Introduction
- Dupixent Cancer Lawyer: If you suffered Dupixent and Cancer call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation. (855)-846-6529 or [email protected]. The only call you will have to make.
-
-
- Avvo Rated Top Lawyer 2024 by AVVO (2025)
- Top 25 Class action lawyerby the National Trial Lawyers Association (2023-present)
- Top 100 Civil Plaintiff Trial Lawyer by the National Trial Lawyers Association (2017-present)
- AV rating from Martindale-Hubble since 2014 (2014-present)
- 2020 Top Rated Litigator by Martindale-Hubble (2019-present)
- 2021 Top Rated Lawyer by Martindale-Hubble (2019-present)
- 2021 Elite Lawyer of the South by Martindale-Hubble (2019-present),
- Avvo Client’s Choice Award in 2021 (AVVO 2021)
- PRR AV Preeminent Rating on Lawyers.com by Martindale-Hubble® (2018-2022).
- Superb Rated Attorney, (10.0 out of 10), the Highest Rating Possible by Avvo.
- 10 rating by Justia, their highest rating for both legal abilities and ethics and professionalism.
-
- Dupixent: Is an injectable medication that was approved by the FDA in 2017. It has become a widely prescribed treatment for conditions such as atopic dermatitis (eczema), asthma, and other conditions involving type 2 inflammation. Over 800,000 patients have received this medication manufactured by Sanofi and Regeneron.
- Dupixent Cancer Claims: However, recent scientific evidence has raised significant concerns about the link between Dupixent and cancer, specifically cutaneous T-cell lymphoma (CTCL) and related malignancies. Emerging research shows that people using Dupixent are at a much higher risk of developing T-cell lymphoma. Studies suggest that the diagnosis rates of CTCL in Dupixent users are more than four times higher compared to those who do not use the medication. Despite this growing evidence, there is currently no FDA Dupixent cancer warning on the medication’s label.
- Competent Dupixent Cancer Lawyer: This article examines why retaining a qualified Dupixent Cancer Lawyer constitutes an indispensable decision for affected patients in 2025. The discussion encompasses the medical evidence linking dupilumab to CTCL, the legal framework governing Dupixent Lawsuits, eligibility criteria for participation in mass tort actions, and the critical role specialized attorneys perform in securing compensation for patients harmed by inadequately disclosed medication risks.
- Othe Pharmaceutical Medications: In addition to Dupixent, other medications like Mounjaro, Trulicity, Zepbound, and Wegovy have also been associated with severe side effects such as vision loss or blindness. For instance, hiring a Mounjaro Vision Loss Lawyer could be crucial if one faces a potential lawsuit related to Mounjaros vision loss side effects. Similarly, understanding the debilitating vision side effects of Trulicity or the worst vision side effects of Zepbound is essential for patients considering these treatments.
- Severe side Effects: Moreover, those who have suffered vision-related complications linked to Wegovy use over the manufacturer’s inadequate warnings may find it beneficial to consult a Wegovy Blindness Lawyer. Understanding these potential risks associated with various medications is vital for patient safety and informed decision-making.
Legal Issues Surrounding Dupixent Cancer Claims
- Emerging Evidence: The emerging evidence linking Dupixent to cutaneous T-cell lymphoma has led to significant legal scrutiny of Sanofi and Regeneron, the manufacturers responsible for developing and marketing dupilumab. Pharmaceutical liability claims against these companies focus on allegations that they failed to adequately disclose the potential cancer risks associated with their highly successful biologic medication.
- Failure to Warn: The failure to warn lawsuits are an important way for patients to hold manufacturers accountable who they believe put profits before patient safety.

Legal Theories Behind Dupixent Cancer Claims
The basis for legal action against Sanofi and Regeneron is built on several interconnected theories of pharmaceutical product liability. Plaintiffs and their legal representatives argue that the manufacturers:
- Knew or should have known about the increased CTCL risk through clinical trial data, post-market surveillance, and scientific literature
- Failed to conduct proper safety studies to identify and quantify the lymphoma risk before widely distributing the drug
- Left out important safety information from prescribing materials, patient medication guides, and direct-to-consumer advertising
- Continued promoting Dupixent without updating warning labels even though there was growing evidence of serious side effects
These allegations highlight the legal responsibility of drug manufacturers to remain vigilant throughout a medication’s lifecycle, from initial development to post-approval monitoring. This duty goes beyond simply reporting known risks; it also requires actively investigating potential safety signals that arise after FDA approval.
