Introduction to the Dupixent Cancer Lawsuit
Dupixent Cancer Lawsuit: Lawsuits have emerged alleging that the manufacturers failed to provide proper warnings about Dupixent and Cancer Risks, specifically the potential for developing lymphoma. These legal actions claim that patients prescribed dupilumab were not adequately informed about the cancer risks associated with the medication, constituting a failure to warn. This situation is similar to other recent cases involving medications like Zepbound which have also faced legal scrutiny due to serious side effects such as vision loss.
Dupixent (dupilumab): Is a biologic medication manufactured by Sanofi and Regeneron, has become a popular treatment option for patients with inflammatory conditions such as moderate-to-severe eczema, asthma, chronic obstructive pulmonary disease (COPD), and chronic rhinosinusitis with nasal polyps. It works by targeting specific immune system pathways that cause inflammation, providing relief to millions of patients worldwide since its U.S Food and Drug Administration (FDA) approval.
Clinical Studies: However, recent scientific evidence has raised serious concerns about the safety of this widely prescribed medication. Studies conducted between 2024 and 2025 have found a possible link between Dupixent use and an increased risk of developing cutaneous T-cell lymphoma (CTCL), a rare and aggressive form of non-Hodgkin lymphoma. These findings have led to questions about whether Sanofi and Regeneron adequately warned patients and healthcare providers about these serious Dupixent and cancer risks.
Dupixent Cancer Lawsuit: For those affected by such severe outcomes following treatment with these biologic medications, seeking legal action is often necessary. Law firms that focus on class action lawsuits, such as the Law Offices of Timothy L. Miles, offer valuable resources and guidance in navigating these complex legal matters.
A Failure to Warn: This article explores the legal aspects of these claims, the scientific evidence supporting the link between Dupixent and CTCL, and the implications for patients who have developed lymphoma after using this biologic medication.
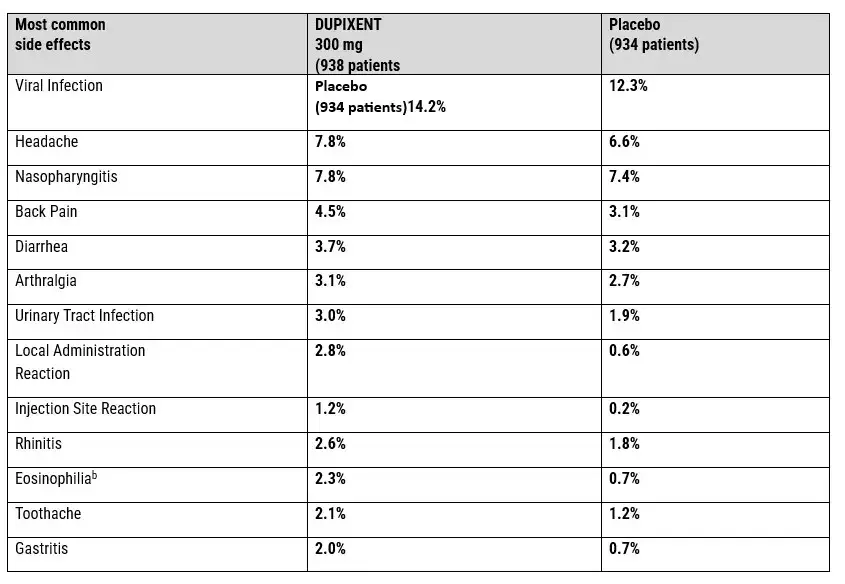
DUPIXENT COMMON SIDE EFFECTS PROFILE
Understanding Dupixent: Mechanism and Uses
Dupixent (dupilumab): Is an advanced biologic medication, specifically designed as a monoclonal antibody to target specific immune system pathways responsible for chronic inflammatory responses. The FDA has approved this treatment for various inflammatory conditions, highlighting its wide-ranging effectiveness in managing diseases caused by persistent immune dysfunction.
