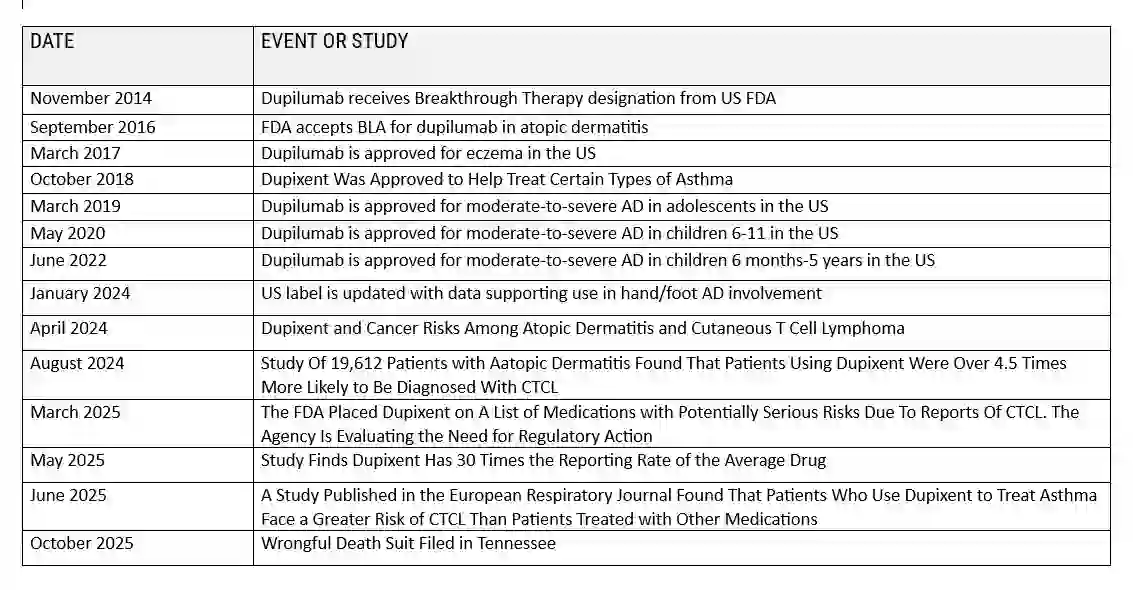
Introduction to Dupixent and Cancer
- Dupixent and Cancer: Recent scientific investigations have raised concerns about a potential link between Dupixent and cancer development, particularly cutaneous T-cell lymphoma (CTCL). Multiple peer-reviewed studies published between 2024 and 2025 have documented cases suggesting an increased cancer risk among certain patient populations receiving dupilumab treatment. These findings have led to regulatory scrutiny and legal action against the drug’s manufacturers, Sanofi and Regeneron.
- Dupixent (dupilumab): Is a significant advancement in biologic therapy, approved for treating multiple inflammatory conditions including atopic dermatitis, eczema, asthma, chronic rhinosinusitis with nasal polyps, and eosinophilic esophagitis. The medication works by targeting specific proteins in the immune system that contribute to chronic inflammation, providing relief for millions of patients worldwide who have struggled with these debilitating conditions.
- Dupixent And Cancer Risks: Understanding the relationship between Dupixent and cancer risks requires careful examination of clinical evidence, regulatory warnings, and ongoing investigations. Patients currently receiving treatment, healthcare professionals prescribing the medication, and individuals considering Dupixent therapy must possess comprehensive knowledge of these emerging safety concerns.
- Comprehensive Guide: This guide provides an analysis of the current evidence linking Dupixent and cancer development, explores the mechanisms behind this potential association, examines regulatory responses, and outlines legal options available to affected individuals. The information presented enables informed decision-making regarding treatment options and potential legal claims.