Disclosure Obligations of Pharmaceutical Companies
- Federal regulations and common law principles set clear expectations for pharmaceutical manufacturers when it comes to disclosing risks. Companies are required to provide healthcare professionals with comprehensive information about potential adverse events so that they can make informed decisions about prescribing. This obligation includes:
- Continuously evaluating safety data from various sources, including submissions to the FDA Adverse Event Reporting System
- Promptly updating prescribing information whenever new risks are identified or existing risks are better understood
- Providing adequate warnings that clearly communicate the nature, severity, and likelihood of serious adverse events
- Offering clear guidance on patient populations who may be at higher risk
- The alleged failure to include appropriate warnings about CTCL risks in Dupixent’s prescribing materials is at the heart of these legal claims. A qualified Dupixent Cancer Lawyer will investigate whether the manufacturers met these disclosure standards or if their omissions amount to actionable negligence.
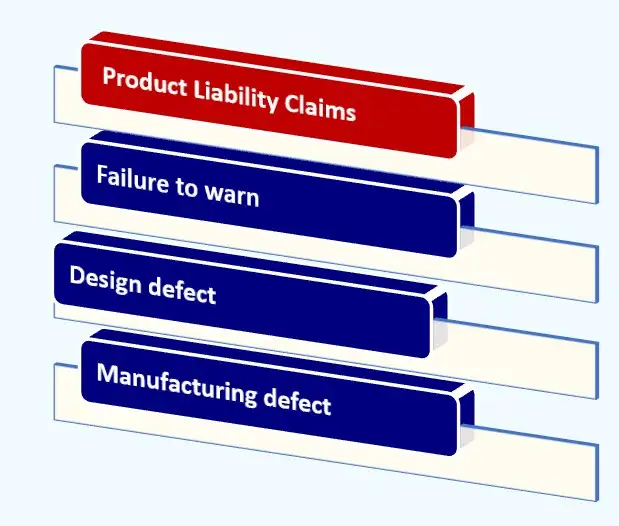
Liability Framework for Biologic Drugs
- Dupixent Cancer Lawyer: Understand that biologic medications like dupilumab pose unique legal challenges. Unlike traditional small-molecule drugs, biologics involve complex manufacturing processes and immune system mechanisms that require deep scientific knowledge for evaluation.
- Legal Framework: The legal framework for holding biologic drug manufacturers accountable includes claims related to design defects, manufacturing defects, and failure-to-warn theories. Among these, failure-to-warn arguments are proving most relevant in ongoing litigation regarding Dupixent’s potential link to cancer.
- Issues with Othe Medications: In addition to these issues, there are also concerns related to other medications such as Zepbound. Recent research published in JAMA Ophthalmology has established a concerning association between GLP-1 receptor agonists, including Zepbound, and certain eye issues. This adds another layer of complexity to the ongoing discussions about pharmaceutical liability and patient safety. For more information on this topic, you can read about the science behind the Zepbound eye issues.

Eligibility Criteria for Joining a Dupixent Cancer Lawsuit or Mass Tort
Determining whether individuals qualify for a Dupixent cancer lawsuit requires careful evaluation of specific medical and legal criteria. Pharmaceutical product liability claims demand rigorous documentation establishing the connection between drug exposure and subsequent adverse health outcomes.
Additionally, potential plaintiffs must demonstrate that they meet the eligibility criteria for joining a mass tort or class action lawsuit. These criteria typically include having a confirmed diagnosis of CTCL or another related cancer, establishing a timeline linking Dupixent use to the development of the condition, and ensuring timely filing within the statute of limitations.
Navigating this process can be challenging, but ab experienced Dupixent Cancer Lawyer can assist in gathering the necessary evidence and guiding individuals through each step.
Documented Dupixent Use and Cancer Diagnosis
Patients seeking to participate in litigation against Sanofi and Regeneron must demonstrate verifiable use of Dupixent as prescribed by a licensed healthcare provider. Medical records confirming the prescription, dosage, duration of treatment, and administration schedule constitute essential evidence. The diagnosis of cutaneous T-cell lymphoma or related malignancies must be established through pathology reports, skin biopsies, and diagnostic imaging studies performed by qualified oncologists or dermatopathologists.