FDA-Approved Indications
The regulatory approval for Dupixent includes several distinct medical conditions:
- Atopic dermatitis (moderate-to-severe eczema) in patients aged six months and older
- Moderate-to-severe asthma with an eosinophilic phenotype or oral corticosteroid-dependent asthma in patients aged six years and older
- Chronic rhinosinusitis with nasal polyps in adults
- Eosinophilic esophagitis in patients aged one year and older weighing at least 15 kilograms
- Prurigo nodularis in adults
- Chronic obstructive pulmonary disease (COPD) with an eosinophilic phenotype
This wide range of approved uses shows the medication’s flexibility in treating inflammatory diseases across different organ systems and patient groups.
Dupixent Mechanism: Blocking Interleukin Pathways
Mechanism of Action: The therapeutic action of Dupixent centers on its ability to inhibit interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling pathways. These cytokines play central roles in type 2 inflammatory responses, driving the cascade of immune reactions that characterize conditions such as atopic dermatitis and asthma. By binding to the IL-4 receptor alpha subunit, dupilumab prevents both IL-4 and IL-13 from activating their respective signaling pathways, thereby reducing the inflammatory cascade at its source.
Therapeutic Efficacy: This targeted approach sets biologic medications apart from traditional immunosuppressants, as the intervention focuses on specific molecular pathways rather than broadly suppressing immune function. The precision of this mechanism theoretically reduces the risk of widespread immunosuppression while maintaining therapeutic efficacy against inflammatory diseases.
Previously Documented Dupixent Side Effects
Documented Dupixent Side Effects: Clinical trials and post-marketing surveillance identified several adverse reactions associated with Dupixent use prior to emerging cancer concerns:
- Injection site reactions including erythema, swelling, and pruritus
- Conjunctivitis and other ocular surface inflammatory conditions
- Oral herpes and cold sores
- Eosinophilia (elevated eosinophil counts)
- Arthralgia (joint pain)
- Nasopharyngitis and upper respiratory tract infections
Safety Profile: These documented side effects formed the basis of the original risk-benefit assessment for Dupixent. The subsequent identification of potential malignancy risks, particularly cutaneous T-cell lymphoma, has prompted reconsideration of the medication’s safety profile and raised questions about whether adequate warnings accompanied the product’s distribution and marketing.

The Link Between Dupixent and Cancer Risks: What Studies Reveal
