Introduction to the Dupixent Cancer Lawsuit
The Dupixent Cancer Lawsuit continues to build momentum as cases continue to be filed. Dupixent (dupilumab) is a prescription biologic injection used to treat inflammatory diseases, including moderate to severe atopic dermatitis (eczema), asthma, chronic rhinosinusitis with nasal polyps, and eosinophilic esophagitis. For many patients, it has been a meaningful therapy that reduces flares and improves quality of life.
However, a growing body of adverse event reports, case literature, and observational research has raised specific and serious concerns regarding the safety of Dupixent. There is an ongoing debate about whether Dupixent use may be associated with an increased risk of certain lymphomas, particularly cutaneous T-cell lymphoma (CTCL), a rare form of non-Hodgkin lymphoma that can initially resemble eczema or dermatitis. This concern has led to several lawsuits against the manufacturers of Dupixent, alleging that they did not provide adequate warnings about these potential risks.
This 2026 update explains what is currently known about the situation, what remains uncertain, and what patients should consider from both a medical and legal perspective. It’s crucial to note that this article is informational only and does not constitute medical or legal advice. Patients are advised not to stop or change a prescribed medication without consulting their treating clinician.
Dupixent is a monoclonal antibody that targets interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling by inhibiting the IL-4 receptor alpha subunit. In clinical practice, it is commonly prescribed for:
- Atopic dermatitis (eczema)
- Asthma (including eosinophilic phenotypes)
- Chronic rhinosinusitis with nasal polyps
- Eosinophilic esophagitis
The current lawsuit and safety discussions do not argue that Dupixent is ineffective for inflammatory disease. The central question is narrower and more consequential: whether, for a subset of patients, Dupixent may be linked to T-cell lymphomas, especially CTCL, and whether manufacturers provided adequate warnings or risk communication as evidence accumulated. For more detailed information on the ongoing lawsuits regarding Dupixent’s safety concerns, you can follow these links for updates on the legal proceedings related to Dupixent’s side effects or visit this page for further insights into the safety debate.
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected].standing Dupixent: Its Role in the Safety Debate
The Core Allegation in the Dupixent Cancer Lawsuit: Dupixent and Cutaneous T-Cell Lymphoma (CTCL)
Cutaneous T-cell lymphoma (CTCL) is a cancer of T-lymphocytes that primarily involves the skin. Early CTCL can mimic common inflammatory skin disease, which can lead to delayed diagnosis.
Reported CTCL-related concerns include:
- An observed pattern of CTCL diagnoses in some Dupixent users
- Claims that Dupixent may accelerate the clinical course or unmask CTCL that was previously misdiagnosed as eczema
- Claims that the drug label lacks a cancer-specific warning sufficient to alert clinicians and patients to this risk signal
Some reports and analyses referenced in public discussion describe substantially increased relative risk estimates for CTCL in certain patient cohorts treated with dupilumab. These figures are not, by themselves, definitive proof of causation, but they are central to the ongoing safety and litigation narrative.
Key Terms Patients Should Understand: Signal vs Association vs Causality
Because cancer-risk reporting often gets distorted online, it helps to separate three concepts that regulators, clinicians, and courts treat differently:
- Safety signal: A pattern of reports that warrants further evaluation. A signal is not proof.
- Association: A statistical relationship observed in data. Association does not necessarily mean the drug caused the outcome.
- Causality: A conclusion that the drug likely caused the outcome, typically requiring stronger evidence, consistency across studies, plausible mechanisms, and exclusion of alternative explanations.
As of 2026, public reporting commonly describes an ongoing evaluation and an emerging safety signal related to CTCL and other T-cell lymphomas. However, a definitive causal finding is not universally established in publicly available sources. Litigation often proceeds in this evidentiary environment, especially when plaintiffs allege that warnings were inadequate given the evolving signal.