Eligible for a Dupixent cancer lawsuit are individuals diagnosed with specific T-cell lymphoma subtypes, including:
- Mycosis Fungoides – the most common form of CTCL characterized by progressive skin patches and plaques
- Sézary Syndrome – an aggressive variant presenting with erythroderma and malignant cells in peripheral blood
- Other cutaneous T-cell lymphoma variants documented in medical literature as potentially associated with dupilumab exposure
Temporal Relationship and Causation Evidence
- Medical Evidence: Must establish a temporal relationship between Dupixent administration and cancer development. Treatment records demonstrating symptom onset during or following dupilumab therapy strengthen causation arguments. Laboratory reports showing abnormal lymphocyte populations, immunohistochemistry results confirming T-cell receptor gene rearrangements, and flow cytometry data identifying malignant cell markers provide scientific support for claims.
- Dupixent Cancer Lawsuit: Patients who experienced treatment failure, rapid progression of skin manifestations, or development of systemic symptoms while using Dupixent possess particularly compelling evidence for compensation in a Dupixent lawsuit.
Timeliness and Statute Limitations in Filing a Claim
The statute of limitations for pharmaceutical claims establishes strict time limits within which injured parties must initiate legal proceedings against manufacturers. Each jurisdiction has its own filing deadlines, usually ranging from one to six years from the date of injury or diagnosis. These statutory periods are non-negotiable legal requirements that, once expired, permanently extinguish the right to pursue compensation regardless of the severity of harm suffered.
Importance of Acting Quickly
To preserve legal rights under these statutory deadlines, immediate action is required upon diagnosis of cutaneous T-cell lymphoma or related conditions following Dupixent administration. The specific timeline for filing a claim varies depending on state law—some jurisdictions calculate from the date of initial injury, while others measure from the moment a reasonable person would have discovered the connection between dupilumab exposure and subsequent cancer development. This variation across state lines makes it crucial to consult with legal counsel promptly in order to determine applicable deadlines that are specific to individual circumstances.
Understanding the Discovery Rule
- The discovery rule provides limited exceptions to standard limitation periods in pharmaceutical litigation. This equitable doctrine recognizes that patients cannot reasonably be expected to file claims before becoming aware of both their injury and its causal relationship to a specific medication. Courts applying this principle may extend filing deadlines when:
-
- The connection between Dupixent use and CTCL diagnosis remained unclear due to complex medical presentations
- Manufacturers actively hid safety data regarding lymphoma risks
- Symptoms developed gradually, making it difficult to immediately recognize drug-related harm
- Confirming the diagnosis required extensive testing and consultations with specialists
- For those considering action due to adverse effects from other medications like Zepbound which has been linked to eye floaters, or individuals involved in MoonLake class action lawsuits, it’s crucial to understand these eligibility criteria thoroughly.
Role of a Dupixent Cancer Lawyer
- Skilled pharmaceutical litigation attorneys, such as those at classactionlawyertn.com, possess the technical expertise and resources necessary to navigate the complexities inherent in Dupixent Cancer Claims. These legal professionals understand the intricate relationship between drug mechanisms, adverse event reporting systems, and regulatory compliance requirements that govern pharmaceutical manufacturers.
- A Dupixent Cancer Lawyer brings specific knowledge of immunomodulatory therapies and their potential to influence lymphoproliferative disorders, enabling them to construct compelling arguments that establish causation between medication exposure and subsequent malignancy development.
Investigative Process
- Investigative Process: The investigative process undertaken by attorneys handling these cases encompasses multiple dimensions of evidence gathering and analysis. Legal teams collaborate with medical experts who specialize in dermatology, oncology, and immunology to review patient medical histories and establish temporal relationships between Dupixent administration and CTCL manifestation. This examination includes:
-
- Analysis of prescription records documenting dosage, duration, and frequency of Dupixent use
- Evaluation of pre-treatment medical assessments to establish baseline health status
- Review of pathology reports and biopsy results confirming CTCL diagnosis
- Assessment of post-diagnosis treatment protocols and patient outcomes
- Investigation of FDA adverse event databases and clinical trial data
Forensic Document Analysis
- Dupixent cancer lawyer Timothy L. Miles employs forensic document analysis to identify potential failures in manufacturer warning obligations.
- This scrutiny extends to examining internal company communications, clinical trial protocols, and regulatory submissions to determine whether Sanofi and Regeneron possessed knowledge of cancer risks prior to adequate disclosure to healthcare providers and patients.