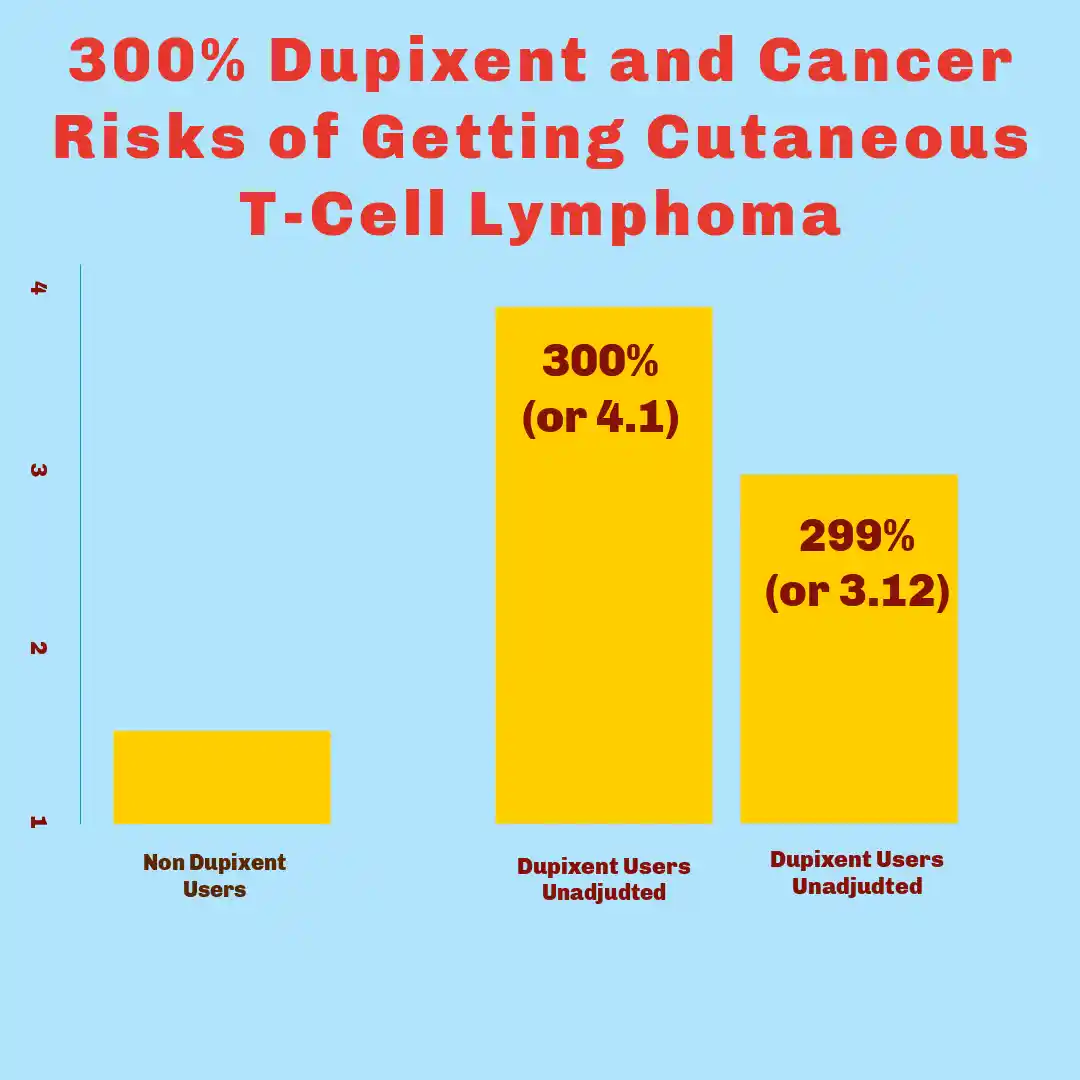
The Link Between Dupixent and Cancer: Recent scientific investigations have brought significant attention to the Dupixent and Cancer Risks, particularly concerning cutaneous T-cell lymphoma (CTCL). Research published between 2024 and 2025 has identified a statistically significant correlation between dupilumab use and the development of this rare malignancy. The dupilumab lymphoma studies 2024 demonstrate an elevated risk profile that has prompted legal scrutiny and patient safety concerns.
Quantifying the Dupixent CTCL Risk
High Dupixent and Cancer Risks: Epidemiological analyses conducted in 2024 revealed alarming statistics regarding Dupixent and cancer. The studies documented a 2 to 4.5 times increased risk of developing CTCL among patients who received dupilumab treatment compared to those who did not use the medication. This risk elevation represents a substantial departure from baseline population rates and has raised questions about the adequacy of pre-market safety testing and post-market surveillance protocols.
Risk Higher for Certain Patient Subgroups: The variability in risk ratios—ranging from twofold to more than fourfold—reflects differences in study methodologies, patient populations examined, and duration of drug exposure analyzed. Certain patient subgroups demonstrated higher susceptibility, suggesting that individual risk factors may interact with dupilumab’s immunomodulatory effects to create conditions favorable for lymphoma development.
Understanding Cutaneous T-Cell Lymphoma (CTCL)
Cutaneous T-cell lymphoma (CTCL): Represents a rare and aggressive variant of non-Hodgkin lymphoma characterized by malignant transformation of T-lymphocytes that primarily manifest in the skin. This cancer type differs fundamentally from other lymphomas due to its cutaneous presentation and the specific immune cells involved in its pathogenesis.
Subtypes: CTCL encompasses several subtypes, with mycosis fungoides and Sézary syndrome being the most common presentations. The disease typically progresses through distinct stages:
- Early stage: Patches or plaques on the skin resembling eczema or psoriasis
- Advanced stage: Tumors, erythroderma, and potential systemic involvement
- Metastatic stage: Spread to lymph nodes, blood, and internal organs
Treatment Options: The aggressive nature of CTCL necessitates prompt diagnosis and intervention. Treatment options include topical therapies, phototherapy, systemic medications, and in severe cases, chemotherapy or stem cell transplantation. The prognosis varies significantly depending on the stage at diagnosis and the specific subtype identified.
