Introduction to Saxenda Eye Problems
- Saxenda Eye Problems: Refers to significant developments with respect to potential ocular convolutions connected to Saxenda use. Reports of serious Saxenda vision side effects, including vision loss and blindness, have resulted in a wave of lawsuits against the manufacturer. These concerns have coalesced into the Saxenda Blindness Lawsuit, with patients seeking representation from legal professionals, including Saxenda Blindness Lawyers, to address alleged inadequate warnings regarding severe and permanent oscular-related risks.
- Saxenda: Is an important breakthrough in the field of medication for long-term weight management. It has been approved by the U.S. Food and Drug Administration (FDA) as a prescription drug for adults who are obese or overweight and have weight-related health issues. The active ingredient in Saxenda is liraglutide, a man-made version of a hormone that acts on the body’s receptors to regulate metabolism and promote weight loss.
- FDA Approved for Weight Loss: Liraglutide works by imitating natural hormones that control hunger and food consumption, making Saxenda an effective option for individuals who have difficulty managing their weight through traditional methods such as dieting and exercising. The FDA granted approval for Saxenda after thorough clinical trials showed its effectiveness in achieving long-term weight loss when used in conjunction with calorie-restricted diets and increased physical activity.
- Saxenda Vision Side Effects: Recent studies and feedback from patients have highlighted the urgent need to understand the potential Saxenda Vision Side Effects. Saxenda eye problems have become a major concern among healthcare professionals, with reported cases of severe visual complications requiring immediate medical attention.
- Be Cognizant of Saxenda Eye Problems: It is crucial for patients taking this weight loss medication to be aware of possible Saxenda vision side effects, as early detection and intervention are vital in preventing irreversible vision damage. The connection between liraglutide use and specific eye disorders calls for vigilant self-monitoring by patients and regular check-ups by healthcare providers throughout the course of treatment.
![Saxenda Eye Problems: Patient Reference Guide [2025]](https://classactionlawyertn.com/wp-content/uploads/2025/11/saxenda-eye-problems-a-patients-guide.webp)
Understanding Saxenda and How It Works
- Mechanism of Action: Saxenda works by targeting the body’s natural system for regulating hunger through a complex biological process. The active ingredient in Saxenda, liraglutide, is a type of medication known as a GLP-1 receptor agonist.
- Mimics GLP-1: It mimics the action of glucagon-like peptide-1 (GLP-1), a hormone that is naturally produced in the intestines after we eat. This imitation effect activates specific receptors throughout the body, especially in the pancreas, brain, and digestive system.
How Liraglutide Works to Control Appetite
- Receptor Agonist: Liraglutide works by attaching itself to GLP-1 receptors in the hypothalamus, which is the part of the brain responsible for controlling appetite and signaling when we are full. When liraglutide binds to these receptors, it triggers a series of responses in the nervous system that:
-
- Decrease feelings of hunger by altering signals related to appetite
-
- Increase sensations of fullness after consuming smaller meals
-
- Reduce cravings for food between meals
-
- Influence reward pathways associated with eating behavior
- Modified Molecular Structure: The medication achieves these effects by continuously activating the receptors, ensuring that therapeutic levels remain in the bloodstream for long periods. This is made possible by its modified molecular structure that prevents it from being broken down quickly by enzymes.
How Saxenda Affects Digestion and Stomach Emptying
- Delayed Gastric Emptying: One of the keyways Saxenda helps with weight management is by slowing down the emptying of the stomach. When food stays in the stomach for a longer time before moving into the small intestine, it prolongs the feeling of fullness after meals and leads to reduced calorie intake.
- This change in how quickly food moves through the digestive system is directly linked to some gastrointestinal issues reported by patients using Saxenda. The most common side effects include:
-
- Nausea, especially during the early stages of treatment
-
- Constipation due to slower movement of waste through the intestines
-
- Discomfort or bloating in the abdomen
-
- Diarrhea as the digestive system adapts to new patterns of movement
Effects Beyond Digestion: How Saxenda Impacts Other Body Systems
- Saxenda Effects on Bodily Functions: GLP-1 receptors are found throughout various parts of our body, not just in areas related to appetite control and digestion. This means that liraglutide can also have effects on other systems such as cardiovascular healtheffects on other systems such as cardiovascular health, kidney function, and eye structures.
- Impacts Blood Flow: The same activation of these receptors that leads to weight loss can also impact blood flow, inflammation responses, and metabolism in different organs. Therefore, it is important for healthcare providers to closely monitor patients receiving Saxenda treatment since side effects may occur in areas unrelated to weight management or digestion.