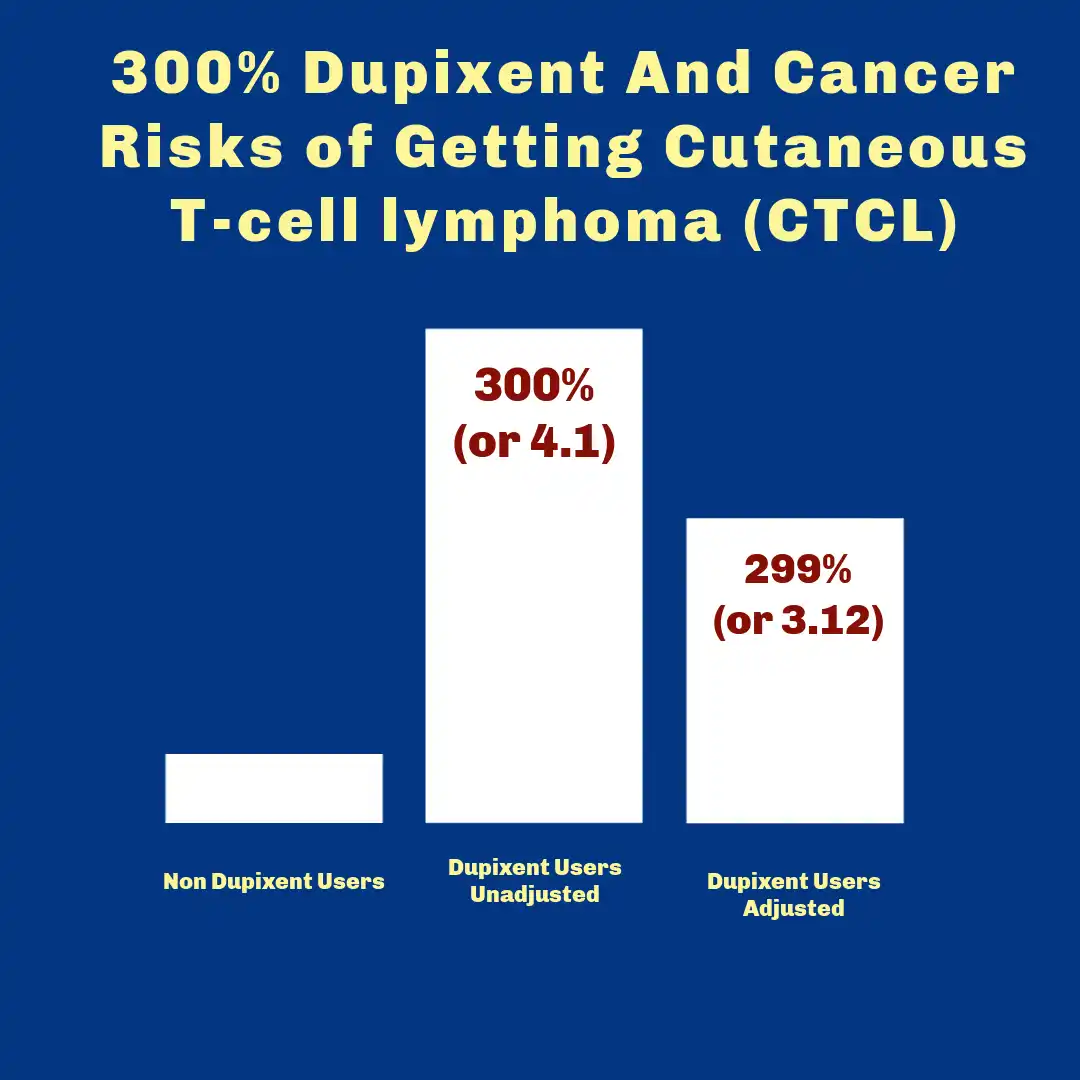
What the Research and Reporting Trend Has Suggested on Dupixent and Cancer
Several categories of evidence have been cited in connection with Dupixent and lymphoma risk. Each has strengths and limitations.
1) Observational findings in atopic dermatitis cohorts
Some published analyses have suggested that dupilumab-treated atopic dermatitis patients may show a higher observed rate of CTCL diagnosis than comparator groups, with relative risk metrics reported in the multiple-fold range in certain summaries.
How to interpret this carefully:
- Atopic dermatitis itself can be a diagnostic “mask” for early CTCL.
- Patients placed on biologics may undergo more dermatology follow-up, biopsy, and escalation of care, increasing detection.
- Confounding by severity is possible because patients with more severe or atypical disease are more likely to be prescribed advanced therapies.
Even with these caveats, elevated relative risks in repeated settings can intensify scrutiny, particularly when paired with real-world adverse event reporting.
2) Adverse event reports submitted to the FDA (FAERS)
Public discussion has referenced nearly 300 reports of T-cell lymphoma among Dupixent users within the U.S. Food and Drug Administraqtion’s (FDA) Adverse Event Reporting System (FAERS).
What FAERS can and cannot prove:
- FAERS is designed to detect potential signals, not to establish incidence or causation.
- Reports may be incomplete, duplicated, or missing diagnostic confirmation.