Clinical Manifestations and Diagnostic Challenges
Dupixent Side Effects: The symptoms associated with CTCL linked to Dupixent use present a diagnostic dilemma for healthcare providers. Patients may experience:
- Persistent dry skin that fails to respond to standard moisturizing treatments
- Unexplained rashes that differ in appearance from typical eczema flares
- Patches of discolored or thickened skin
- Intense pruritus (itching) disproportionate to visible skin changes
- Lymph node swelling in the neck, armpits, or groin
- Unexplained fatigue and malaise
Dupixent and Cancer Risks: The overlap between these CTCL symptoms and the underlying inflammatory conditions for which Dupixent is prescribed creates significant diagnostic complexity. Eczema patients routinely experience dry skin and rashes, making it challenging to distinguish between disease activity and potential malignancy. This symptomatic overlap may have delayed recognition of cancer development in some patients, allowing the disease to progress to more advanced stages before definitive diagnosis.
FDA Investigation into Dupixent: Safety Data and Concerns Raised
FDA Investigation: The FDA’s Adverse Event Reporting System (FAERS) serves as a critical repository for documenting potential safety concerns associated with pharmaceutical products, including biologics like Dupixent. This database collects voluntary reports from healthcare professionals, patients, and manufacturers regarding adverse events that occur during or after medication use. The accumulation of reports linking dupilumab to lymphoma cases has prompted regulatory scrutiny and raised questions about the adequacy of existing safety warnings.
Adverse Event Reports in the FAERS Database
Patient Reports: Submitted to the FAERS database have documented cases of cutaneous T-cell lymphoma and other lymphoproliferative disorders among individuals prescribed Dupixent for various inflammatory conditions. These reports detail the temporal relationship between drug initiation and the development or progression of malignancies, providing regulatory authorities with real-world evidence of potential safety signals. The database contains information on patient demographics, dosing regimens, concurrent medications, and clinical outcomes, enabling pattern recognition across diverse patient populations.
Dupixent and Cancer Risks: Healthcare providers and patients have reported instances where lymphoma symptoms emerged during active Dupixent therapy or within months of treatment initiation. The reports describe presentations ranging from isolated skin lesions to systemic involvement with lymphadenopathy and organ infiltration. The voluntary nature of FAERS reporting means the actual incidence of adverse events may exceed documented cases, as not all events are reported to the system.
Regulatory Response and Investigation Status
Regulatory Response: The FDA’s evaluation of safety data involves analyzing individual case reports, reviewing clinical trial data, and assessing epidemiological studies to determine whether a causal relationship exists between dupilumab exposure and cancer development. Regulatory authorities examine whether reported malignancies represent background rates expected in the treated population or constitute a drug-related safety signal requiring label modifications or risk mitigation strategies.
The agency’s investigation into potential cancer risks linked to dupilumab use encompasses several analytical approaches:
- Signal detection analysis comparing observed versus expected rates of lymphoma in Dupixent-exposed populations
- Review of preclinical and clinical trial data for evidence of carcinogenic potential or immunological mechanisms that could promote malignancy
- Assessment of biological plausibility regarding how IL-4 and IL-13 pathway blockade might influence cancer surveillance or tumor development
- Evaluation of confounding factors such as underlying disease severity and prior treatment exposures that could contribute to cancer risk
The FDA investigation Dupixent Process: Involves collaboration with the manufacturers, Sanofi and Regeneron, requiring submission of post-marketing safety data and analysis of their pharmacovigilance databases. The regulatory timeline for completing such investigations varies based on the complexity of the safety signal and the availability of definitive evidence. The emerging concerns documented in the Adverse Event Reporting System FAERS have become central to the Dupixent Cancer Lawsuit claims, as plaintiffs allege manufacturers possessed safety data that warranted enhanced warnings to prescribers and patients.
Legal Grounds for the Dupixent Cancer Lawsuit: Failure to Warn Claims Explained
Failure to warn lawsuits are a specific type of product liability claims based on the principle that manufacturers have a responsibility to inform consumers and healthcare providers about known or reasonably foreseeable risks associated with their products. The legal actions against Sanofi and Regeneron Pharmaceuticals allege that these companies failed to provide sufficient warnings regarding the potential connection between Dupixent use and the development of cutaneous T-cell lymphoma.
