Introduction to the Dupixent Cancer Lawsuit
- Dupixent Cancer Lawsuit: Recent scientific investigations have raised concerns about a potential link between Dupixent and cancer development, particularly cutaneous T-cell lymphoma (CTCL). Multiple peer-reviewed studies published between 2024 and 2025 have documented cases suggesting an increased cancer risk among certain patient populations receiving dupilumab treatment. These findings have led to regulatory scrutiny and legal action against the drug’s manufacturers, Sanofi and Regeneron.
- Dupixent (dupilumab): Is a significant advancement in biologic therapy, approved for treating multiple inflammatory conditions including atopic dermatitis, eczema, asthma, chronic rhinosinusitis with nasal polyps, and eosinophilic esophagitis. The medication works by targeting specific proteins in the immune system that contribute to chronic inflammation, providing relief for millions of patients worldwide who have struggled with these debilitating conditions.
- Dupixent And Cancer Risks: Understanding the relationship between Dupixent and cancer risks requires careful examination of clinical evidence, regulatory warnings, and ongoing investigations. Patients currently receiving treatment, healthcare professionals prescribing the medication, and individuals considering Dupixent therapy must possess comprehensive knowledge of these emerging safety concerns.
- 30 Frequently Asked Questions: It’s crucial for current and former users of Dupixent to understand the implications of its side effects related to cancer development. This comprehensive guide aims to answer 30 commonly asked questions about the Dupixent cancer lawsuit. It covers important topics such as medical evidence, legal processes, compensation options, and the latest updates on ongoing lawsuits. By providing this information, we hope to empower patients in making informed decisions about their rights and healthcare choices.
A COMPLETE TIMELINE OF SIGNIFICANT EVENTS
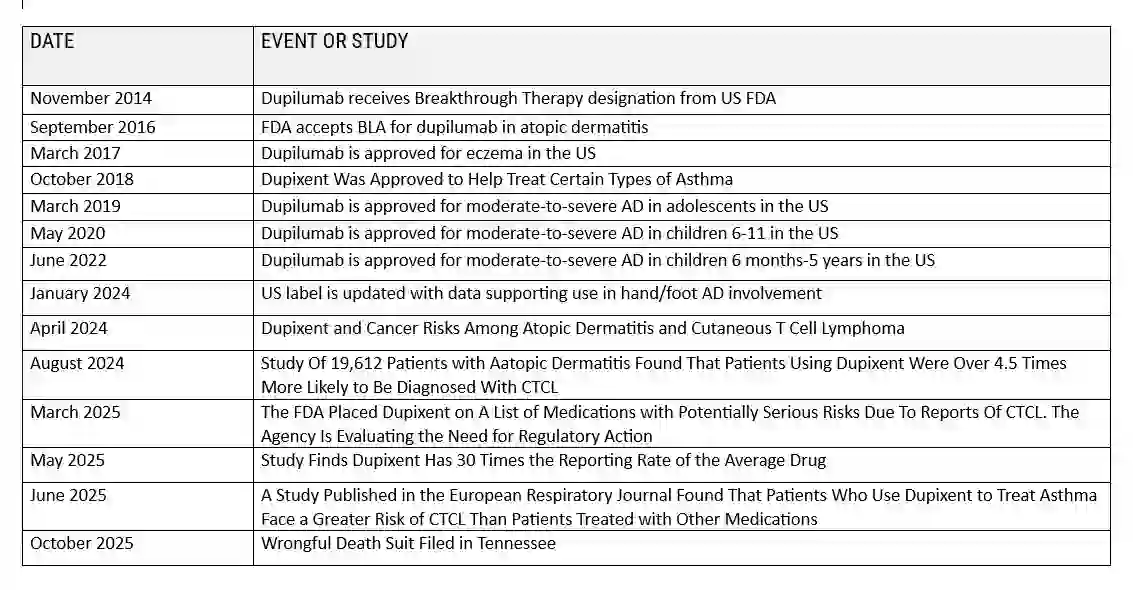
1. What Is Cutaneous T-Cell Lymphoma (CTCL)
Cutaneous T-cell lymphoma is a type of non-Hodgkin lymphoma where cancerous T-lymphocytes primarily affect the skin. This uncommon cancer occurs when T-cells, which are white blood cells that play a role in the immune system, become cancerous and gather in the skin. There are several subtypes of this disease, each with different levels of severity and clinical presentations.
