Introduction to Saxenda Vision Changes
Saxenda vision changes hae effected numerous individuals which has led to numerous lawsuits against the manufacture. Saxenda (liraglutide) is an important breakthrough in the field of weight management medications. It works as a glucagon-like peptide-1 (GLP-1) receptor agonist, helping to control appetite and regulate glucose metabolism.
Saxenda operates by imitating the action of natural incretin hormones in the body. These hormones send signals to the brain indicating that we are full and also slow down the process of emptying our stomachs. As a result, this leads to gradual and sustained weight loss in adults who are classified as obese or overweight, especially those with weight-related health issues.
As more people start using Saxenda and other similar GLP-1 receptor agonists in 2025, new research has shown that both patients and healthcare providers need to be aware of possible eye-related problems that may arise from this treatment. One specific concern that has come up is Saxenda dry eyes, which has been reported as a common Saxenda vision side effects. However, it’s also important to pay attention to other potential Saxenda Eye Problems.
Being aware of these possible complications is becoming increasingly crucial as we gather more long-term safety data and treat a wider range of patients.
This guide aims to provide comprehensive information for patients regarding various vision changes associated with Saxenda. It covers both common symptoms such as dry eye syndrome and less frequently reported issues like blurry vision and floaters. Additionally, it addresses rare but severe conditions that could potentially lead to permanent vision loss.
By understanding these potential complications, patients can make better decisions about their treatment options and be proactive in seeking medical attention when necessary.
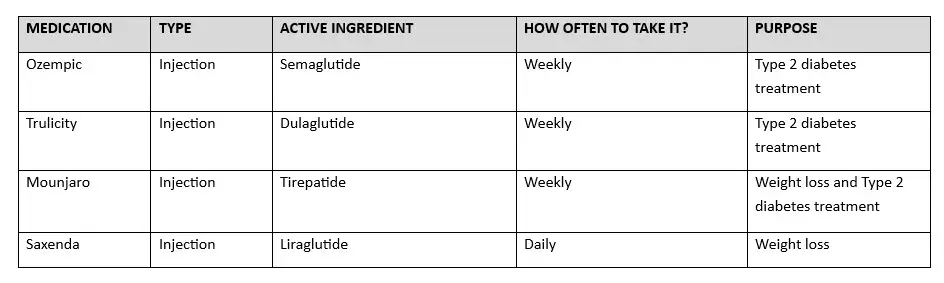
SAXENDA COMPARISON TO OTHER GLP-1 DRUGS
Dry Eyes: The Most Common Vision Side Effect of Saxenda
Dry eyes have become an eye problem reported by patients using Saxenda, a medication that helps with weight loss. Both doctors and patients have noticed that this treatment can cause irritation on the surface of the eyes, which needs to be addressed by healthcare professionals and those undergoing therapy.
Symptoms of Dry Eyes Caused by Saxenda
Patients experiencing this condition typically report a range of symptoms that interfere with daily visual comfort:
- Persistent feeling of grittiness or something foreign in the eyes
- Burning or stinging sensations, especially during long periods of looking at screens
- Excessive tearing as a response to insufficient tear film stability
- Redness and swelling of the conjunctival tissues
- Increased sensitivity to environmental factors such as wind, air conditioning, or screen exposure
- Difficulty wearing contact lenses due to reduced eye moisture
How Saxenda May Cause Dry Eyes
The exact reasons why Saxenda may lead to dry eyes are not fully understood, but there are several possible explanations:
- Effects on Tear Production: Research suggests that liraglutide, the active ingredient in Saxenda, may affect how tears are produced and what they consist of. GLP-1 receptors, which are activated by liraglutide, can be found in various parts of the eye including the glands responsible for making tears.
- Impact on Autonomic Nervous System: The autonomic nervous system controls many involuntary bodily functions, including tear production. It’s possible that Saxenda’s effects on metabolism and inflammation could also influence this system and lead to changes in tear production.
- Existing Conditions: Patients who already have conditions like diabetes or autoimmune disorders may be more prone to developing dry eyes while taking Saxenda because their baseline tear film stability is already compromised.
Importance of Monitoring Dry Eyes in Saxenda Users
Since dry eyes are such a common side effect of Saxenda, it is important for both healthcare providers and patients to keep an eye out for any signs or symptoms related to this issue. By being proactive about monitoring and addressing dry eye concerns early on, we can help prevent them from becoming more severe problems down the line.
Other Saxenda Vision Changes Linked to its Use
Beyond dry eyes, patients using the medicine have documented a spectrum Saxenda vision changes that warrant clinical attention. These Saxenda vision problems extend to various ocular manifestations, each presenting distinct challenges for individuals undergoing weight management therapy with this GLP-1 receptor agonist.
