Introduction to a Patient Reference Guide to Dupixent and Cancer Risk
- Dupixent and Cancer Risk: Numerous lawsuits have been filed against Sanofi and Regeneron, the makers of Dupixent, alleging the company failed to adequately warn patients and doctors about a potential link between the drug and the development of cutaneous T-cell lymphoma (CTCL).
- Dupixent (dupilumab): Is an injectable biologic medication that is used to treat particular types of inflammatory diseases. The medicine is currently the subject of a growing number of lawsuits alleging it causes or accelerates the development of cutaneous T-cell lymphoma (CTCL), a rare form of cancer. As of November 2025, the litigation is in early stages, with no settlements or jury verdicts yet.
- Recent studies on Dupixent and Cancer: Have shown that Dupixent use may increase the risk of certain types of cancers known as Cutaneous T-cell Lymphomas (CTCL), including its subtypes Mycosis fungoides and Sezary syndrome, by as much as 300% or more in people who used the medication.
- Potential Compensation: People who used Dupixent and were later diagnosed with CTCL may be eligible to file a Dupixent Cancer Lawsuit and potential entitled to substantial compensation. If you suffered Dupixent and Cancer call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation. (855)-846-6529 or [email protected].
THE DUPLXENT COMMON SIDE EFFECCTS PROFILE
Summary of Key Information on Dupixent and Cancer Risk
- Type of Cancer: The primary concern is an increased risk of developing cutaneous T-cell lymphoma (CTCL), a rare non-Hodgkin lymphoma that affects the skin. Subtypes such as mycosis fungoides and Sézary syndrome have also been reported.
- Increased Dupixent and Cancer Risk Factor: One 2024 study published in the Journal of the American Academy of Dermatology found that atopic dermatitis patients treated with Dupixent were more than four times (4.1-fold) more likely to develop CTCL compared to patients who did not use the drug. Another study suggested that Dupixent users reported CTCL 30 times more often than users of all other medications in FDA safety data.
- Second Dupixent and Cancer Study: A second study in 2024, published in Dermatologic Therapy, similarly reported an increased risk of CTCL among Dupixent users. The study alson noted that the risk was highest within the first year of treatment, and people over 60 were more susceptible.
- Mechanism: Dupixent works by blocking two immune system messengers, IL-4 and IL-13, to reduce inflammation. Researchers hypothesize this mechanism might allow malignant T-cells to grow unchecked or “unmask” a pre-existing, slow-growing lymphoma that was initially misdiagnosed as severe eczema.
- FDA and Label Warnings: The U.S. Food and Drug Administration (FDA) has not added a specific cancer warning to the Dupixent label as of late 2025. The current label mentions other side effects but no mention of cancer risk.
- Dupixent Lawsuits: Manufacturers Sanofi and Regeneron are facing lawsuits alleging they failed to adequately warn patients and physicians about the potential CTCL risks.
- Other Cancers: While the link to CTCL is the primary concern, some studies have noted individual cases of other solid tumors; however, a clear association for other cancers has not been widely established, with some research suggesting no significant negative effects on most other tumor types.

Recommendations for Patients
- Do not stop taking Dupixent without consulting your doctor:. The benefits of managing conditions like severe eczema and asthma may outweigh the potential risks, but a medical professional should make that assessment.
- Be Cognizant of Warning Signs: Patients currently using Dupixent should watch for warning signs and discuss any concerns with their healthcare provider:
-
- Skin rashes that do not heal or are atypical.
- Unexplained lumps or thickened skin patches.
- Swollen lymph nodes.
- Hair loss (in the context of new skin changes).
- Increase Skin Examinations: Requesting more frequent skin examinations may be advisable for those with pre-existing risk factors or concerns.
The Dupixent Mechanism of Action
- Targeted Biologic Therapy: Dupixent (dupilumab) is a targeted biologic therapy that works by blocking the signaling of two key proteins, or cytokines, in the immune system: interleukin-4 (IL-4) and interleukin-13 (IL-13).
- Blocks Two Key Proteins: These interleukins are central drivers of type 2 inflammation, which is the underlying cause of conditions like severe asthma, atopic dermatitis (eczema), and chronic rhinosinusitis with nasal polyps.
Key Aspects of the Mechanism of Action
- Monoclonal Antibody: Dupilumab is a fully human monoclonal antibody (IgG4 subclass) designed to specifically target a particular part of the body’s inflammatory response.