Primary Types of CTCL
The two main types of cutaneous T-cell lymphoma are:
- Mycosis Fungoides: This is the most common subtype, making up about 50-60% of all CTCL cases. It usually progresses slowly through different stages:
- Patch stage: Flat, scaly patches that look like eczema or psoriasis appear on the skin.
- Plaque stage: Raised, thickened lesions with more defined edges develop.
- Tumor stage: Nodules or tumors form, indicating advanced disease progression.
- Sézary syndrome: This is a more aggressive form of CTCL that affects about 3-5% of patients. It is characterized by widespread redness (erythroderma) covering large areas of skin, intense itching (pruritus), and the presence of malignant T-cells in the bloodstream. Sézary syndrome tends to progress more quickly than Mycosis Fungoides.
Clinical Manifestations and Disease Progression
The symptoms of cutaneous T-cell lymphoma go beyond just skin issues. Patients often experience:
- Persistent skin lesions that do not respond to standard dermatological treatments.
- Severe itching that affects their quality of life and sleep.
- Skin discoloration ranging from pink to deep red or brown patches.
- Enlarged lymph nodes (lympadenopathy) in advanced stages.
- Hair loss (alopecia) in affected areas of the scalp.
- Thickened, cracked skin resembling chronic dermatitis.
In advanced cases, CTCL may spread beyond the skin to involve lymph nodes, blood, and internal organs such as the liver, spleen, and bone marrow. The rate at which the disease progresses varies widely among patients. Some may have a slow-moving disease for decades while others experience rapid advancement requiring aggressive treatment.
2. What Is the Link Between Dupixent and Cancer Risks
Scientific investigations have revealed a statistically significant association between Dupixent use and elevated CTCL risk with Dupixent. Research published in peer-reviewed medical journals demonstrates that patients treated with dupilumab exhibited a substantially higher incidence of cutaneous T-cell lymphoma compared to control populations. The data indicates a risk elevation ranging from 2.5 to 4.59 times greater than baseline rates observed in patients not receiving the medication.
The most compelling evidence emerged from post-marketing surveillance studies and clinical trial analyses conducted after Dupixent received FDA approval in 2017. These investigations tracked thousands of patients across multiple therapeutic indications, including atopic dermatitis, asthma, and chronic rhinosinusitis with nasal polyposis. The link between Dupixent and cancer became apparent when researchers identified clusters of CTCL diagnoses among patients with no prior history of lymphoproliferative disorders.
Key Research Findings:
- Patients with moderate-to-severe atopic dermatitis treated with dupilumab demonstrated a 4.59-fold increased risk of developing CTCL
- The median time from treatment initiation to CTCL diagnosis ranged from several months to multiple years
- Risk appeared dose-dependent and duration-dependent in certain patient populations
- Younger patients without traditional risk factors for lymphoma developed aggressive forms of CTCL
The mechanism underlying Dupixent and cancer risks remains under active investigation. Dupilumab functions as an interleukin-4 receptor alpha antagonist, blocking signaling pathways involved in type 2 inflammatory responses. Researchers hypothesize that sustained immunomodulation may create conditions favorable for malignant T-cell proliferation in susceptible individuals. Alternative theories suggest the medication may unmask pre-existing subclinical lymphoproliferative conditions or directly influence T-cell differentiation pathways.
These findings prompted regulatory agencies to scrutinize safety data and manufacturers to update prescribing information, though questions persist regarding the adequacy and timing of risk communication to healthcare providers and patients.
3. Who is Being Sued?
Both Sanofi and Regeneron Pharmaceuticals are named as defendants in the Dupixent Cancer Lawsuit because they both played a role in developing and selling the drug. Sanofi has the main marketing rights in the United States, while Regeneron is heavily involved in the scientific development and ongoing research of the drug. The lawsuits claim that both companies knew about potential cancer risks based on clinical trial data and post-market surveillance reports but failed to properly inform doctors and patients about these risks.
4. What Are the Main Claims Against the Manufacturers?
The main claims in these lawsuits focus on the manufacturers’ alleged failure to warn about cancer risks associated with Dupixent use. Specifically, plaintiffs argue that:
- Sanofi and Regeneron had clinical data showing increased cancer risks but did not provide adequate warnings on product labeling
- The companies did not conduct enough post-market studies to identify and measure cancer risks in real-world patient populations
- Healthcare providers did not receive enough information to make informed prescribing decisions and properly counsel patients about potential cancer risks
- Product marketing materials highlighted benefits while downplaying or ignoring serious side effects, including cancer development
The plaintiffs argue that if they had received complete and accurate information about the risk of developing cutaneous T-cell lymphoma and other Dupixent side effects, they would have chosen not to use Dupixent or sought alternative treatment options. The lawsuits aim to prove that the manufacturers’ failure to disclose this information constitutes negligence, breach of warranty, and violations of consumer protection laws.