Overview of Eye Problems Associated with Saxenda
Saxenda Eye Problems: Patients prescribed Saxenda for weight management have reported various Saxenda vision side effects that require careful attention and monitoring throughout treatment. The Saxenda eye side effects observed in clinical studies and post-marketing surveillance show a range of vision issues from mild temporary changes to severe conditions needing immediate medical intervention.
Documented Vision Issues
Saxenda Vision Problems: The vision problems with Saxenda include multiple forms of visual impairment affecting different aspects of eye function. Clinical reports and patient testimonials have identified several distinct patterns of vision problems associated with liraglutide therapy:
- Blurred vision is the most common complaint, characterized by reduced visual clarity and difficulty focusing on objects at varying distances
- Tunnel vision presents as a narrowing of the peripheral visual field, limiting the patient’s ability to see objects outside their direct line of sight
- Eye pain or swelling manifests as discomfort, pressure sensations, or visible inflammation around the eye structures
- Halos around lights appear as bright circles or rings surrounding light sources, particularly noticeable during nighttime or low-light conditions
- Sudden vision changes include rapid onset of visual distortion, partial vision loss, or complete blindness in one or both eyes
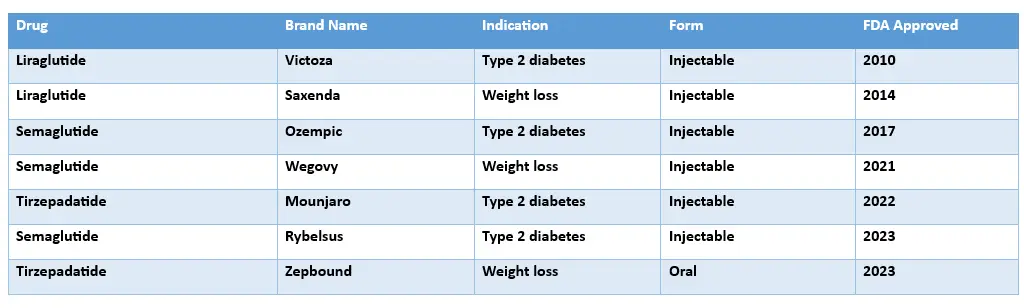
OZEMPIC VS. TRULICITY VS. MOUNJARO V. SAXENDA
|
MEDICATION |
TYPE | ACTIVE INGREDIENT | HOW OFTEN TO TAKE IT? | PURPOSE |
|
Ozempic |
Injection | Semaglutide | Weekly |
Type 2 diabetes treatment |
|
Trulicity |
Injection | Dulaglutide | Weekly |
Type 2 diabetes treatment |
|
Mounjaro |
Injection | Tirepatide | Weekly |
Weight loss and Type 2 diabetes treatment |
| Saxenda | Injection | Liraglutide | Daily |
Weight loss |
Prevalence and Reporting Patterns
- Saxenda Blindness Lawsuit: The Saxenda vision side effects have been documented through various channels, including adverse event reporting systems, clinical studies, and direct patient communications with healthcare providers.
- Diverse Symptom Profile: While the exact incidence rates vary across different populations, the consistency of reported symptoms across diverse patient groups has prompted increased scrutiny from medical professionals and regulatory bodies.
Critical Importance of Early Detection
- Saxenda Vision Side Effects: Recognition of adverse reactions of Saxenda affecting vision requires vigilant self-monitoring by patients and systematic assessment by healthcare providers. The timing between starting the medication and symptom onset varies significantly among individuals, with some experiencing immediate changes while others develop complications after prolonged use.
- Early Identification of Saxenda vision problems: Is crucial in preventing permanent eye damage. Patients who quickly report visual changes enable healthcare providers to carry out appropriate diagnostic evaluations, adjust treatment plans, or stop medication before irreversible harm occurs. The opportunity for effective intervention becomes much smaller as certain conditions worsen, making immediate recognition and response vital parts of safe Saxenda therapy.
- Record Ocular Changes: Recording any visual changes, no matter how minor they may seem, creates an important medical record that supports both clinical management and potential legal considerations if complications arise.
Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION) and Saxenda
- Saxenda Eye Problems: Non-arteritic anterior ischemic optic neuropathy represents the most severe ocular complication associated with Saxenda use. This condition occurs when blood flow to the optic nerve becomes compromised, resulting in sudden damage to the nerve tissue responsible for transmitting visual information from the eye to the brain. Unlike arteritic forms of optic neuropathy caused by inflammatory conditions, NAION develops without underlying arterial inflammation, making it particularly challenging to predict and prevent.