The Common Dupixent Side Effects
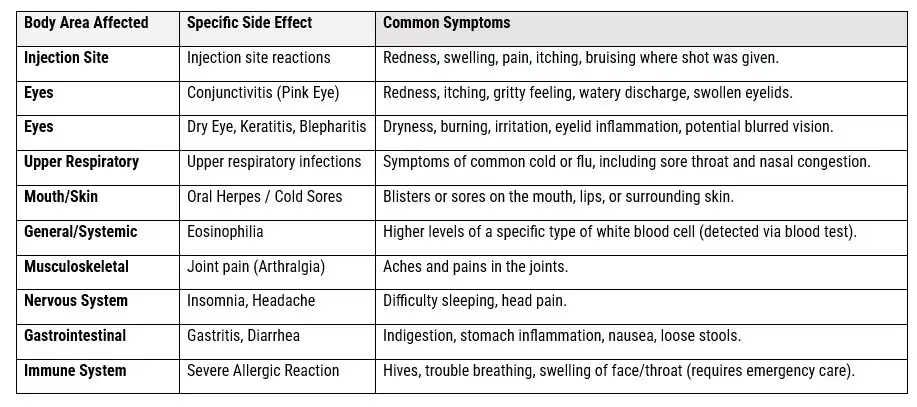
Understanding Dupixent and Its Approved Uses
- Dupixent: Is an important advancement in biologic therapy, containing the active ingredient dupilumab. This monoclonal antibody works by targeting and blocking specific proteins in the immune system, specifically interleukin-4 (IL-4) and interleukin-13 (IL-13). These cytokines are key players in driving type 2 inflammatory responses, which contribute to various chronic conditions characterized by ongoing inflammation and immune system dysfunction.
- Pharmaceutical Collaboration: Between Sanofi and Regeneron Pharmaceuticals brought Dupixent to market, combining Sanofi’s global reach in immunology with Regeneron’s expertise in antibody development. The partnership has resulted in one of the most commercially successful biologic medications in recent pharmaceutical history, with widespread use across multiple therapeutic areas.
FDA Approved Uses
The Food and Drug Administration (FDA) has approved Dupixent for several medical conditions:
- Atopic Dermatitis: Dupixent received initial FDA approval in 2017 for treating moderate-to-severe atopic dermatitis in adult patients whose condition cannot be adequately controlled with topical therapies. Subsequent approvals extended its use to pediatric populations, including adolescents and children as young as six months.
- Asthma: The medication gained approval for moderate-to-severe eosinophilic asthma and oral corticosteroid-dependent asthma in patients aged 12 years and older, providing an alternative treatment pathway for individuals with difficult-to-control respiratory symptoms.
- Chronic Rhinosinusitis with Nasal Polyps: FDA authorization expanded to include adults with inadequately controlled chronic rhinosinusitis accompanied by nasal polyposis, addressing both inflammatory burden and polyp formation.
- Eosinophilic Esophagitis: The most recent FDA approval encompasses treatment of eosinophilic esophagitis in patients aged 12 years and older weighing at least 40 kilograms, marking Dupixent as the first biologic therapy approved for this gastrointestinal condition.
Each approved indication reflects the drug’s ability to modulate type 2 inflammatory pathways across different organ systems and patient populations.
Understanding the Connection Between Dupixent and Cancer Risks
- Dupixent and Cancer: Recent scientific studies have found worrying trends regarding Dupixent and cancer risks, raising concerns within the medical community.
- Dupixent And Cancer Risks: Multiple peer-reviewed studies have suggested a possible connection between Dupixent and cancer, each providing important information to help us understand this potential relationship.