5. Who Can Qualify for a Dupixent Cancer Lawsuit?
Individuals seeking to qualify for a Dupixent cancer lawsuit must meet specific eligibility criteria established through legal precedent and medical documentation requirements. The primary qualification centers on demonstrating a direct connection between Dupixent usage and subsequent cancer diagnosis.
Primary Eligibility Requirements
To be eligible for a Dupixent lawsuit, claimants must satisfy the following fundamental criteria:
- Confirmed CTCL Diagnosis: Medical records must document a diagnosis of Cutaneous T-Cell Lymphoma, including Mycosis Fungoides or Sézary syndrome, following Dupixent treatment
- Documented Dupixent Use: Prescription records and pharmacy documentation establishing regular use of dupilumab for an FDA-approved condition
- Temporal Relationship: Evidence demonstrating the cancer diagnosis occurred during or after Dupixent treatment, establishing a plausible timeline
- Medical Documentation: Comprehensive medical records including biopsy results, pathology reports, and oncology consultations confirming CTCL diagnosis

Additional Qualifying Factors
Beyond the primary requirements, several supplementary factors strengthen eligibility:
- Duration of Dupixent treatment prior to cancer diagnosis
- Absence of pre-existing CTCL or related conditions before initiating Dupixent therapy
- Documentation of adverse events reported to healthcare providers during treatment
- Evidence of inadequate warnings or insufficient informed consent regarding cancer risks
Exclusionary Circumstances
Certain situations may disqualify potential claimants from pursuing legal action:
- Pre-existing CTCL diagnosis before commencing Dupixent treatment
- Insufficient medical documentation linking cancer development to medication use
- Expiration of applicable statutes of limitations in the claimant’s jurisdiction
- Previous settlement or legal resolution related to Dupixent-associated injuries
Individuals who qualify for a Dupixent cancer lawsuit should gather comprehensive medical records, prescription histories, and documentation of all treatment-related expenses. The strength of a potential claim depends substantially on the quality and completeness of medical evidence establishing the connection between Dupixent exposure and subsequent CTCL development.
6. What Is the Compensation in a Dupixent Cancer Lawsuit?
Individuals who develop cutaneous T-cell lymphoma following Dupixent treatment may pursue compensation in a Dupixent cancer lawsuit to address the substantial financial and personal burdens resulting from their diagnosis. The legal framework governing pharmaceutical litigation recognizes multiple categories of recoverable damages designed to restore plaintiffs to their pre-injury status to the extent monetary compensation permits.
Economic Damages
Economic Damages constitute the quantifiable financial losses directly attributable to the cancer diagnosis. These damages for medical expenses encompass:
- Diagnostic testing and imaging procedures required to confirm CTCL
- Oncology consultations and specialist evaluations
- Chemotherapy, radiation therapy, or targeted biological treatments
- Hospitalizations and emergency medical interventions
- Prescription medications and supportive care therapies
- Ongoing monitoring and surveillance for disease progression
- Future medical care necessitated by the condition
Lost income represents another critical component of economic damages. Patients undergoing cancer treatment frequently experience work interruptions ranging from temporary absences to permanent disability. Compensation addresses:
- Wages lost during treatment periods and recovery
- Diminished earning capacity resulting from long-term health complications
- Loss of employment benefits including health insurance and retirement contributions
- Reduced career advancement opportunities due to medical limitations
Non-Economic Damages
Non-Economic Damages address intangible losses that profoundly impact quality of life. These include physical pain and suffering endured throughout diagnosis and treatment, emotional distress stemming from a cancer diagnosis, disfigurement or scarring associated with CTCL manifestations, and loss of enjoyment of life activities previously accessible to the plaintiff.
Punitive Damages
Certain jurisdictions permit punitive damages when manufacturers demonstrate reckless disregard for patient safety. These damages serve to punish egregious corporate conduct and deter similar behavior across the pharmaceutical industry. The availability and calculation of punitive damages vary significantly based on state-specific legal standards and the particular facts surrounding each case.