Blurry Vision and Visual Disturbances
Saxenda blurry vision represents a significant concern among patients, with reports indicating episodes of reduced visual acuity that can occur at various points during treatment. These Saxenda vision side effects manifest as:
- Intermittent loss of visual clarity affecting near or distance vision
- Difficulty focusing on objects, particularly during reading or computer work
- Fluctuating vision quality throughout the day, often correlating with medication timing
- Temporary inability to distinguish fine details or read small print
The impact of Saxenda and blurry vision on daily life extends beyond mere inconvenience. Patients have reported challenges with essential activities such as driving, particularly during nighttime conditions, and performing work-related tasks requiring visual precision. The unpredictable nature of these episodes creates additional concern, as individuals cannot anticipate when visual clarity may diminish. Some patients describe the sensation as viewing objects through a fog or experiencing a film-like obstruction across their visual field.
The mechanism underlying these Saxenda vision side effects may relate to alterations in blood glucose levels, fluid retention patterns, or direct effects on ocular tissues. Clinical observations suggest that some patients experience these symptoms during dose escalation phases, while others report persistent issues throughout their treatment duration.
Saxenda and Eye Floaters
Saxenda and eye floaters represent another dimension of Saxenda vision side effects associated with this medication. Eye floaters appear as small, dark shapes that drift across the visual field, manifesting as:
- Spots or specks that move when attempting to focus on them
- Thread-like strands or cobweb patterns
- Shadow-like formations in peripheral vision
- Increased visibility against bright backgrounds
The significance of these Saxenda eye problems extends beyond typical age-related floaters. Emerging evidence suggests a potential connection between Saxenda use and retinal fluid accumulation, a condition that may precipitate or exacerbate floater development. Retinal swelling, medically termed macular edema, can alter the vitreous humor composition, leading to increased floater formation. This retinal fluid accumulation requires immediate medical evaluation, as it may indicate more serious underlying pathology affecting the posterior segment of the eye.
SAXENDA AND TYPE 2 DIABETICS HAD A 179% HIGHER LIKELIHOOD OF DEVELOPING NAION

Serious Vision Problems Including Vision Loss Associated with Saxenda Use
While most vision-related complications associated with Saxenda remain manageable, rare instances of severe eye conditions have emerged in clinical observations and patient reports. Non-arteritic anterior ischemic optic neuropathy (NAION) represents one of the most concerning complications documented among individuals using this GLP-1 receptor agonist medication. This condition results from inadequate blood flow to the optic nerve, potentially causing sudden and permanent vision loss in affected patients.
Understanding NAION and Its Clinical Presentation
NAION manifests through distinct symptoms that require immediate medical attention:
- Sudden, painless vision loss in one eye, typically noticed upon waking
- Presence of visual field defects, particularly affecting the lower or upper portions of vision
- Altered color perception and reduced visual acuity
- Swelling of the optic disc observable during ophthalmologic examination
The condition develops when blood supply to the optic nerve head becomes compromised, leading to ischemic damage that cannot be reversed through conventional treatment approaches. Patients experiencing these symptoms require urgent evaluation by ophthalmology specialists to confirm diagnosis and implement appropriate management strategies.
Evidence Linking Saxenda to Increased NAION Risk
Recent pharmacovigilance data and clinical studies have identified a potential association between Saxenda and vision loss, specifically regarding NAION incidence. Research examining the risk of NAION with Saxenda use suggests that individuals prescribed GLP-1 receptor agonists may face elevated susceptibility compared to the general population. The mechanisms underlying this association remain under investigation, with hypotheses focusing on vascular effects, metabolic alterations, and inflammatory processes potentially contributing to optic nerve vulnerability.
The emergence of these findings has prompted increased scrutiny from regulatory bodies and healthcare providers. Multiple Saxenda vision loss lawsuits have been filed by patients who developed NAION during treatment, with legal proceedings consolidated through multidistrict litigation frameworks. Patients experiencing sudden vision changes should consult both medical professionals and, when appropriate, a Saxenda vision loss lawyer to understand their rights and available recourse options.
Safety Precautions and Monitoring Recommendations for Patients on Saxenda
Saxenda safety warnings encompass multiple critical areas that patients must understand before starting treatment. The FDA has issued comprehensive guidance regarding the medication’s risk profile, requiring healthcare providers to inform patients of potential adverse events that extend beyond vision-related complications.