New Findings from Clinical Research
The amount of research looking into Dupixent And Cancer Risks has grown significantly. Here are some key findings:
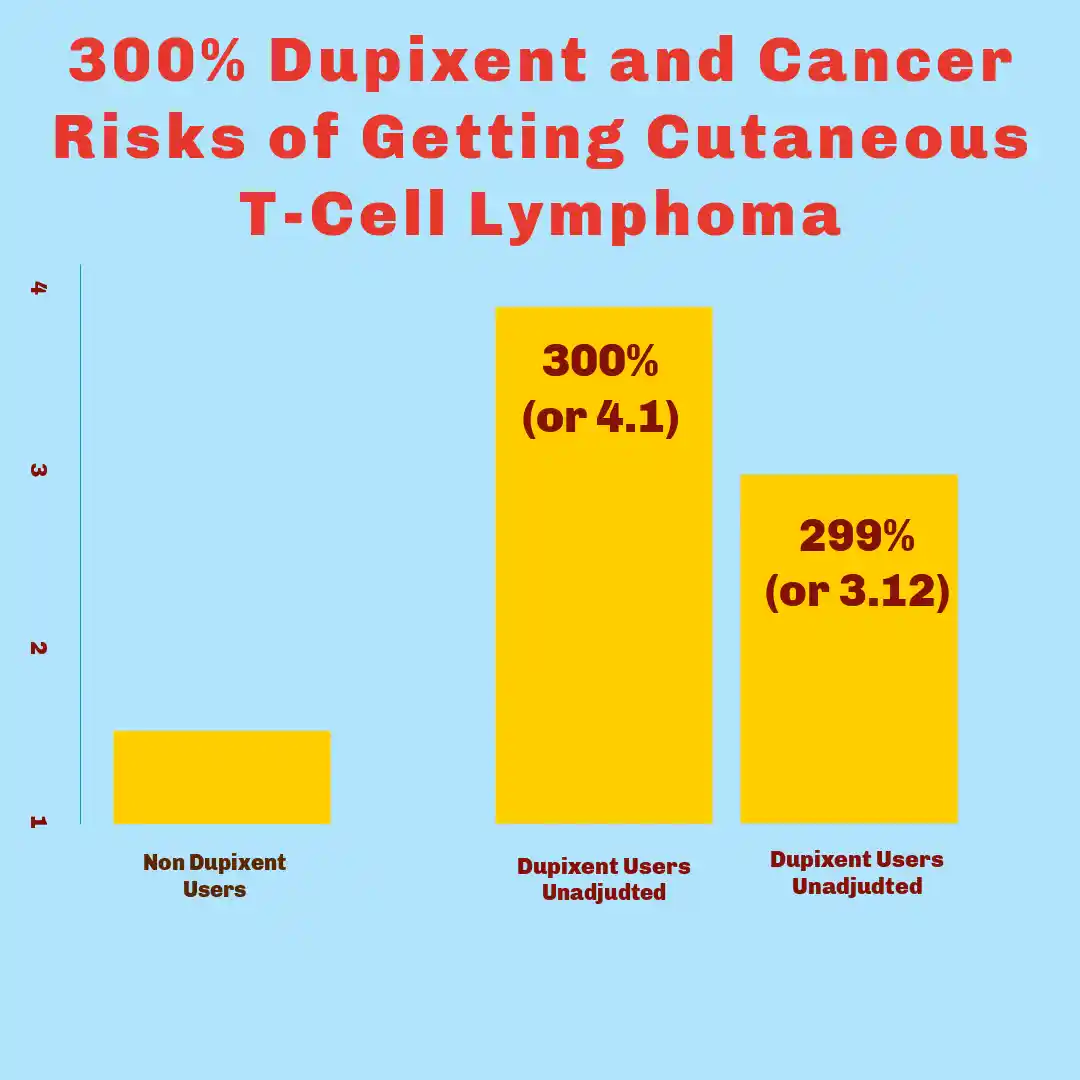
- Hasan et al. (2024): Documented cases where patients receiving dupilumab therapy subsequently developed cutaneous T-cell lymphoma (CTCL), a rare form of non-Hodgkin lymphoma affecting the skin. Their research identified temporal relationships between treatment initiation and lymphoma diagnosis that warranted additional investigation.
- Mandel et al. (2024): Conducted a comprehensive analysis of adverse event databases, revealing elevated incidence rates of CTCL among dupilumab-treated populations compared to control groups. The study examined patient cohorts across multiple dermatological conditions, noting that the cancer risk appeared independent of the underlying disease being treated.