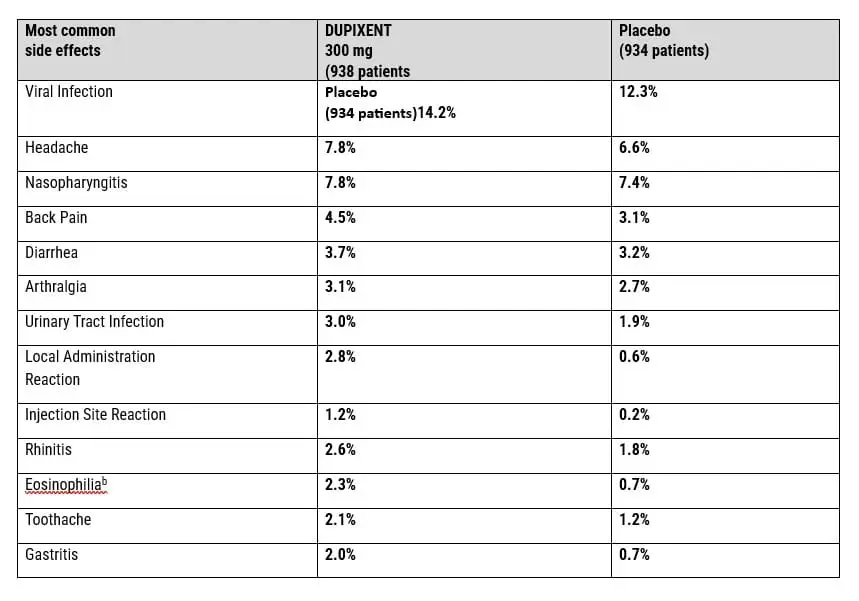
SAFETY AND EFFICACY DATA OF DUPIXENT SIDE EFFECTS FROM CLINICAL TRIALS
7. How to Find the Right Lawyer for Your Dupixent Cancer Lawsuit?
Choosing the right Dupixent Cancer Lawyer is crucial for anyone pursuing a Dupixent Cancer Lawsuit. These cases can be complicated, so it’s important to have an attorney who knows about mass tort cases, regulations, and medical causation principles. Lawyers with a lot of experience in pharmaceutical litigation understand how drug manufacturers, regulatory agencies, and patient safety obligations all work together.
Timothy L. Miles is the type of lawyer you need to navigate a Dupixent Cancer Lawsuit. Attorneys who practice in pharmaceutical mass torts have specific skills that are important for building a strong case:
- Regulatory expertise: Knowledge of the FDA approval process and requirements for monitoring a drug after it has been approved
- Scientific literacy: Ability to understand clinical trial data and studies that connect Dupixent to CTCL
- Resource networks: Connections with medical experts, toxicologists, and oncologists who can support your claims about causation
- Litigation infrastructure: Capability to handle complex discovery processes and work with multiple plaintiffs
8. Why You Should Consider Hiring an Experienced Pharmaceutical Litigation Attorney for your Dupixent Cancer Lawsuit?
Experienced pharmaceutical litigation attorneys typically offer contingency fee arrangements, which means you don’t have to pay them upfront. This fee structure aligns the attorney’s compensation with the outcome of your case, ensuring that they are fully committed to representing you throughout the litigation process.
When hiring an attorney, it’s important to discuss their communication style and how often you can expect updates on your case. Make sure they are transparent about potential timelines, settlement negotiations, and trial preparation strategies. At the Law Offices of Timothy L. Miles we take all cases on a contingency basis including a Dupixent Cancer Lawsuit.
9. What Is the Role of Qualified Counsel in Pharmaceutical Mass Torts?
The attorney-client relationship in pharmaceutical mass torts goes beyond just legal representation. Qualified counsel should also act as an advocate who understands the medical, emotional, and financial challenges caused by CTCL diagnoses.
Legal teams with experience in pharmaceutical mass torts often have established relationships with medical professionals who can provide documentation on the progression of cutaneous T-cell lymphoma and its link to Dupixent exposure. This can be valuable evidence in supporting your Dupixent Cancer Lawsuit.
10. What is Dupixent (dupilumab) and how does it work in the body?
Dupixent functions as a monoclonal antibody that inhibits interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling pathways. These cytokines play critical roles in inflammatory responses associated with atopic dermatitis, asthma, and chronic rhinosinusitis with nasal polyposis. By blocking these pathways, dupilumab reduces inflammation and alleviates symptoms in patients with moderate-to-severe presentations of these conditions.