- Receptor Antagonist: It acts as an interleukin-4 receptor alpha (IL-4Rα) antagonist. Dupixent binds to this specific subunit, which is a shared component of two different types of receptors found on various cells throughout the body:
-
- Type 1 Receptor: Found mainly on immune cells like mast cells, basophils, eosinophils, and T cells. IL-4 typically binds to this receptor.
-
- Type 2 Receptor: Found on epithelial cells, smooth muscle cells, and fibroblasts in tissues like the lungs and skin. Both IL-4 and IL-13 bind to this receptor.
- Dual Blockade: By binding to the shared IL-4Rα subunit, Dupixent effectively prevents both IL-4 and IL-13 from attaching to their respective receptors and initiating inflammatory signaling.
- Inhibition of Inflammatory Responses: This dual blockade stops the downstream signaling cascade that normally leads to the symptoms of type 2 inflammation. The effects include:
-
- Reduced release of pro-inflammatory substances like chemokines and nitric oxide.
- Decreased production of immunoglobulin E (IgE), an antibody involved in allergic reactions.
- Inhibition of eosinophil (a type of white blood cell) trafficking and infiltration into inflamed tissues.
- Reduced mucus overproduction and smooth muscle contraction in the airways.
- The Impact: This targeted action helps to “calm” an overreactive immune system and mitigate the chronic inflammation responsible for the symptoms of associated diseases, without causing broad immunosuppression. By targeting the common pathway, Dupixent can treat multiple type 2 inflammatory conditions simultaneously.
Most Common Side Effect of Dupixent on the Eyes
Most Common Side Effect: The most common side effect of Dupixent on the eyes is conjunctivitis (pink eye). Other potential eye-related side effects include dry eyes, eyelid inflammation (blepharitis), and corneal inflammation (keratitis). These issues occur most frequently in patients being treated for atopic dermatitis (eczema)
Common Ocular Side Effects
Most eye problems associated with Dupixent are mild to moderate and can be managed with treatment, often allowing the patient to continue using the medication.
Conjunctivitis: This is the most frequently reported side effect, affecting as many as 1 in 4 patients in some studies.
-
- Symptoms: Redness, itching, a gritty feeling (like a foreign body is in the eye), watery or sticky discharge, and swollen eyelids.
- Dry Eye: This can cause dryness, burning, and irritation. The drug blocks a protein (IL-13) that helps maintain mucus production and the stability of the tear film, leading to dryness and irritation.
- Blepharitis: Inflammation along the edges of the eyelids
-
- Symptoms: Swollen eyelids, a burning/gritty sensation, flaking or crusting on the eyelashes, and sticky eyelids upon waking.
- Keratitis: Inflammation of the cornea (the clear front surface of the eye).
-
- Symptoms: Eye pain, blurred vision, light sensitivity (photophobia), a sensation that something is in the eye, and potentially a visible white spot on the cornea in severe cases.
- Blurred Vision: A general complaint that can be linked to any of the above conditions.
- Eye Pruritus: Severe itching of the eyes
Rare but Serious Complications
While less common, more severe issues have been reported and require urgent medical attention.
- Corneal Ulceration/Perforation: In rare cases, severe inflammation can lead to ulcers or perforation of the cornea, which can threaten vision.
- Punctal Stenosis: The narrowing of the tear drainage ducts, leading to excessive tearing.
- Cicatricial Conjunctivitis: A chronic condition leading to scarring and potential vision impairment
When to Seek Medical Help
It is important to promptly report any new or worsening eye symptoms to your healthcare provider. They may refer you to an ophthalmologist (eye specialist) for an exam.
Seek urgent ophthalmology evaluation if you experience:
- Significant eye pain that doesn’t go away.
- Sudden changes in vision or vision loss.
- Intense light sensitivity.
- Visible damage or an opaque spot on the cornea.