The Core Allegations Against Manufacturers
Dupixent Cancer Lawsuit: The lawsuits assert that Sanofi and Regeneron knew—or should have known through reasonable investigation—about the increased CTCL risk among Dupixent users. Plaintiffs argue that despite this knowledge, the pharmaceutical companies:
- Failed to update prescribing information to adequately reflect the cancer risk
- Neglected to communicate emerging safety data to prescribing physicians through appropriate channels
- Omitted critical risk information from patient medication guides and direct-to-consumer marketing materials
- Continued aggressive marketing campaigns without disclosing the potential for serious adverse events
Negligence Across the Product Lifecycle
The legal claims go beyond simple communication failures and include broader allegations of negligence throughout the manufacturing, marketing, and distribution processes. Plaintiffs argue that the defendants breached their duty of care by:
Manufacturing Negligence: Allegations suggest inadequate post-market surveillance systems to identify and respond to emerging safety signals related to lymphoma development in patient populations using dupilumab.
Marketing Negligence: Claims assert that promotional materials emphasized benefits while minimizing or entirely excluding information about serious risks, creating an imbalanced representation of the drug’s safety profile that influenced prescribing decisions.
Distribution Negligence: The Dupixent Lawsuits contend that the companies maintained distribution channels without implementing adequate risk mitigation strategies or restricted access programs that might have limited exposure among particularly vulnerable patient populations.
The Legal Standard for Pharmaceutical Warnings
Product Liability Law: Requires pharmaceutical manufacturers to provide warnings that adequately convey the nature, severity, and likelihood of known risks. The warnings must be:
- Scientifically accurate and based on current medical knowledge
- Sufficiently detailed to enable informed decision-making by healthcare providers
- Prominently displayed and clearly communicated to ensure visibility
- Updated promptly as new safety information becomes available through clinical experience or research
Failure to Warn: The failure to meet these standards forms the basis of the legal claims against Dupixent’s manufacturers, with plaintiffs asserting that proper warnings would have changed treatment decisions and prevented their diagnoses of cutaneous T-cell lymphoma.
Severe Side Effects: In addition to these concerns, there are also reports highlighting severe side effects associated with other medications such as Zepbound. For instance, Zepbound Vision Loss Lawsuit has emerged as a significant legal issue, drawing attention from patients who have experienced vision loss due to this medication. Similar to the Dupixent case, these lawsuits underscore the critical need for pharmaceutical companies to disclose all potential risks associated with their products.

Who is Eligible for a Dupixent Cancer Lawsuit? Understanding the Criteria
Eligibility: Determining who can file a lawsuit for Dupixent cancer requires careful evaluation of specific medical and timing factors that connect the medication to harmful health effects. Individuals looking to qualify for a Dupixent cancer lawsuit must meet certain evidence standards that show a direct link between taking dupilumab and developing cancer.
Primary Eligibility Requirements
The foundation of any Dupixent lawsuit rests upon documented medical evidence establishing the following criteria:
1. Confirmed CTCL Diagnosis
Potential plaintiffs must have a definitive diagnosis of cutaneous T-cell lymphoma made by qualified oncology or dermatology specialists. This diagnosis usually involves skin biopsy results, immunohistochemistry testing, and pathological examination showing malignant T-cell growth. The diagnosis must meet established clinical criteria for CTCL classification, including mycosis fungoides or Sézary syndrome variants.
2. Documented Dupixent Use
Medical records must show that dupilumab was prescribed for FDA-approved uses such as atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyps, or eosinophilic esophagitis. Prescription history, pharmacy records, and treatment logs establish how long and how much medication was taken.
3. Temporal Relationship
The importance of medical records linking drug use with the start or worsening of lymphoma symptoms cannot be overstated. Eligible claimants must demonstrate that CTCL symptoms appeared during active Dupixent therapy or within a medically reasonable time after starting treatment. Documentation should reflect the progression of symptoms—including persistent skin lesions, unexplained rashes, swollen lymph nodes, or systemic manifestations—corresponding with the period when the medication was being used.
Supporting Documentation Standards
Medical Records: Comprehensive medical documentation strengthens eligibility determination. Records should include baseline dermatological assessments prior to Dupixent initiation, treatment monitoring notes, laboratory results indicating abnormal lymphocyte populations, imaging studies revealing lymph node involvement, and oncological consultations confirming malignancy diagnosis. The evidentiary chain must establish that CTCL development represents a departure from the patient’s pre-treatment health status, attributable to dupilumab exposure rather than pre-existing conditions or alternative causative factors.