11. What types of cancer have been associated with Dupixent use?
Research has identified a statistically significant association between dupilumab treatment and Cutaneous T-Cell Lymphoma (CTCL), specifically Mycosis Fungoides and Sézary syndrome. These malignancies originate from mature T-lymphocytes that infiltrate the skin, potentially progressing to affect lymph nodes, blood, and internal organs. The medical science FAQs about dupilumab consistently highlight CTCL as the primary cancer concern in ongoing litigation.
13. How much does Dupixent increase the risk of developing CTCL?
Clinical studies and post-marketing surveillance data indicate that Dupixent users face an elevated risk of CTCL ranging from 2.5 to 4.59 times higher than the general population. The precise risk magnitude varies based on treatment duration, patient demographics, and underlying health conditions that may predispose individuals to lymphoproliferative disorders.
14. Is there a Dupixent class action lawsuit?
No Dupixent class action lawsuit currently exists for Dupixent-related cancer claims. The litigation proceeds through mass tort litigation, which differs fundamentally from class action proceedings. Mass tort cases allow each plaintiff to maintain an individual lawsuit while benefiting from coordinated discovery and shared legal resources. This structure proves advantageous for pharmaceutical injury claims where individual damages, medical histories, and exposure patterns vary significantly among affected patients.
15.What distinguishes individual lawsuits from class action lawsuits in Dupixent cases?
Individual lawsuits permit plaintiffs to present unique medical evidence, specific treatment timelines, and personalized damage calculations. Each case receives independent evaluation based on:
- Individual medical records and diagnosis dates
- Specific duration and dosage of Dupixent exposure
- Unique economic losses and medical expenses
- Personal pain and suffering experiences
- Distinct causation evidence linking dupilumab to CTCL development
Class actions require common questions of law and fact applicable to all members, which pharmaceutical injury cases rarely satisfy due to varying individual circumstances.
16. How does mass tort litigation work for a Dupixent cancer lawsuit?
Mass tort litigation consolidates similar cases for pretrial proceedings while preserving individual claim characteristics. Courts may establish multidistrict litigation (MDL) to streamline discovery, expert witness testimony, and motion practice. This coordination reduces redundant legal work and litigation costs while maintaining each plaintiff’s right to individual compensation based on specific damages.
17.What is the statute of limitations for filing a Dupixent cancer lawsuit?
Statutes of limitations vary by jurisdiction, typically ranging from one to six years from the date of diagnosis or discovery of harm. The discovery rule applies in most states, meaning the limitation period begins when the plaintiff knew or reasonably should have known about the connection between Dupixent use and cancer development. Consultation with qualified Dupixent Cancer Lawyer ensures compliance with applicable deadlines specific to each jurisdiction.

18. What evidence is required to file a Dupixent cancer lawsuit?
Plaintiffs must establish several evidentiary elements in a Dupixent Lawsuit:
- Medical documentation confirming CTCL diagnosis through biopsy and pathology reports
- Prescription records demonstrating Dupixent use prior to cancer diagnosis
- Treatment history showing temporal relationship between dupilumab exposure and CTCL development
- Medical causation opinion from qualified experts linking Dupixent and cancer development
- Damage documentation including medical bills, employment records, and impact statements
19. Who can be named as defendants in a Dupixent cancer lawsuit?
Defendants typically include:
- Sanofi S.A. and its subsidiaries as co-developer and manufacturer
- Regeneron Pharmaceuticals, Inc. as co-developer and manufacturer
- Potentially prescribing physicians if medical malpractice contributed to harm
- Healthcare facilities where treatment occurred if institutional negligence existed
Most cases focus on manufacturer liability based on failure to warn, design defect, or inadequate testing claims.
20. What legal theories support a Dupixent cancer lawsuit?
Plaintiffs pursue multiple legal theories:
Failure to Warn: Manufacturers possessed knowledge of cancer risks but failed to provide adequate warnings to healthcare providers and patients.
Design Defect: The drug’s formulation presents unreasonable dangers that outweigh therapeutic benefits for certain patient populations.
Negligence: Defendants breached duties of care in testing, manufacturing, marketing, or post-market surveillance of dupilumab.
21. How Much Does if Cost to Hire a Dupixent Cancer Lawyer?
- No Fee: It does not cost anything to hire a lawyer if you are eligible for a Dupixent Cancer Lawsuit. We take all cases on a contingency basis which means we do not get paid unless we win or settle your case.
- Talk with a Lawyer Free of Charge: A lawyer can explain the process of a Dupixent Cancer Lawsuit and answer any questions you may have free of charge. The call is free and so is the fee unless we will or settle your case, so call Dupixent Cancer Lawsuit Timothy L. Miles today to see if you may be entitled to significant compensation.