- The pathophysiology of NAION involves acute ischemia to the anterior portion of the optic nerve head, leading to swelling and subsequent atrophy of nerve fibers. This damage typically manifests unilaterally, though bilateral involvement can occur in approximately 15% of cases over time. The condition ranks as the second most common optic neuropathy in adults over 50 years of age, with certain risk factors including hypertension, diabetes, and sleep apnea increasing susceptibility.

Quantified Risk Associated with Liraglutide
- Severe Saxenda Vision Side Effects: Recent epidemiological data demonstrates a statistically significant elevation in NAION risk among patients utilizing liraglutide-based medications such as Saxenda. Research indicates an approximate 25% increased risk of developing non-arteritic anterior ischemic optic neuropathy in individuals prescribed this GLP-1 receptor agonist compared to those not using the medication. This elevated risk profile necessitates heightened vigilance from both prescribing physicians and patients throughout the treatment duration.
- Link to Optic Nerve Damage: The mechanism linking liraglutide to optic nerve damage remains under investigation, though hypotheses include alterations in microvascular perfusion, changes in intraocular pressure regulation, and potential effects on autoregulatory mechanisms within the optic nerve head circulation.
Clinical Manifestations of NAION
- NAION Symptoms: Patients experiencing NAION typically present with characteristic symptoms requiring immediate medical evaluation:
-
- Sudden, painless vision loss in one eye, often noticed upon awakening
- Altitudinal visual field defects, where vision loss affects either the upper or lower half of the visual field
- Reduced color perception or color desaturation in the affected eye
- Relative afferent pupillary defect (RAPD), observable during clinical examination
- Optic disc edema with associated hemorrhages visible on fundoscopic examination
- Persistent visual impairment that does not improve with corrective lenses
- Ocular Disturbances: The onset of symptoms typically occurs acutely, with maximal vision loss developing within hours to days. Unlike transient visual disturbances, NAION-related vision loss demonstrates permanence, with limited potential for spontaneous recovery.
- Saxenda Eye Problems: Patients may describe the visual loss as a “curtain coming down” or a dark shadow obscuring portions of their visual field, distinguishing this presentation from other Saxenda eye problems such as temporary blurred vision or refractive changes.
COMPARISON OF GLP-1 DRUG PROFILES
Recognizing Symptoms and When to Seek Medical Attention
- Monitor Vision: If you are starting or continuing Saxenda treatment, it’s important to have a plan in place for monitoring your vision.
- Early Detection: This will help you catch any potential eye problems early on. During the first six months of treatment, when the risk of complications is higher, make sure to check your eyesight regularly.
How to Assess Your Vision
- Self-Assessment: To assess your vision accurately, follow these steps:
-
- Cover one eye with your hand or an eye patch.
- Look at an object straight ahead with the uncovered eye.
- Pay attention to any changes in clarity, color perception, or field of vision.
- Repeat this process with the other eye.
- Identify Changes in Vision: By doing this self-assessment regularly, you’ll be able to identify any patterns or changes in your vision that may require medical attention.
Keeping Track of Your Visual Experiences
- Keep a Written Log: In addition to self-assessment, it’s also helpful to keep a written log of your visual experiences. This can include any unusual symptoms you notice, such as blurred vision or difficulty seeing at night. By documenting these experiences, you’ll have a clearer picture of your visual health and be better prepared for discussions with your healthcare provider.
Tip: Use a notebook or a digital app to record your visual experiences. Be as specific as possible about when and how often these symptoms occur.
When to See an Eye Doctor
- Seek Immediate Medical Attention: While regular self-assessments and logging of visual experiences are important, there are certain situations where you should seek immediate medical attention from an ophthalmologist. These critical symptoms include:
-
- Sudden loss of vision or significant decrease in eyesight in one or both eyes
- Dark spots, shadows, or curtain-like obstructions in your field of vision
- Blurred vision that doesn’t go away with blinking or resting
- Halos, rainbow-colored rings, or sensitivity to light
- Eye pain accompanied by redness, swelling, or discharge
- Flashing lights or floating objects that become more frequent
- Promptly Seek Medical Intervention: If you experience any of these symptoms while taking Saxenda, don’t delay—make an appointment with an eye doctor right away.
The Role of Healthcare Providers
- Thorough Examination: As a patient on Saxenda, it’s not just up to you to monitor your eye health—your healthcare providers also play a crucial role in this process. Before starting Saxenda treatment, make sure that your prescribing a thorough eye examination. This is especially important if you have pre-existing risk factors such as diabetes, high blood pressure (hypertension), or cardiovascular disease.