FDA Black Box Warnings and Primary Safety Concerns
The medication carries a black box warning regarding thyroid C-cell tumors, including medullary thyroid carcinoma (MTC). Clinical studies in rodents demonstrated dose-dependent and treatment-duration-dependent increases in thyroid C-cell tumors at clinically relevant exposures. Patients with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) should not use Saxenda under any circumstances.
Acute pancreatitis represents another significant safety concern explicitly addressed in Saxenda safety warnings. Patients must recognize symptoms including:
- Persistent severe abdominal pain, often radiating to the back
- Nausea and vomiting accompanying abdominal discomfort
- Elevated pancreatic enzyme levels upon laboratory evaluation
Discontinuation of Saxenda becomes necessary if pancreatitis is suspected, with no resumption of treatment if pancreatitis is confirmed.
Comprehensive Ophthalmologic Monitoring Protocol
Establishing a baseline ophthalmologic assessment prior to Saxenda initiation provides essential reference data for detecting subsequent vision changes. Patients should undergo complete eye examinations that include:
- Visual acuity testing using standardized protocols
- Intraocular pressure measurement to screen for glaucoma risk
- Dilated fundoscopic examination to evaluate retinal health
- Assessment of tear film quality and dry eye indicators
Ongoing monitoring during treatment requires scheduled eye examinations at intervals determined by individual risk factors, pre-existing conditions, and symptom development. Patients with diabetes, hypertension, or pre-existing ocular conditions warrant more frequent ophthalmologic surveillance. Any new visual symptoms—including sudden vision changes, persistent dry eyes, or visual field defects—necessitate immediate ophthalmologic evaluation rather than waiting for scheduled appointments.
SAXENDA COMMON SIDE EFFECTS PROFILE

Legal Considerations for Patients Experiencing Vision Side Effects from Saxenda
Patients who have experienced Saxenda vision side effects may have legal options through ongoing lawsuits against the medication’s manufacturer. The legal situation surrounding these claims has changed significantly, with many individuals reporting permanent vision impairment after using Saxenda and similar GLP-1 receptor agonists.
Understanding Multidistrict Litigation (MDL)
The establishment of multidistrict litigation MDL 3094 is an important development in bringing together vision-related injury claims. This federal court process combines cases that involve accusations that manufacturers did not adequately warn patients and healthcare providers about the risk of non-arteritic anterior ischemic optic neuropathy (NAION) and other serious eye complications.
Key Benefits of the MDL Process
The MDL process offers several advantages for handling these cases:
- Streamlined pretrial proceedings: By consolidating similar cases, the MDL process allows for more efficient management of pretrial activities such as discovery.
- Potentially expedited resolution: With multiple cases being heard together, there is a possibility of faster outcomes for claims involving common factual and legal issues.
Legal Claims Against Pharmaceutical Companies
Plaintiffs in these cases argue that pharmaceutical companies were aware of potential vision risks but failed to provide adequate warnings in product labeling or marketing materials. Common legal claims include:
- Failure to warn about known risks
- Defective design of the medication
- Negligent misrepresentation of safety profiles
- Breach of implied warranty
Preserving Evidence for Your Case
Patients who have experienced vision changes or loss while taking Saxenda should take steps to preserve relevant medical records. This includes:
- Ophthalmological examinations: Keep copies of any eye exams conducted by an eye specialist.
- Diagnostic imaging: If you underwent any imaging tests such as OCT (optical coherence tomography) or fundus photography, obtain those reports.
- Treatment documentation: Maintain records of all treatments received for your vision issues, including medications prescribed or procedures performed.
Seeking Legal Guidance
Consulting with a lawyer who practices in pharmaceutical litigation can provide valuable insights into your eligibility for compensation and the strength of your potential claims. They can help you navigate the complexities of the legal system and advocate on your behalf.
Taking Action: Managing Eye Side Effects While Using Saxenda Responsibly
Patients experiencing eye symptoms while undergoing Saxenda treatment can implement several evidence-based strategies to reduce discomfort and protect their vision. Managing Saxenda eye problems requires a proactive approach that balances therapeutic benefits with eye health considerations.
Practical Strategies for Alleviating Dry Eyes
Artificial tears and preservative-free lubricating eye drops provide immediate relief for dry eye symptoms. Patients should apply these solutions multiple times daily, particularly during activities that worsen dryness such as prolonged screen time or exposure to air conditioning. Humidifiers in living and working spaces maintain optimal moisture levels, reducing environmental factors that contribute to eye discomfort. Omega-3 fatty acid supplementation supports tear film stability and may reduce inflammatory processes affecting the surface of the eye.