22. If I Was Diagnosed with Dupixent and Cancer, How Do I Get Started?
- Get a Free Case Evaluation: If you or a loved on developed Dupixent and Cancer after taking Dupixent, the most important thing to do now is get a free case evaluation from a Dupixent Cancer Lawyer to see if you qualify for a Dupixent Cancer Lawsuit. These are not typical cases any lawyer can litigate. Contact Dupixent Cancer Lawyer Timothy L. Miles who can tell you if you are eligible for a Dupixent Cancer Lawsuit and can explain more about what will happen next. (855) 846–6529 or [email protected].
- Medical Records Review: The earlier your records are reviewed, the stronger the foundation becomes, especially in cases where the biopsy history is limited or where the initial diagnosis was “eczema” that simply never responded the way it should have. A lawyer experienced in drug litigation can help secure the complete medical file, obtain an independent pathology review if necessary, and establish a clear timeline that courts and juries can follow.
- Time Is of the Essence: If you have a valid case, then it is vital to preserve your rights under the statute of limitations in your state. These deadlines are different in different states, but it normally starts running from the time of diagnosis or from the point when a reasonable person should have realized the connection between the diagnosis and the drug. Time is of the essence so give Dupixent Cancer Lawyer Timothy L. Miles a call today.
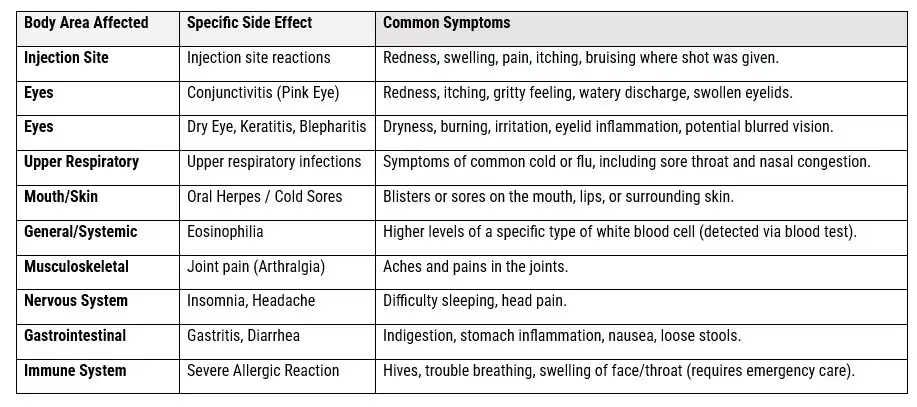
23. What Are the Common Dupixent Side Effects?
THE DUPIXENT COMMON SIDE EFFECTS PROFILE
24. What Is the Dupixent Lawsuit Status?
25. Is there Research that Supports the Link Between Dupixent and Cancer?
28. Are There Warnings About Dupixent’s Cancer Risks?
29. Do I need to stop using Dupixent before filing a Dupixent Lawsuit?
30. What Are the Key Warning Signs for Dupixent and Cancer?
Conclusion
The Dupixent Cancer Lawsuit is an important case in pharmaceutical legal battles. It emphasizes the need for honest communication about risks in medication safety. Patients who developed Dupixent and Cancer are facing significant medical, financial, and emotional challenges that require legal investigation.
Understanding the link between dupilumab treatment and increased CTCL risk helps affected individuals make informed choices about their legal options. The claims against Sanofi and Regeneron Pharmaceuticals focus on their failure to properly inform patients and healthcare providers about these serious cancer risks, which could jeopardize patient safety and informed consent.
For individuals diagnosed with CTCL after using Dupixent, taking decisive action becomes essential:
- Document medical records establishing treatment history and diagnosis
- Preserve evidence of Dupixent prescriptions and usage patterns
- Consult qualified healthcare professionals regarding ongoing medical management
- Seek expert legal representation pharmaceutical litigation to evaluate claim viability
The next steps of filing a Dubixent cancer lawsuit require careful attention to statutory deadlines and procedural requirements. Experienced attorneys in pharmaceutical mass torts possess the resources, knowledge, and litigation capabilities necessary to navigate complex medical evidence and corporate defense strategies.
Affected patients deserve accountability, appropriate compensation, and recognition of the harm suffered. Legal consultation provides clarity regarding eligibility, potential recovery, and the pathway toward justice in these challenging circumstances.