- Sheng-Kai Ma (2025): Contributed additional findings through retrospective analysis of long-term dupilumab users. This research identified specific patient characteristics that appeared to correlate with heightened susceptibility to lymphoproliferative disorders, including duration of treatment and baseline immunological profiles.
How Dupixent Might Affect Cancer Risks
The way Dupixent could potentially impact cancer risks involves intricate immune system processes. Here’s a simplified explanation:
- How It Woks: Dupilumab works by blocking certain signals in the body called interleukin-4 (IL-4) and interleukin-13 (IL-13). These signals are important for regulating how our immune system detects and gets rid of cancer cells.
- Signals Disrupted: When these signals are disrupted, it may make it harder for our body to recognize and eliminate cancer cells in their early stages.
- Effects on Immune System: Additionally, the effects of Dupilumab on the immune system could create an environment that allows certain immune cells called lymphocytes to change in a way that promotes cancer growth.
- Signals Blocked: IL-4 and IL-13 also play a role in maintaining proper functioning of another type of immune cell called T-cells. If these signals are blocked for a long time, it might upset the balance between different types of T-cells and lead to uncontrolled growth of cells characteristic of CTCL.
Regulatory Response and Ongoing Investigations
The FDA’s investigation into Dupixent and cancer risks is an important part of ensuring patient safety. Here’s what we know so far:
- The FDA is reviewing data on reported cases of malignancies among patients treated with dupilumab after its approval.
- They are looking at factors such as when these cancers were diagnosed in relation to when treatment started, how much medication was given, and any other factors that could explain the connection between Dupixent and cancer.
- The agency has also asked manufacturers for more safety information, including follow-up data from clinical trial participants and real-world evidence from patient registries.
- This investigation is ongoing, which means we can expect updates as new information becomes available.
By understanding these potential risks associated with Dupixent, healthcare providers can make more informed decisions when prescribing this medication and monitor patients closely for any signs of malignancy during treatment.
How Dupixent Treatment May Impact CTCL Development or Progression
Clinical Challenges: The way Dupixent works presents significant clinical challenges when evaluating patients for cutaneous T-cell lymphoma (CTCL). Dupilumab, the active ingredient in Dupixent, works by blocking interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling pathways.
Inflammatory Response Patterns: This directly affects the inflammatory response patterns that clinicians rely on to identify early-stage CTCL.
Masking CTCL Symptoms Due to Dupixent Treatment
Masking CTCL Symptoms: The effects of Dupixent on the immune system create a diagnostic dilemma by suppressing the visible signs of CTCL. The medication’s anti-inflammatory properties reduce skin inflammation, redness, and itching—symptoms that are critical for detecting CTCL. Patients receiving Dupixent therapy may temporarily see improvement in their skin symptoms while the underlying malignant T-cell growth goes unnoticed. This suppression of symptoms delays the recognition of malignant transformation, allowing CTCL to progress to more severe stages before an accurate diagnosis is made.
Timing of Treatment: The timing between starting treatment and changes in symptoms adds to the complexity of diagnosis. Healthcare providers may interpret the initial improvement in skin condition as a positive response to Dupixent, unintentionally ignoring the possibility of concurrent CTCL development. The medication’s effectiveness in reducing inflammatory markers hides the abnormal processes typical of early-stage lymphoma, creating a false sense of therapeutic success.
Challenges in Differentiating CTCL from Eczema During Treatment
The clinical presentation of CTCL shares substantial overlap with atopic dermatitis and other inflammatory skin conditions for which Dupixent is prescribed. Both conditions show similar features on the skin:
- Persistent red patches or plaques
- Chronic itching resistant to standard treatments
- Scaling and thickening of affected skin areas
- Distribution patterns affecting similar body parts
The similarity between these conditions requires heightened clinical awareness. A thorough examination by a dermatopathologist becomes necessary when patients show unusual responses to Dupixent therapy or experience worsening skin changes despite following treatment guidelines. The medication’s ability to modify inflammation requires clinicians to be more proactive in performing skin biopsies and analyzing tissue samples.