When to See a Specialist
If patients experience persistent or worsening vision changes, they should seek immediate evaluation from an eye doctor. Specialist consultation is necessary when the following occurs:
- Sudden vision loss or significant visual field defects
- Progressive blurriness unresponsive to conservative management
- New onset of floaters accompanied by flashes of light
- Severe eye pain or persistent redness
Alternative weight management therapies should be considered when eye side effects negatively impact quality of life or pose risks to visual function. Endocrinologists and ophthalmologists can work together to determine whether continuing Saxenda treatment aligns with individual risk-benefit profiles, especially for patients with pre-existing retinal conditions or diabetes-related eye disease.
Conclusion
Patient awareness 2025 requires proactive involvement in all aspects of medication safety, especially when it comes to eye health during Saxenda treatment. People who are thinking about or currently using this weight management therapy must make saxenda vision monitoring an essential part of their healthcare routine.
The information shared in this Saxenda Dry Eyes: A Patient Guide [2025] emphasizes the importance of being watchful for any changes in vision, whether it’s minor discomfort from dry eyes or more serious conditions that need immediate medical attention. Here are the responsibilities that patients have:
- Scheduling thorough eye exams before starting therapy
- Going for regular eye check-ups throughout the entire treatment period
- Keeping track of any changes in vision, no matter how insignificant they may seem
- Informing healthcare providers promptly about any eye symptoms
- Knowing the difference between manageable side effects and warning signs of serious complications
It’s crucial for patients, prescribing doctors, and eye specialists to work together and carefully consider the benefits of Saxenda against any potential risks to vision. By paying close attention to eye health, we can catch any negative effects early on, which will allow us to intervene promptly and protect our eyesight throughout the entire treatment process.
Frequently Asked Questions About Saxenda Vision Side Effects
Can Saxenda cause vision loss?
Yes, recent studies suggest a possible link between Saxenda and vision loss, specifically a condition called non-arteritic anterior ischemic optic neuropathy (NAION), which can cause sudden and permanent vision loss. Saxenda, like other GLP-1 receptor agonists, may increase the risk of NAION by potentially reducing blood flow to the optic nerve, though the overall risk is considered low.
How common are Saxenda Vision Problems?
Eye problems are considered rare with Saxenda use, but recent studies have linked it to an increased risk of a serious condition called NAION which can cause sudden and permanent vision loss. Other potential Saxenda vision side effects include short-term vision changes, and for those with diabetes, a possible increased risk of developing or worsening diabetic eye conditions like macular degeneration or retinopathy.
Are there ongoing lawsuits regarding Saxenda and vison loss? What are they about?
Yes, several lawsuits have been filed alleging that Saxenda caused serious eye problems—including partial or total blindness—in some users. These Saxenda Vision Loss Lawuit, generally claim that Novo Nordisk (the manufacturer) failed to adequately warn patients and healthcare providers about potential risks to eye health.
Plaintiffs often seek compensation for medical expenses, lost income due to disability, pain and suffering, and sometimes punitive damages if negligence can be shown. Central issues in the lawsuits include whether there was sufficient scientific evidence linking Saxenda to these risks at the time of approval and whether warning labels should have included clearer information about potential vision side effects.
What should I do if I experience any Saxenda Vision Side Effects?
If you notice any Saxenda vision side effects—such as blurriness, double vision, seeing spots or flashes, eye pain, or sudden loss of sight—stop using Saxenda immediately and contact your healthcare provider without delay. Vision problems can signal underlying conditions like increased intraocular pressure, retinal issues, or even stroke-like events that require urgent assessment.
Your doctor will likely perform a complete eye examination and may refer you to an ophthalmologist for further evaluation. They will also review your medication history, including your Saxenda vision side effects, and may conduct blood tests to rule out other causes such as uncontrolled blood sugar levels or hypertension.
Do not resume Saxenda until your doctor has determined it is safe to do so. Early intervention greatly improves the chance of recovery if the problem is drug-related.
Can a lawyer help with a Saxenda Vision Loss Lawsuit?
Yes—a lawyer experienced in pharmaceutical litigation can assess your Saxenda Vision Loss Lawsuit and help determine if you are eligible for a Saxenda Vision Loss Lawsuit due to alleged harm from Saxenda. Contact Saxenda Vision Loss Lawyer Timothy L. Miles for a free case evaluation about a Saxenda Vision Loss Lawsuit. (855) 846–6529 or [email protected].
How soon can Saxenda vision side effects appear after starting Saxenda?
Saxenda vision side effects could theoretically occur at any time during treatment but are most likely within the first few weeks if they are related to the medication.