- Get Regular Follow-Up Care: Regular follow-up appointments every three months during the first year can help detect any subtle changes in your eyesight before they become irreversible. It is also essential for healthcare providers to document visual function throughout Saxenda treatment. This creates important medical records that can be used for both clinical management and potential legal purposes if any adverse outcomes occur.
- Comprehensive Assessment: By working together—patients actively monitoring their own vision and healthcare providers conducting comprehensive assessments—we can ensure better eye health outcomes for those using Saxenda.
Legal Issues Surrounding Saxenda Eye Problems and Vision Loss
- Saxenda Eye Problems: Patients who have experienced vision loss or blindness while using Saxenda have initiated legal action against Novo Nordisk, the pharmaceutical manufacturer. These Saxenda blindness lawsuits center on allegations that the company failed to provide adequate warnings about the serious risks of vision-related complications, particularly NAION, associated with liraglutide use. Plaintiffs assert that had they been properly informed of these risks, they would have made different treatment decisions or pursued alternative weight management options.
- Saxenda Vision Loss: The consolidation of multiple cases has resulted in the establishment of multidistrict litigation MDL 3094, which centralizes pretrial proceedings for efficiency and consistency. This MDL encompasses claims related to Saxenda vision loss and other serious adverse effects linked to GLP-1 receptor agonists.
- MDL Coordination: The centralization allows for coordinated discovery, expert testimony, and the development of consistent legal standards across numerous individual cases, streamlining what would otherwise be a fragmented litigation landscape across multiple jurisdictions.
- Saxenda Blindness Lawyer: Pharmaceutical injury litigation requires experience and extensive knowledge due to the complex medical and regulatory issues involved. Attorneys experienced in this field, such as Timothy L. Miles, possess the necessary knowledge about the drug approval processes, post-market surveillance obligations, and manufacturer liability.
- Saxenda Eye Problems: These lawyers understand the scientific evidence required to establish causation between medication use and adverse outcomes, as well as the regulatory framework governing pharmaceutical warnings and disclosures.
- Saxenda Vision Side Effects: The legal action against Novo Nordisk represents a significant development in pharmaceutical accountability, particularly regarding the transparency of risk communication for medications approved for chronic use in otherwise healthy individuals seeking weight management.
- Saxenda Vision Loss: These cases examine whether the manufacturer conducted adequate post-market surveillance, appropriately updated prescribing information as new safety data emerged, and fulfilled its duty to warn both healthcare providers and patients about evolving risk profiles associated with long-term liraglutide therapy.
Eligibility and Process for Filing a Saxenda Eye Side Effects Lawsuit
- Saxenda Lawsuit Eligibility: depends on several specific criteria that establish a direct connection between the medication and resulting vision complications. Patients who developed Saxenda eye problems including non-arteritic anterior ischemic optic neuropathy (NAION), significant vision loss, or blindness while using the medication may qualify to pursue legal action.
- Saxenda Eye Problems: The timing of symptom onset relative to Saxenda use proves critical, as documented medical records demonstrating vision deterioration during or shortly after treatment strengthen eligibility. Patients must provide comprehensive medical documentation including ophthalmological examinations, diagnostic imaging results, and treatment records that establish the causal relationship between liraglutide exposure and ocular damage.
- Saxenda Eye Problems: The legal process for pursuing compensation for vision loss from Saxenda follows a structured pathway designed to establish liability and quantify damages. Initial case evaluation involves consultation with a Saxenda blindness lawyer who reviews medical records, treatment history, and the extent of visual impairment.
- Saxenda Vision Side Effects: The attorney conducts a thorough investigation to determine whether Novo Nordisk failed to provide adequate warnings about vision-related risks associated with the medication. This investigation includes analysis of clinical trial data, FDA communications, and expert medical testimony regarding the manufacturer’s knowledge of potential ocular complications.
Finding a lawyer experienced in pharmaceutical cases requires careful consideration of specific qualifications and experience. Prospective clients should seek attorneys with:
-
- Demonstrated expertise in pharmaceutical injury litigation and product liability law
-
- Track record of successful outcomes in cases involving prescription medication adverse effects
-
- Resources to conduct extensive medical and scientific research supporting complex pharmaceutical claims
-
- Membership in professional organizations focused on pharmaceutical litigation
-
- Access to medical experts who can provide credible testimony regarding causation and damages
The litigation process typically operates on a contingency fee basis, meaning attorneys receive compensation only upon successful resolution of the claim. This arrangement eliminates upfront costs for patients pursuing justice while ensuring legal representation remains accessible to individuals who suffered serious vision complications from Saxenda eye problems.