Accelerated Disease Progression in Diagnosed CTCL Patients
Confirmed CTCL Diagnose: Patients with confirmed CTCL diagnoses who later receive Dupixent treatment face specific risks related to disease acceleration. The change in cytokine profiles caused by blocking IL-4 and IL-13 may disrupt the immune balance needed to control malignant T-cell populations. Clinical observations suggest that administering Dupixent to CTCL patients leads to rapid progression from early-stage disease to advanced forms, such as tumor formation and involvement of other body systems.
Dupixent and Cancer: The connection between Dupixent and cancer progression in diagnosed CTCL cases calls for thorough screening protocols before treatment and ongoing monitoring throughout the duration of therapy.
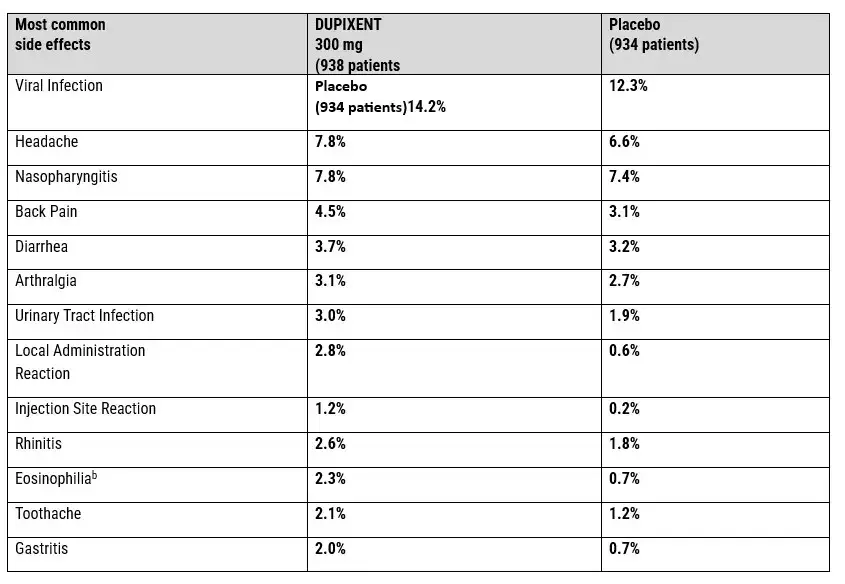
Reported Dupixent Side Effects Related to Cancer Risks
- Safety Profile: The safety profile of Dupixent includes a range of adverse reactions documented through post-marketing surveillance and clinical trial data.
- FAERS: The FDA Adverse Event Reporting System (FAERS) is an important resource for tracking Dupixent side effects that occur during real-world use, providing healthcare professionals and regulatory agencies with crucial information about potential safety concerns.
Common Adverse Reactions and Malignancy Concerns
Dupixent side effects reported in FAERS Adverse Event Reporting System reports show a pattern of adverse events that require careful examination. The database contains numerous reports of malignancies occurring in patients receiving dupilumab therapy, with particular emphasis on blood cancers. These reports include:
- Cases of cutaneous T-cell lymphoma emerging during treatment
- Diagnoses of non-Hodgkin lymphoma in patients with extended exposure to the medication
- Manifestations of mycosis fungoides in individuals previously treated for inflammatory conditions
- Development of Sézary syndrome in patients receiving long-term therapy
The timing between Dupixent and cancer diagnosis varies significantly across reported cases. Some patients developed malignancies within months of starting therapy, while others experienced cancer diagnoses after years of continuous treatment. This variability makes it difficult to establish definitive cause-and-effect relationships but highlights the need for ongoing monitoring of drug safety.
Dermatologic Manifestations Preceding Malignancy
- Specific Skin-Related Adverse Events: Documented in FAERS reports deserve special attention because they may be linked to underlying malignancy. Patients have reported persistent red patches, plaques that do not respond to standard dermatologic treatments, and unusual skin lesions that were later diagnosed as cutaneous lymphoma.
- The Challenge: Lies in differentiating these malignant presentations from the inflammatory skin conditions for which Dupixent was originally prescribed.