- FAERS lacks a true denominator; it cannot directly answer “how often” something occurs in all users.
Even with limitations, clusters of rare events can matter. In litigation, these reports are often used to argue that manufacturers and regulators had enough notice to evaluate and potentially strengthen warnings sooner.
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected].standing Dupixent: Its Role in the Safety Debate
3) Reports of multiple T-cell lymphoma subtypes
In addition to CTCL, reports have referenced other T-cell and NK-cell lymphomas in some dupilumab-treated populations, including patients treated for asthma.
This broadens the discussion beyond a single diagnosis and may influence how risk is framed in expert testimony, pharmacovigilance analysis, and labeling arguments.
Why CTCL Is So Often Missed: The Eczema Overlap Problem
A central clinical issue is that early CTCL can resemble chronic dermatitis. Patients may cycle through topical steroids, systemic immunosuppressants, phototherapy, and biologics before someone considers lymphoma and orders a biopsy.
CTCL symptoms that can resemble eczema
- Persistent itching (pruritus)
- Red, scaly, thickened, peeling, or discolored skin
- Patches or plaques that do not behave like typical eczema
Symptoms that may suggest CTCL progression
- Raised bumps, nodules, sores, or ulcers
- Tumor-like lesions on the skin
- Hair loss in affected areas
- Enlarged lymph nodes
- Systemic symptoms such as fatigue or unexplained weight loss (in more advanced contexts)
From a patient-safety perspective, the concern is not only whether Dupixent increases risk, but also whether its use in atypical “eczema” cases may contribute to diagnostic delay if the underlying condition is actually CTCL.
Warning Signs of Dupixent and Cancer That Merit a Re-Evaluation
Patients and clinicians often focus on efficacy. In this specific controversy, a different set of “red flags” matters. Reports have highlighted warning signs that, in context, may justify a biopsy or specialist reassessment.
Commonly Cited Red Flags Include:
- Failure to respond to Dupixent in a manner consistent with typical eczema improvement
- Progression from patches to plaques or tumors
- Sudden acceleration of skin symptoms after starting therapy
- Development of systemic symptoms (for example, fevers, night sweats, unexplained weight loss)
- New or worsening lymph node enlargement
- A rash that becomes more asymmetric, fixed, or treatment-resistant
These signs do not prove CTCL, but they support a prudent, proactive approach: re-check the diagnosis rather than reflexively escalating anti-inflammatory therapy.
Dupixent Side Effects: What the Label Commonly Emphasizes
Dupixent’s safety profile includes side effects that have been documented in clinical development and post-marketing experience. Public summaries often reference trials including 1,874 adult patients across two clinical trials, with common adverse effects occurring in at least 2% of patients and more frequently than placebo.
Commonly discussed side effects include:
- Injection site reactions
- Eye-related adverse events (for example, conjunctivitis and other ocular inflammation)
- Joint aches and pain (arthralgia) in some patients
Serious side effects may include:
- Allergic reactions
- Significant eye problems requiring ophthalmologic care
- Other adverse events that warrant urgent clinical evaluation
The core legal criticism raised in Dupixent cancer lawsuits is not that common side effects were hidden, but that the label allegedly lacked adequate cancer-specific warning language related to CTCL or T-cell lymphomas as a potential risk.
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected].standing Dupixent: Its Role in the Safety Debate
The Current Legal Landscape (2026): Lawsuits, but No Formal MDL Yet
As of this 2026 update, public reporting has described Dupixent cancer litigation as emerging and expanding, with expectations that more cases could lead to coordinated proceedings.
What exists now
- Individual product liability cases alleging inadequate warnings and harm related to CTCL and other lymphomas
- Reports of the first wrongful death lawsuit filed in Tennessee alleging fatal T-cell lymphoma linked to Dupixent
What does not appear to be established yet (in the common public summaries)
- A formal nationwide multidistrict litigation (MDL) or certified class action for Dupixent cancer claims
Why consolidation matters
When multiple similar product liability cases accumulate, courts sometimes centralize pretrial proceedings in an MDL to:
- Standardize discovery
- Coordinate expert evidence
- Reduce inconsistent rulings
- Encourage global settlement discussions if liability risk becomes clearer
If filings continue, consolidation remains a realistic possibility, but it is not a certainty and depends on case volume, factual overlap, and judicial decisions.