Negotiating with Pharmaceutical Manufacturers
When it comes to negotiating with major pharmaceutical manufacturers like Sanofi (the maker of Dupixent) or Regeneron, having an attorney who understands corporate defense strategies is essential. An experienced Dupixent Cancer Lawyer knows how these companies operate and what tactics they may employ during negotiations.
In addition, it is important for attorneys practicing in pharmaceutical injury claims to be familiar with settlement valuation methodologies. This knowledge allows them to assess the worth of potential settlements based on various factors such as:
- Strength of causation evidence linking Dupixent exposure to specific subtypes of cutaneous T-cell lymphoma
- Availability of internal company documents that reveal any prior knowledge held by these manufacturers about lymphoma risks associated with their product
- Jurisdictional precedents that establish liability for pharmaceutical manufacturers
- The health status of the client involved in the case and their need for an expedited resolution

Preparing for Trial
While settlement negotiations are often preferred, there may be instances where going to trial becomes necessary. In such situations, qualified lawyers must have comprehensive strategies prepared that involve expert testimony from various professionals such as oncologists (cancer specialists), dermatopathologists (experts in skin diseases), and regulatory specialists (individuals knowledgeable about drug regulations).
The decision-making process regarding whether to pursue settlement negotiations or proceed with a trial depends on multiple factors including:
- Strength of causation evidence on the link between Dupixent and cancer
- Availability of internal company documents revealing prior knowledge held by these manufacturers about lymphoma risks
- Jurisdictional precedents concerning liability for pharmaceutical manufacturers
- The health status of the client involved in the case and their need for an expedited resolution
Providing Compassionate Guidance
Dealing with a cancer diagnosis while simultaneously pursuing legal action can be emotionally overwhelming. It is crucial for individuals facing such circumstances a to have a Dupixent Cancer Lawyer who offers compassionate guidance throughout what may be prolonged litigation processes.
Essential Documentation Requirements
- Comprehensive Medical Records: Form the foundation of any viable claim. Dermatological examination notes documenting the progression of skin manifestations—from initial patches to plaques or tumors—create a temporal narrative linking Dupixent administration to malignant transformation.
- Test Results: Laboratory results demonstrating abnormal lymphocyte populations, elevated lactate dehydrogenase levels, and flow cytometry findings identifying clonal T-cell populations provide objective markers of disease development. Treatment records showing the timing of dupilumab injections relative to symptom onset establish the critical exposure-outcome relationship necessary for causation arguments.
Pathological Evidence Standards
- Dermatopathologists reports serve as definitive proof of CTCL diagnosis. Skin biopsy immunohistochemical staining reveals the specific cellular markers characteristic of mycosis fungoides or Sézary syndrome, including aberrant T-cell phenotypes with loss of CD7 or CD26 expression.
- These pathology slides demonstrate malignant transformation at the cellular level, providing visual evidence that expert witnesses can present to juries. The microscopic documentation of atypical lymphocytes infiltrating the epidermis, particularly when showing epidermotropism patterns, becomes irrefutable evidence of lymphoma development during or following dupilumab therapy.

Compensation Available in Dupixent Cancer Lawsuits
- Patients who develop cutaneous T-cell lymphoma or related malignancies following prescribed Dupixent treatment may pursue substantial legal compensation for Dupixent side effects through pharmaceutical product liability claims.
- The compensation framework in these cases recognizes the profound medical, financial, and personal consequences of developing cancer potentially linked to dupilumab exposure.
Economic Damages
- Medical Expenses Reimbursement constitutes a primary category of recoverable damages in Dupixent cancer litigation. Claimants may seek compensation for:
-
- Diagnostic procedures including skin biopsies, pathology analysis, and imaging studies required to confirm CTCL
- Oncology consultations and specialized dermatology care for lymphoma management
- Chemotherapy, radiation therapy, phototherapy, or targeted immunotherapy treatments
- Hospitalization costs associated with severe lymphoma progression or treatment complications
- Prescription medications for cancer treatment and symptom management
- Ongoing monitoring and surveillance protocols following CTCL diagnosis
- Lost Wages and Diminished Earning Capacity represent quantifiable economic losses stemming from cancer diagnosis and treatment. Compensation types in pharmaceutical product liability cases include reimbursement for time away from employment during medical appointments, treatment sessions, and recovery periods. Claimants experiencing permanent disability or reduced work capacity due to CTCL progression may recover damages for future income loss and career advancement opportunities foreclosed by their medical condition.