Other Serious Side Effects Related to Saxenda Use
Saxenda’s impact extends beyond eye problems, encompassing a range of serious adverse reactions that require careful patient awareness and clinical supervision. The medication carries significant warnings that reflect the seriousness of potential systemic effects requiring thorough risk assessment.
Thyroid Tumors and Black Box Warning
The FDA mandates a black box warning for Saxenda regarding thyroid C-cell tumors, specifically medullary thyroid carcinoma (MTC). Clinical studies in rodents demonstrated dose-dependent and treatment-duration-dependent increases in thyroid C-cell tumors at clinically relevant exposures. Patients with a personal or family history of MTC or those diagnosed with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) face contraindications for Saxenda use. Healthcare providers must evaluate thyroid function and monitor for symptoms including:
- Persistent neck mass or swelling
- Difficulty swallowing (dysphagia)
- Persistent hoarseness
- Shortness of breath
Pancreatitis and Gallbladder Complications
Pancreatitis from liraglutide represents another critical concern, with acute pancreatitis observed in clinical trials and post-marketing surveillance. Patients experiencing persistent severe abdominal pain radiating to the back, with or without vomiting, require immediate medical evaluation and potential discontinuation of treatment. Gallbladder problems with Saxenda include cholelithiasis and cholecystitis, occurring at rates exceeding those in control populations. Substantial weight loss itself increases gallstone formation risk, compounding the medication’s direct effects.
Renal Impairment
Dehydration-induced kidney problems manifest through acute kidney injury and worsening chronic renal insufficiency. The mechanism involves severe gastrointestinal adverse reactions—nausea, vomiting, diarrhea—leading to volume depletion. Patients with pre-existing renal disease require enhanced monitoring of renal function parameters and hydration status throughout treatment. Clinical protocols mandate assessment of serum creatinine and estimated glomerular filtration rate at baseline and during therapy intensification periods.

Patient Guidance on Using Saxenda Safely Regarding Vision Health
Understanding Saxenda side effects on eyes requires proactive measures from patients throughout their treatment course. Establishing baseline vision assessments before initiating therapy provides essential reference points for detecting subsequent changes. Patients should document any pre-existing eye conditions, including diabetic retinopathy, glaucoma, or previous optic nerve disorders, as these factors may influence risk profiles.
Essential monitoring practices include:
- Scheduling comprehensive ophthalmological examinations every three to six months during active treatment
- Maintaining detailed logs of any visual disturbances, including date of onset, duration, and specific symptoms experienced
- Performing daily self-assessments of visual acuity, peripheral vision, and color perception
- Reporting any changes immediately to prescribing physicians rather than waiting for scheduled appointments
Patient monitoring for vision loss demands heightened awareness during the initial treatment phases when systemic adjustments to liraglutide occur. Individuals should establish clear communication protocols with their healthcare teams, designating specific providers responsible for addressing vision-related concerns. This coordination ensures rapid evaluation when symptoms emerge.
Managing side effects with healthcare providers requires transparent disclosure of all visual symptoms, regardless of perceived severity. Patients experiencing sudden vision changes, persistent eye pain, or progressive visual field deficits must seek immediate medical evaluation. Delaying assessment of these symptoms risks permanent optic nerve damage that may prove irreversible despite treatment discontinuation.
Conclusion
Saxenda Eye Problems: Are an important factor for patients and healthcare providers to consider when evaluating liraglutide therapy for weight management. The known link between Saxenda use and vision issues, especially the increased risk of NAION, requires extra caution throughout the treatment process.
Informed decision-making on weight management medication involves a thorough understanding of both the therapeutic benefits and potential negative effects. Patients prescribed Saxenda need to weigh the medication’s effectiveness in achieving weight loss goals against the documented risks to eye health and overall bodily function. This evaluation requires open communication between patients and prescribing doctors, along with a detailed review of individual risk factors and medical history.
Balancing benefits and risks of liraglutide therapy goes beyond just making initial prescription decisions. Continuous monitoring, immediate reporting of any visual disturbances, and regular eye examinations are all crucial parts of responsible medication management. Patients who experience changes in their vision should not hesitate to seek immediate medical evaluation and consider consulting with qualified legal professionals if insufficient warnings contributed to preventable harm.
The ongoing legal cases surrounding Saxenda highlight the responsibility of the pharmaceutical industry to provide proper safety information. Patients have the right to pursue legal action when manufacturers fail to adequately disclose known risks. By actively engaging with healthcare providers and being aware of their legal options, patients can empower themselves to protect both their health and their rights throughout treatment with liraglutide-based medications.