What Plaintiffs Typically Allege in a Dupixent Cancer Lawsuit
While each complaint differs, reported allegations often follow a consistent framework seen in pharmaceutical product liability litigation:
- Failure to warn: The manufacturers allegedly did not provide adequate warning regarding CTCL or lymphoma risk.
- Inadequate risk communication: Plaintiffs may argue prescribers were not sufficiently informed through labeling, safety communications, or post-marketing actions.
- Delayed diagnosis and worsened disease: Because CTCL can mimic eczema, plaintiffs may allege that Dupixent use contributed to a delay in diagnosing cancer, allowing progression.
- Deficient post-marketing surveillance response: Plaintiffs may argue that accumulating reports should have prompted faster or stronger warnings and guidance.
The defense in these cases commonly focuses on causation challenges, confounding factors, alternative explanations, and the argument that the benefit-risk profile remains favorable for indicated populations.
For specific instances of these allegations leading to lawsuits, you can refer to detailed accounts like this wrongful death lawsuit or this product liability case.
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected].standing Dupixent: Its Role in the Safety Debate
Eligibility: Who May Qualify for a Dupixent Cancer Lawsuit
Eligibility is fact-specific and depends on medical records, diagnosis, and timing. Public-facing criteria commonly referenced include the following:
You may qualify for a Dupixent Cancer Lawsuit if you:
- Were prescribed or used Dupixent (dupilumab), and
- Were later diagnosed with a T-cell lymphoma, including cutaneous T-cell lymphoma (CTCL), and
- Experienced serious harm, complications, or substantial medical intervention, and
- Are within your state’s statute of limitations
Because time limits vary, many attorneys evaluate the filing deadline from:
- The date of diagnosis, or
- The date you reasonably discovered a possible connection between Dupixent and the diagnosis (depending on state law)
A timely review of your medical record is often decisive in determining viability. For more detailed information about the eligibility criteria for a Dupixent cancer lawsuit, you can refer to this resource.
Compensation: What Damages May Be Sought
In product liability cancer cases, successful plaintiffs may seek compensatory damages, and in certain circumstances punitive damages.
Compensatory damages may include:
- Past and future medical expenses (treatment, hospitalizations, oncology care)
- Lost wages and loss of earning capacity
- Pain and suffering
- Out-of-pocket costs (travel, caregiving, supportive therapies)
Punitive damages (in some cases)
Punitive damages may be pursued where plaintiffs allege egregious conduct, such as knowing concealment of risk. Availability varies significantly by state and by the evidence developed in discovery.
Settlement Value: Why It Is Difficult to Estimate in 2026
Patients often ask for a settlement range. As of 2026, widely circulated public summaries emphasize that it remains difficult to estimate values because:
- Large bellwether verdicts may not yet exist (or are not yet conclusive)
- Liability, causation, and damages vary widely among individual plaintiffs
- The litigation posture may shift if an MDL is formed, if new studies emerge, or if labeling changes occur
In practice, valuation depends on:
- Confirmed diagnosis subtype (CTCL stage and progression)
- Treatment burden and prognosis
- Proof of exposure and timeline
- Evidence supporting diagnostic delay
- Workup history, biopsies, pathology confirmation
- Economic losses, age, and life impact
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected].

What Patients Should Do Now (Without Assuming the Worst)
A forward-looking approach is appropriate here: proactive documentation, proactive medical follow-up, and proactive legal rights preservation. That is true whether you plan to file a claim or simply want to protect your options.
1) Do not self-discontinue medication
Medical decisions must be individualized. Abruptly stopping therapy can lead to serious flares, asthma destabilization, or other complications. Discuss risks with your prescribing clinician.
2) If symptoms are atypical or worsening, request a diagnostic re-check
Ask about:
- Dermatology re-evaluation
- Skin biopsy (or repeat biopsy if prior results do not match clinical behavior)
- Referral to a cutaneous lymphoma specialist if suspicion persists
3) Preserve records early
If you are concerned about CTCL or lymphoma risk, preserve:
- Dupixent prescription records and dosing history
- Dermatology notes, photographs, pathology reports
- Biopsy results, immunophenotyping, staging workup
- Oncology records and treatment timeline
A clear timeline is often the foundation for both clinical clarity and legal evaluation.
4) Track symptom progression
Maintain a dated log of:
- Rash distribution, severity, and new lesion types
- Systemic symptoms (fatigue, fevers, weight change)
- Response or non-response to treatments
- Lymph node changes
How a Dupixent Lawyer Typically Builds a Case
In drug injury litigation, the early phase often looks less like courtroom drama and more like structured evidence development.