Non-Economic Damages
Pain and Suffering Compensation acknowledges the substantial quality-of-life impacts experienced by patients diagnosed with cutaneous T-cell lymphoma following Dupixent use. This category encompasses:
- Physical discomfort from persistent skin lesions, erythroderma, and intense pruritus characteristic of CTCL
- Psychological distress including anxiety, depression, and emotional trauma associated with cancer diagnosis
- Disfigurement and permanent scarring resulting from lymphoma progression or treatment interventions
- Loss of enjoyment of life activities previously accessible before cancer development
- Relationship strain and social isolation stemming from visible skin manifestations
Wrongful Death Claims
- Fatal cases involving severe lymphoma progression linked to dupilumab exposure may warrant wrongful death claims filed by surviving family members. These actions seek compensation for funeral and burial expenses, loss of financial support, loss of companionship and consortium, and the decedent’s pain and suffering prior to death. Wrongful death litigation holds pharmaceutical manufacturers accountable when inadequate risk disclosure allegedly contributes to preventable fatalities among patients prescribed Dupixent for inflammatory conditions.
- In addition to these serious health risks associated with Dupixent, other medications like Mounjaro have also been reported to cause significant side effects such as blurry vision. This highlights the broader issue of adverse effects linked to certain pharmaceuticals, underscoring the importance of patient awareness and informed consent in the prescription process.
The Importance of Free Case Evaluations
Many reputable Dupixent cancer lawyers like Timothy L. Miles offer free consultations or case evaluations. Take advantage of this opportunity to discuss your situation with multiple attorneys before making a decision.
- During these evaluations, lawyers will review your medical records, treatment history, and any relevant documentation related to your cancer diagnosis. They will assess whether there are sufficient grounds for pursuing legal action against Sanofi and Regeneron.
Preserving Your Legal Rights
It’s important to remember that there are time limits (statutes of limitations) within which you must file a lawsuit if you wish to pursue legal action. By consulting with a Dupixent cancer lawyer promptly after learning about your diagnosis or potential claim, you can ensure that your legal rights are preserved.
Additionally, acting quickly can help secure evidence while it is still fresh and witnesses are available to testify if needed.
Conclusion
Finding the right Dupixent cancer lawyer requires careful consideration and research. By understanding what qualifications to look for in an attorney practicing in pharmaceutical litigation, asking relevant questions during consultations, accessing free case evaluations offered by reputable lawyers, and being mindful of statutory deadlines – you’ll increase your chances of finding effective legal representation for your case.
What are the potential benefits of a Dupixent Cancer Lawsuit?
Are There Warnings About Dupixent’s Cancer Risks?No, the FDA-approved labeling for Dupixent does not include an explicit warning about T-cell lymphoma.
How long do I have to file a Dupixent Lawsuit?
What Is Cutaneous T-cell lymphoma?
-
What it is: A type of non-Hodgkin lymphoma that starts in T-cells in the skin.
-
Common symptoms:
-
Itchy, scaly patches or plaques
-
Redness on the skin
-
Thicker, raised lesions or tumors
-
Sometimes, the skin redness can cover a large portion of the body, a condition called erythroderma
-
Progression: CTCL is often slow-growing, but some types can be aggressive and spread to other parts of the body, such as the lymph nodes, blood, or organs like the liver or lungs.
-
Diagnosis: Diagnosis can be challenging due to similar-looking symptoms and often involves a skin biopsy, physical exam, blood tests, and imaging.
-
Treatment: Treatments are chosen based on the stage of the cancer and can include creams, light therapy, radiation, or systemic treatments like chemotherapy or other medications.
How many different types of CTCL are there?
-
Mycosis Fungoide (MF) – The most common type, characterized by patches, plaques, and tumors on the skin.
-
Sézary Syndrome (SS) – A more aggressive form of MF that affects the blood as well as the skin.
-
Lymphomatoid papulosis – A benign form of CTCL that causes small, itchy bumps on the skin.
-
Granulomatous slack skin – A rare, indolent form of CTCL that causes loose, pendulous folds of skin.
-
Anaplastic large cell lymphoma – An aggressive form of CTCL that can spread to other organs.
-
Extranodal NK/T-cell lymphoma: A rare type of CTCL that affects the skin and lymph nodes.
