May 2025 analysis revealed that Dupixent and Cancer reporting rates exceeded the average drug by approximately 30-fold, intensifying regulatory scrutiny. The current product labeling contains no specific warnings regarding CTCL risk, representing a critical gap between emerging safety data and patient information. The FDA’s ongoing evaluation may result in mandatory label modifications, prescribing restrictions, or additional post-market surveillance requirements to address these safety concerns.

Clinical Trials and Long-Term Safety Data of Dupixent
The approval and widespread use of dupilumab is based on a large number of Phase III clinical trials that have studied its safety and effectiveness in various inflammatory conditions.
Atopic Dermatitis Trials
The LIBERTY AD clinical trial program showed significant improvements in disease severity, skin clearance, and itch reduction among patients with moderate-to-severe atopic dermatitis. Here are some key findings from these trials:
- Participants receiving dupilumab along with topical corticosteroids had better results compared to those receiving placebo treatments.
- The Phase 3 LIBERTY-AD-HAFT trial specifically looked at hand and foot atopic dermatitis, showing significant symptom improvement in patients treated with dupilumab compared to placebo.
Asthma Trials
Clinical trials studying dupilumab’s effectiveness in asthma patients established it as an effective additional treatment for those with eosinophilic or oral corticosteroid-dependent asthma. These studies found improvements in lung function, reductions in severe asthma attacks, and decreased need for systemic corticosteroids.
Pediatric Safety Data
Long-term safety data gathered from pediatric populations, including children aged six months to five years, supported the medication’s safety profile across different age groups.
Critical Limitations in Trial Design
The design and length of clinical trials have limitations when it comes to identifying rare side effects such as cutaneous T-cell lymphoma (CTCL). Here are some critical limitations:
- Selection Bias: Trial populations usually exclude patients with pre-existing cancers or significant immune system abnormalities, which may create a bias in selecting participants and hide cancer risks in real-world populations.
- Short Duration: Most Phase III trials last between 16 to 52 weeks, which is not enough time to detect slow-growing cancers like CTCL that may develop over months or years of exposure.
- Statistical Improbability: Clinical trials involve hundreds to thousands of participants, but rare events occurring at rates of 1 in 10,000 or less cannot be detected within these sample sizes. The absence of CTCL cases in pre-approval trials does not prove safety; instead, it shows the statistical unlikelihood of finding exceptionally rare events within controlled study environments.
After millions of patients receive treatment, post-marketing surveillance and real-world evidence become crucial for identifying such risks.
Legal Actions Related to Dupixent Cancer Claims
- Dupixent Cancer Lawsuit: The legal landscape surrounding Dupixent and cancer risks is still in its early stages. Individual lawsuits have been filed against the manufacturers, Sanofi and Regeneron Pharmaceuticals, in various jurisdictions across the United States.
- Dupixent and Cancer Allegations: These legal actions allege that the biologic medication caused or worsened the development of cutaneous T-cell lymphoma and other lymphomas in patients who used Dupixent as prescribed for inflammatory conditions.
Current Status of Individual Lawsuits
- Early Stages of Litigation: As of November 2025, there is no Dupixent class action lawsuit or Multi-District Litigation (MDL) established for Dupixent-related cancer claims. The lack of consolidated litigation is due to the unique nature of damages experienced by individual plaintiffs. Each case presents distinct medical histories, treatment timelines, and cancer progression patterns.
- Individual lawsuits: Continue to be filed in federal and state courts. Plaintiffs are seeking compensation for medical expenses, pain and suffering, and other damages related to their CTCL diagnoses.
- Additional Patient Information: For more comprehensive insights into ongoing legal cases and potential class action suits related to dangerous drugs like Dupixent, you might find this authoritative legal blog useful.
- Dupixent Lawsuit for Wrongful Death: A significant wrongful death lawsuit was filed in Tennessee in October 2025, highlighting the seriousness of the allegations against the manufacturers. The complaint claims that Dupixent sped up the T-cell lymphoma process, leading to rapid clinical decline and ultimately the patient’s death. This case emphasizes the potentially life-threatening consequences associated with Dupixent use.

Prospective Timeline for Future Legal Developments
Dupixent and Cancer Claims: The future legal developments for Dupixent cancer claims depend on several factors:
- The accumulation of additional cases
- Regulatory actions by the FDA
- Emerging scientific evidence
Dupixent and Cancer MDL: Attorneys involved believe that if the number of cases reaches a critical point, there may be a consolidation into an MDL. This would streamline pretrial proceedings and discovery processes.