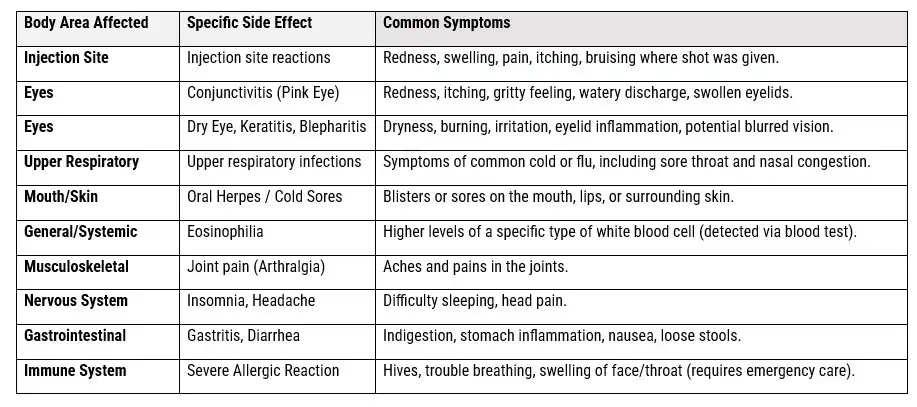
Other Common Side Effects (Non-Ocular)
The most frequent side effects that are not related to the eyes include:
- Injection site reactions: This is the most common side effect, involving redness, swelling, pain, itching, or bruising at the site of the injection.
- Upper respiratory tract infections: Symptoms similar to the common cold or flu (nasopharyngitis).
- Oral herpes: An increased risk of developing cold sores on the mouth or lips.
- Oropharyngeal pain: Pain in the mouth or back of the throat.
- Eosinophilia: Higher than normal levels of a certain type of white blood cell (eosinophils) in the blood.
- Joint pain (arthralgia): This can range from mild aches to severe pain that may affect mobility.
- Insomnia: Trouble sleeping, reported more commonly in patients with chronic rhinosinusitis with nasal polyps.
How to Manage Ocular Side Effects
- Mild Dubixent Side Effects: These side effects of Dupixent are mainly mild, resolve on their own, and are bit critical enough to lead to stopping the medication. Neverthesee, in certain instances, additional treatment may become necessary to manage Dupixent side effects.
- Management of Dupixent side effects to the Eyes: The management of vision Dupixent side effects depends on the severity of the condition. Mild cases may only need over-the-counter (OTC) and home remedies, such as:
-
- artificial tears (eye drops to improve the eye’s moisture)
- hyaluronic eye drops
- cold compresses
- warm compresses
- antihistamines to manage inflammation
- gentle eyelid scrubs for blepharitis
The Basis for a Dupixent Cancer Lawsuit
- Primary Basis Dupixent and Cancer Lawsuits: The central claim in the lawsuits is “failure to warn“. Plaintiffs argue that the manufacturers knew or should have known about the potential link between Dupixent and CTCL based on emerging medical studies and adverse event reports, yet did not update the product’s warning label.
- Key Points: Critical points supporting the litigation include:
-
- Medical Studies: Multiple peer-reviewed studies published in 2024 and 2025 have suggested an increased risk of CTCL among Dupixent users. Some studies reported that Dupixent patients had a 300% to 450% higher risk of developing CTCL compared to non-users.
-
- FDA Investigation: In March 2025, the U.S. Food and Drug Administration (FDA) identified CTCL as a “potential signal of a serious risk” associated with Dupixent and opened a formal safety investigation. The FDA is currently evaluating whether to require a label change.
-
- Masking Symptoms: CTCL often initially appears as skin patches that can be mistaken for eczema. Lawsuits allege that Dupixent’s mechanism of action may “unmask” a pre-existing, undiagnosed lymphoma or accelerate its progression, delaying proper cancer diagnosis and treatment.
-
- First Wrongful Death Suit: A wrongful death lawsuit was filed in October 2025 by the family of a Tennessee woman who died from T-cell lymphoma shortly after starting Dupixent treatment, marking a significant development in the litigation.
Who Can Qualify for a Dupixent Cancer Lawsuit?
- Eligibility: Individuals who used Dupixent and were later diagnosed with cutaneous T-cell lymphoma (CTCL) or related T-cell lymphomas (such as mycosis fungoides or Sézary syndrome) or other severe Dupixent side effects may be eligible to file a personal injury or wrongful death lawsuit.
- Contact Dupixent Cancer Lawyer Timothy L. Miles: If you or a loved one developed CTCL after using Dupixent, or suffered other severe Dupixent side effects it is recommended to consult with a personal injury attorney promptly, as state laws impose strict deadlines (statutes of limitations) for filing a claim. If you suffered Dupixent and Cancer call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation. (855)-846-6529 or [email protected].