-
Extranodal NK/T-cell lymphoma, nasal type – A rare, aggressive lymphoma that can affect the skin, among other places.
-
Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) – A rare, slow-growing type that causes nodules deep in the fatty layer of the skin, most often on the legs.
-
Primary cutaneous peripheral T-cell lymphoma (unspecified) – This is a category for rare entities that do not fit into other classifications.
Do I need to stop using Dupixent before filing?
What is the main concern linking Dupixent to cancer?
Recent studies and post-marketing reports indicate a potential association between Dupixent (dupilumab) and an increased risk of developing cutaneous T-cell lymphoma (CTCL), a rare form of non-Hodgkin lymphoma affecting the skin. This has led to a surge in Dupixent cancer lawsuits and ongoing investigations into whether pharmaceutical manufacturers provided adequate warnings about these cancer risks.
What is cutaneous T-cell lymphoma (CTCL)?
Cutaneous T-cell lymphoma is a rare type of non-Hodgkin lymphoma that primarily affects the skin. It often presents as persistent itchy rashes, red patches, plaques, or nodules. Early symptoms can mimic eczema or psoriasis, making diagnosis challenging for patients using immunomodulatory drugs like Dupixent.
How do I know if I qualify for a Dupixent cancer lawsuit?
Eligibility typically depends on:
- Documented use of Dupixent prior to a CTCL diagnosis
- Confirmation of cutaneous T-cell lymphoma via biopsy and medical records
- Evidence that risks were not adequately disclosed by drug manufacturers
- Timely filing within legal statutes of limitations
Consulting with an experienced Dupixent cancer lawyer can help determine your specific eligibility for compensation.
What compensation can be sought in a Dupixent cancer lawsuit?
Qualified plaintiffs may seek damages covering:
- Medical expenses related to CTCL treatment
- Lost wages or diminished earning capacity
- Pain, suffering, and emotional distress
- Punitive damages against manufacturers for alleged failure to warn
Are there other drugs linked to similar cancer or severe side effect lawsuits?
Yes. In addition to Dupixent lawsuits, several other medications are under scrutiny:
- Mounjaro: Linked to vision loss and macular edema claims (Mounjaro Vision Loss Lawyer)
- Trulicity: Associated with vision impairment (Trulicity Vision Loss Lawyer)
- Saxenda: Noted for serious eye problems (Saxenda Vision Loss Lawsuit)

What should I do if I notice new or worsening skin symptoms while on Dupixent?
Immediately contact your healthcare provider if you develop persistent rashes, red patches, nodules, or any skin changes that do not improve with standard eczema treatments. Early detection and prompt diagnostic procedures (such as skin biopsies) are critical for effective management.
How does the litigation process work in a Dupixent class action or individual lawsuit?
The process generally involves:
- Initial case evaluation by a qualified attorney
- Compilation of medical documentation proving Dupixent exposure and CTCL diagnosis
- Filing claims individually or joining consolidated multidistrict litigation (MDL)
- Participation in discovery, expert witness testimony, and potential trial proceedings
- Settlement negotiations or court verdicts determining compensation
Who can help me with my claim?
A specialized Dupixent Cancer Lawyer—such as Timothy L. Miles—has experience handling pharmaceutical litigation involving adverse drug reactions and immunomodulatory therapies.
Is there current scientific research supporting the link between Dupixent and CTCL?
Yes. Recent publications in dermatology and oncology journals discuss immune system modulation through IL-4/IL-13 inhibition by dupilumab as a possible mechanism contributing to lymphoproliferative disorders like CTCL. Ongoing surveillance data continues to inform both medical practice and legal proceedings.
What are some keywords related to this topic for further research?
Some important keywords include:
- Dupixent cancer lawsuit
- Cutaneous T-cell lymphoma claims
- Non-Hodgkin lymphoma drug risks
- Dupilumab class action lawsuit
- Pharmaceutical product liability litigation
- Biologic medication adverse effects
- Drug manufacturer failure-to-warn cases
- Immunomodulator cancer risk attorney
How can current users protect themselves from potential cancer risks associated with Dupixent?
Patients should:
- Engage in regular full-body self-examinations for unusual skin changes.
- Schedule routine dermatology check-ups.
- Maintain open communication with healthcare providers regarding any new symptoms.
- Stay updated on new research findings and FDA safety alerts regarding dupilumab.
- Document all medication use and side effects for reference in potential legal actions.