Finding a Qualified Dupixent Cancer Lawyer: Tips for Your Search
When looking for a lawyer to represent you in a Dupixent Cancer Lawsuit, it is important to find someone who has the right aqualifications and experience. These lawsuits can be complicated, so you’ll need a lawyer who knows how to take on big pharmaceutical companies and understands the science behind biologic medication claims.
Essential Qualifications for Dupixent Cancer Lawyers
Here are some key qualifications to look for in a lawyer handling Dupixent cancer cases:
- Pharmaceutical litigation history: They should have a track record of representing plaintiffs in drug injury cases against large pharmaceutical corporations such as Sanofi and Regeneron.
- Biologic medication knowledge: It’s important that they understand how biologic drugs work, including the mechanisms of IL-4 and IL-13 pathway inhibition.
- Lymphoma expertise: Look for a lawyer who is familiar with cutaneous T-cell lymphoma (CTCL) diagnosis, treatment protocols, and relevant medical literature establishing causation links.
- Failure to warn experience: They should have proven success in cases involving inadequate safety warnings and negligence on the part of pharmaceutical manufacturers.
- Resource capacity: Make sure they have access to medical experts, toxicologists, and scientific consultants who can help build strong cases.

Compensation in a Dupixent Cancer Lawsuit: What You Need to Know
Plaintiffs pursuing compensation in a Dupixent cancer lawsuit may seek recovery for multiple categories of damages resulting from cutaneous T-cell lymphoma diagnoses allegedly linked to dupilumab use. The scope of compensation dupilumab cancer lawsuit claims encompasses both quantifiable economic losses and non-economic harm stemming from the manufacturer’s alleged failure to provide adequate warnings about cancer risks.
Economic Damages
Medical expenses: Constitute a primary component of compensation claims in pharmaceutical litigation involving Dupixent. These costs include:
- Diagnostic procedures for CTCL confirmation, including biopsies and imaging studies
- Chemotherapy, radiation therapy, or immunotherapy treatments
- Hospitalization expenses for intensive cancer care
- Prescription medications for managing lymphoma symptoms
- Ongoing monitoring and follow-up appointments with oncologists and dermatologists
- Future medical care projected for continued CTCL management
Lost wages and diminished earning capacity represent another critical element of economic damages. Plaintiffs may recover compensation for income lost during treatment periods, reduced work hours necessitated by illness, and permanent impairment of professional capabilities resulting from CTCL complications.
Non-Economic Damages
Pain and Suffering: The physical suffering and psychological distress associated with cancer diagnoses warrant compensation for pain and suffering. CTCL patients experience significant discomfort from skin lesions, systemic symptoms, and the emotional trauma of confronting a rare and aggressive malignancy.
Loss of quality of life damages address the profound impact lymphoma has on daily activities, personal relationships, and overall well-being. The disfigurement and chronic health challenges associated with cutaneous T-cell lymphoma substantially diminish patients’ ability to enjoy life as they did before diagnosis.
Punitive Damages
In cases demonstrating egregious conduct by pharmaceutical manufacturers—such as deliberate concealment of known cancer risks or reckless disregard for patient safety—courts may award punitive damages designed to punish wrongdoers and deter similar behavior across the pharmaceutical industry.
Current Status and Future Outlook of the Litigation: What Lies Ahead?
Dupixent Cancer Lawsuit: The dupilumab lawsuits entered a significant phase with the filing of the first federal complaint in Tennessee federal court during early 2025. This inaugural case established the legal framework for subsequent claims against Sanofi and Regeneron, centering on allegations that the pharmaceutical manufacturers failed to provide adequate warnings about cutaneous T-cell lymphoma risks associated with Dupixent use.
Preliminary Stages: The case timeline remains in preliminary stages, with discovery proceedings expected to unfold throughout 2025. Plaintiffs’ attorneys are gathering medical records, internal company documents, and expert testimony to substantiate claims that the defendants possessed knowledge of cancer risks yet failed to communicate these dangers to healthcare providers and patients.
Multidistrict Litigation: Legal observers anticipate the possibility of MDL status (Multi-District Litigation) consolidation if additional plaintiffs file similar claims across multiple federal jurisdictions. MDL designation would centralize pretrial proceedings before a single judge, streamlining discovery processes and promoting consistent rulings on common legal questions. This procedural mechanism has proven effective in managing complex pharmaceutical litigation involving numerous plaintiffs with shared allegations against the same defendants.