A Dupixent attorney may:
- Obtain complete medical and pharmacy records
- Construct a treatment and symptom timeline
- Identify whether early CTCL signs were present before Dupixent initiation
- Engage independent experts (dermatology, oncology, pharmacovigilance)
- Evaluate whether warning language and risk communication were adequate
- File product liability claims and manage litigation deadlines
- Negotiate with defendants or proceed toward trial if necessary
Early medical record review is frequently emphasized because it can prevent avoidable delays and preserve key documents that later become difficult to obtain.
For further guidance on managing potential drug-related injuries such as those from Dupixent, consider exploring this comprehensive patient guide which offers valuable insights into understanding complex conditions like CTCL.
Statute of Limitations: The Deadline That Can Decide Everything
Many states allow one to three years to file a product liability claim, but the rule is not uniform. The triggering date can vary, and exceptions may apply.
Because the clock may begin running at diagnosis or at reasonable discovery of the connection, patients who wait too long can lose the right to pursue any compensation, regardless of the merits. A time-sensitive evaluation is often the most practical step for anyone diagnosed with CTCL after Dupixent exposure.
Contact for Case Review About a Dupixent Cancer Lawsuit (As Provided)
If you want evaluation to see if you may be eligible for a Dupixent Cancer Lawsuit, based on the contact information you provided, you may contact:
- Timothy L. Miles
- Phone: (855) 846–6529
- Email: [email protected]
If you speak with any attorney, ask direct questions about deadlines, fee structure, record collection, and what documentation they need to assess causation and damages.
The Most Responsible Takeaway for 2026
Dupixent remains an important biologic for multiple inflammatory conditions, and many patients benefit substantially. However, concerns about cutaneous T-cell lymphoma (CTCL) and other T-cell lymphomas have intensified due to accumulating reports and observational findings that have been characterized as a potential safety signal.
For patients, the path forward should be deliberate and evidence-driven:
- Re-check the diagnosis when symptoms do not behave like eczema.
- Escalate evaluation when red flags appear.
- Preserve medical records and timelines.
- Protect your legal rights under state deadlines if you have been diagnosed with CTCL after Dupixent exposure.
In a landscape defined by evolving evidence, proactive measures matter. Clarity matters. Documentation matters. Early action is often the difference between unanswered questions and an informed, defensible path forward.
FAQs (Frequently Asked Questions)
What is Dupixent and what conditions is it prescribed for?
Dupixent (dupilumab) is a prescription biologic injection used to treat inflammatory diseases such as moderate to severe atopic dermatitis (eczema), asthma, chronic rhinosinusitis with nasal polyps, and eosinophilic esophagitis. It works by targeting interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling pathways.
What are the safety concerns associated with Dupixent?
There is an ongoing safety debate about whether Dupixent use may be linked to an increased risk of certain lymphomas, particularly cutaneous T-cell lymphoma (CTCL), a rare form of non-Hodgkin lymphoma that can initially resemble eczema or dermatitis. Reports, case literature, and observational research have raised these concerns.
What is cutaneous T-cell lymphoma (CTCL) and how is it related to Dupixent?
CTCL is a cancer of T-lymphocytes primarily involving the skin. Early CTCL can mimic common inflammatory skin diseases like eczema, leading to delayed diagnosis. Some evidence suggests that Dupixent may accelerate or unmask CTCL in patients previously misdiagnosed with eczema, though definitive causality has not been established.
Are there lawsuits related to Dupixent and Cancer?
Yes, several individuals have filed a Dupixent Cancer Lawsuit against Dupixent manufacturers alleging they failed to provide adequate warnings about the potential risk of developing T-cell lymphomas such as CTCL. These legal actions focus on whether sufficient risk communication was provided as evidence accumulated.
How do experts differentiate between safety signals, associations, and causality concerning Dupixent and Cancer risks?
A safety signal is a pattern of reports indicating a possible risk that needs further evaluation but is not proof. An association means a statistical relationship observed in data but does not confirm causation. Causality requires strong evidence showing the drug likely caused the outcome, including consistent studies and plausible mechanisms. Currently, only an emerging safety signal related to CTCL has been reported for Dupixent without definitive causal conclusions.
Should patients stop using Dupixent due to these safety concerns?
Patients are advised not to stop or change their prescribed medication without consulting their treating clinician. While there are ongoing investigations into Dupixent’s safety profile regarding lymphoma risk, any treatment decisions should be made in consultation with healthcare professionals considering individual patient circumstances.