FDA Designation May Surge Litigation: The FDA’s March 2025 designation of Dupixent as a “Potential Signals of Serious Risks” medication may lead to more lawsuits as patients and healthcare providers become more aware of the risks associated with CTCL.
Early Stages of Litigation: As of November 2025, there have been no settlements, recalls, or jury verdicts recorded for Dupixent cases. This indicates that litigation is still in its early stages, with ongoing discovery and motion practice.
Who is Eligible for a Dupixent Cancer Lawsuit?
To qualify for a Dupixent cancer lawsuit, individuals must meet specific criteria established through legal precedent and medical documentation requirements. The eligibility determination process requires comprehensive evidence linking Dupixent use to subsequent cancer diagnosis.
Primary Eligibility Requirements
The foundational requirement for pursuing legal action is documented prescription and administration of Dupixent. Patients must provide medical records demonstrating:
- Prescription history showing Dupixent treatment for atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyps, or eosinophilic esophagitis
- Pharmacy records confirming medication dispensation and refill patterns
- Healthcare provider documentation of treatment initiation dates and dosing protocols
- Insurance claims or payment records substantiating medication acquisition
Cancer Diagnosis Criteria
Dupixent Cancer Lawsuit Criteria: A confirmed diagnosis of cutaneous T-cell lymphoma or related lymphomas following Dupixent treatment initiation represents the critical medical element. Qualifying diagnoses include:
- Mycosis fungoides
- Sézary syndrome
- Lymphomatoid papulosis
- Anaplastic large cell lymphoma
- Other peripheral T-cell lymphomas affecting cutaneous tissue
Medical Confirmation: Diagnostic confirmation through skin biopsy, pathology reports, and oncological evaluation establishes the medical foundation for legal claims. The temporal relationship between Dupixent administration and cancer diagnosis requires documentation showing symptom onset or diagnosis occurred during or after treatment.
Statute of Limitations Considerations
The timeframe for filing legal action varies by jurisdiction. Individuals must initiate claims within their state’s prescribed statute of limitations period, typically calculated from the date of diagnosis or when the connection between Dupixent and cancer became apparent. Cases filed in Tennessee and other jurisdictions demonstrate the active pursuit of Dupixent lawsuit claims, though no Dupixent class action lawsuit has been established as of November 2025.
What Compensation Can Plaintiffs Expect in a Dupixent Cancer Lawsuit?
Compensation in a Dupixent cancer lawsuit includes various types of damages aimed at addressing the overall impact of a cutaneous T-cell lymphoma diagnosis after receiving dupilumab treatment. Plaintiffs taking legal action against Sanofi and Regeneron may seek compensation for both financial and non-financial losses resulting from their condition.
Economic Damages
The financial burden of cancer treatment forms the foundation of economic damages in these cases. Plaintiffs may recover:
- Medical expenses for CTCL diagnosis, including skin biopsies, blood tests, and imaging studies
- Ongoing cancer treatment costs encompassing topical therapies, phototherapy, radiation, chemotherapy, or systemic medications
- Future medical care required for disease management and monitoring
- Lost wages resulting from inability to work during treatment periods
- Reduced earning capacity when the disease or its treatment permanently affects employment prospects
Non-Economic Damages
Compensation in Dupixent cancer lawsuit claims extends beyond quantifiable financial losses. Non-economic damages address:
- Pain and suffering from the physical manifestations of CTCL, including persistent itching, skin lesions, and treatment side effects
- Emotional distress associated with cancer diagnosis, disease progression, and uncertainty regarding prognosis
- Mental anguish resulting from potential misdiagnosis or delayed recognition of CTCL symptoms
- Loss of quality of life due to chronic symptoms and treatment requirements
- Disfigurement from visible skin manifestations characteristic of cutaneous lymphomas
Punitive Damages
In cases demonstrating egregious conduct by pharmaceutical manufacturers—such as failure to adequately warn about known risks or concealment of safety data—courts may award punitive damages to penalize defendants and deter similar behavior.

How to Start a Dupixent Cancer Lawsuit?
Starting a Dupixent Cancer Lawsuit requires strategic legal guidance from attorneys experienced in pharmaceutical litigation. The complexity of proving that dupilumab treatment caused cutaneous T-cell lymphoma to develop requires experience and knowledge of both medical evidence and product liability law.