The trajectory of future claims depends heavily on several factors:
- Outcomes of ongoing FDA investigations into Dupixent safety data
- Publication of additional peer-reviewed studies examining the dupilumab-CTCL connection
- Disclosure of internal company communications regarding risk assessment
- Resolution of initial bellwether trials that may establish precedent for settlement negotiations
Conclusion
The Dupixent Cancer Lawsuit: Is an important case that holds pharmaceutical companies accountable. It focuses on accusations that the manufacturers did not properly inform patients and healthcare providers about the potential risks of cutaneous T-cell lymphoma associated with dupilumab treatment. The legal principles behind failure to warn claims clearly state that pharmaceutical companies have a responsibility to disclose significant risks that could impact treatment choices.
Dupixent Side Effects: Patients who have been prescribed Dupixent for inflammatory conditions have a reasonable expectation to be fully informed about all major side effects, including rare but severe cancers. The discovery of scientific evidence indicating a higher occurrence of CTCL among users raises important questions about the effectiveness of pre-market testing procedures and post-market monitoring systems.
Dupixent and Cancer: The legal situation is constantly changing as more individuals come forward with documented diagnoses after receiving Dupixent treatment. A Dupixent Cancer Lawsuit will likely investigate internal communications, clinical trial information, and adverse event reporting practices to determine whether Sanofi and Regeneron were aware of cancer risks that required earlier disclosure.
Documentation and Legal Advice: If you have been diagnosed with cutaneous T-cell lymphoma after using Dupixent, it is crucial to keep all medical records that establish a connection between the drug and the onset of symptoms. Seeking advice from experienced attorneys specializing in pharmaceutical litigation is vital to assess potential claims and understand the available legal options. The outcomes of these cases will have an impact on future regulations regarding safety disclosures for biologic medications and transparency obligations for manufacturers.
FREQUENTLY ASKED QUESTIONS THE DUPIXENT LAWSUIT
What are the potential benefits of a Dupixent Cancer Lawsuit?
How long will the Dupixent Cancer Lawsuit take to resolve?
What Is Cutaneous T-cell lymphoma?
-
What it is: A type of non-Hodgkin lymphoma that starts in T-cells in the skin.
-
Common symptoms:
-
-
Itchy, scaly patches or plaques
-
-
-
Redness on the skin
-
-
-
Thicker, raised lesions or tumors
-
-
-
Sometimes, the skin redness can cover a large portion of the body, a condition called erythroderma
-
-
Progression: CTCL is often slow-growing, but some types can be aggressive and spread to other parts of the body, such as the lymph nodes, blood, or organs like the liver or lungs.
-
Diagnosis: Diagnosis can be challenging due to similar-looking symptoms and often involves a skin biopsy, physical exam, blood tests, and imaging.
-
Treatment: Treatments are chosen based on the stage of the cancer and can include creams, light therapy, radiation, or systemic treatments like chemotherapy or other medications.
How can I join the Dupixent Lawsuit?
Is There a Dupixent Class Action Lawsuit?
What is cutaneous T-cell lymphoma (CTCL)?
How many different types of CTCL are there?
-
Mycosis Fungoide (MF) – The most common type, characterized by patches, plaques, and tumors on the skin.
-
Sézary Syndrome (SS) – A more aggressive form of MF that affects the blood as well as the skin.
-
Lymphomatoid papulosis – A benign form of CTCL that causes small, itchy bumps on the skin.
-
Granulomatous slack skin – A rare, indolent form of CTCL that causes loose, pendulous folds of skin.
-
Anaplastic large cell lymphoma – An aggressive form of CTCL that can spread to other organs.
-
Extranodal NK/T-cell lymphoma: A rare type of CTCL that affects the skin and lymph nodes.
-
Extranodal NK/T-cell lymphoma, nasal type – A rare, aggressive lymphoma that can affect the skin, among other places.
-
Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) – A rare, slow-growing type that causes nodules deep in the fatty layer of the skin, most often on the legs.
-
Primary cutaneous peripheral T-cell lymphoma (unspecified) – This is a category for rare entities that do not fit into other classifications.