Essential Steps in the Legal Process
The pathway to pursuing a Dupixent Cancer Lawsuit begins with securing a free case evaluation Dupixent lawyer consultation. During this initial assessment, attorneys examine the specific circumstances of each case to determine whether the individual may qualify for a Dupixent Cancer Lawsuit. This evaluation involves:
1. Medical Documentation Assembly
- Comprehensive prescription records documenting Dupixent administration dates and dosages
- Pathology reports confirming CTCL diagnosis with biopsy results
- Treatment timelines establishing temporal relationship between dupilumab use and cancer development
- Physician notes detailing symptom progression and diagnostic findings
2. Evidence Preservation and Analysis
Specialized Dupixent Cancer Lawyer teams conduct thorough investigations into the medical history, examining whether warning signs were present and adequately communicated. This process includes securing expert medical testimony to establish the connection between dupilumab exposure and lymphoma development.
3. Statute of Limitations Considerations
Time constraints vary by jurisdiction, making prompt action essential. Legal counsel evaluates applicable deadlines based on the state where treatment occurred and where the plaintiff resides. Delays in seeking Dupixent lawyer contact information may result in forfeited legal rights. Contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation. (855) 846–6529 or [email protected].
4. Case Filing and Litigation Strategy
Attorneys prepare comprehensive complaints detailing allegations against Sanofi and Regeneron, including failure to warn claims and design defect theories. The legal team manages all procedural requirements, discovery processes, and negotiations with pharmaceutical company defense counsel.
However, it’s not just Dupixent that has been linked to severe health issues. For instance, if you’ve taken Zepbound, another medication, and developed vision problems as a result, you may want to consider seeking legal advice regarding a potential Zepbound vision loss lawsuit.
Conclusion
The evolving understanding of Dupixent and Cancer necessitates heightened vigilance among patients, healthcare providers, and regulatory authorities. The dupilumab safety profile summary continues to develop as post-marketing surveillance reveals potential associations between treatment and cutaneous T-cell lymphoma that were not fully apparent during initial clinical trials.
Patients currently receiving dupilumab therapy bear responsibility for monitoring side effects dupilumab users may experience, particularly dermatological manifestations that deviate from expected treatment responses. Any persistent or unusual skin changes—including unexplained rashes, plaques, nodules, or erythroderma—warrant immediate medical evaluation rather than dismissal as disease progression or treatment-related irritation.
Proactive engagement between patients and medical professionals remains essential for balancing therapeutic benefits against potential risks in dupilumab therapy.
FREQUENTLY ASKED QUESTIONS THE DUPIXENT LAWSUIT
What are the potential benefits of a Dupixent Cancer Lawsuit?
How long will the Dupixent Cancer Lawsuit take to resolve?
What Is Cutaneous T-cell lymphoma?
-
What it is: A type of non-Hodgkin lymphoma that starts in T-cells in the skin.
-
Common symptoms:
-
-
Itchy, scaly patches or plaques
-
-
-
Redness on the skin
-
-
-
Thicker, raised lesions or tumors
-
-
-
Sometimes, the skin redness can cover a large portion of the body, a condition called erythroderma
-
-
Progression: CTCL is often slow-growing, but some types can be aggressive and spread to other parts of the body, such as the lymph nodes, blood, or organs like the liver or lungs.
-
Diagnosis: Diagnosis can be challenging due to similar-looking symptoms and often involves a skin biopsy, physical exam, blood tests, and imaging.
-
Treatment: Treatments are chosen based on the stage of the cancer and can include creams, light therapy, radiation, or systemic treatments like chemotherapy or other medications.
How can I join the Dupixent Lawsuit?
Is There a Dupixent Class Action Lawsuit?
What is cutaneous T-cell lymphoma (CTCL)?
How many different types of CTCL are there?
-
Mycosis Fungoide (MF) – The most common type, characterized by patches, plaques, and tumors on the skin.
-
Sézary Syndrome (SS) – A more aggressive form of MF that affects the blood as well as the skin.
-
Lymphomatoid papulosis – A benign form of CTCL that causes small, itchy bumps on the skin.
-
Granulomatous slack skin – A rare, indolent form of CTCL that causes loose, pendulous folds of skin.
-
Anaplastic large cell lymphoma – An aggressive form of CTCL that can spread to other organs.
-
Extranodal NK/T-cell lymphoma: A rare type of CTCL that affects the skin and lymph nodes.
-
Extranodal NK/T-cell lymphoma, nasal type – A rare, aggressive lymphoma that can affect the skin, among other places.
-
Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) – A rare, slow-growing type that causes nodules deep in the fatty layer of the skin, most often on the legs.
-
Primary cutaneous peripheral T-cell lymphoma (unspecified) – This is a category for rare entities that do not fit into other classifications.