Immunologic Alterations and Cancer Risk
Immune System: The way Dupixent affects immune function raises theoretical concerns about its impact on the body’s ability to detect and eliminate cancer cells. Reports submitted to FAERS describe changes in lymphocyte populations, abnormal patterns of immune cell activation, and shifts in cytokine profiles that could potentially influence cancer surveillance mechanisms. These immunological changes are an important area of study for understanding the relationship between biologic therapy and cancer development.
Dupixent And Cancer Risks: The ongoing collection of adverse event reports continues to inform regulatory assessments and clinical decision-making processes. When prescribing Dupixent for approved indications, healthcare providers must carefully consider both the therapeutic benefits and the emerging Dupixent and cancer risks.
Legal Actions Surrounding Dupixent and Its Alleged Cancer Risks
Dupixent Cancer Lawsuit: Multiple plaintiffs have initiated legal proceedings against Sanofi and Regeneron Pharmaceuticals, asserting that the manufacturers failed to provide adequate warnings regarding the potential association between Dupixent and cutaneous T-cell lymphoma development. These ongoing lawsuits against Sanofi and Regeneron regarding failure to warn about potential cancer risks associated with their drug represent a significant development in pharmaceutical litigation, particularly concerning biologic medications marketed for chronic inflammatory conditions.
Failure to Warn: The central allegation in these cases focuses on the manufacturers’ purported knowledge of potential cancer risks prior to widespread public disclosure. Plaintiffs contend that Sanofi and Regeneron possessed information from clinical trials, post-marketing surveillance data, and adverse event reports that suggested a possible connection between dupilumab administration and CTCL development. The lawsuits assert that despite this knowledge, the companies failed to adequately communicate these risks through product labeling, prescribing information, or direct warnings to healthcare providers and patients.
Core Legal Claims
The litigation encompasses several distinct legal theories:
- Failure to Warn: Allegations that manufacturers did not provide sufficient information about cancer risks to enable informed consent
- Design Defect: Claims that the drug’s formulation presents unreasonable dangers that outweigh its therapeutic benefits for certain patient populations
- Negligence: Assertions that the companies breached their duty of care in monitoring, reporting, and communicating safety signals
- Fraudulent Misrepresentation: Accusations that marketing materials and safety communications minimized or omitted material information about Dupixent and cancer risks
Scope of Current Litigation
Dupixent Cancer Lawsuit: The legal actions include both individual lawsuits filed in various state and federal courts, as well as potential consolidation efforts for cases sharing common factual and legal questions. Plaintiffs typically include individuals who developed CTCL or other lymphomas following Dupixent treatment, with documented medical records establishing both the drug exposure and subsequent cancer diagnosis.
Patient Safety: The manufacturers face allegations that their pre-market clinical trials and post-approval safety monitoring systems identified signals warranting enhanced warnings, yet the companies allegedly prioritized commercial interests over patient safety. Discovery proceedings in these cases focus on internal company communications, safety committee deliberations, regulatory submissions, and the timing of when specific cancer risk information became known to corporate decision-makers.
Qualifying for a Dupixent Cancer Lawsuit: Criteria and Requirements
- Eligible for a Dupixent Cancer Lawsuit: To pursue a claim related to Dupixent’s alleged association with cancer development, specific criteria must be met. These requirements are essential in establishing a direct connection between the medication and subsequent cancer diagnosis.
- Dupixent Lawsuit: Patients considering joining a Dupixent Cancer Lawsuit must meet certain fundamental prerequisites that prove both causation and damages resulting from the pharmaceutical intervention.
Primary Qualification Criteria
Individuals seeking to qualify for a Dupixent Lawsuit typically must demonstrate the following essential elements:
- Documented Dupixent Usage: Verifiable prescription records confirming treatment with Dupixent (dupilumab) for an FDA-approved condition, including the duration and dosage of therapy administered
- Cancer Diagnosis: Medical confirmation of cutaneous T-cell lymphoma (CTCL) or related malignancy diagnosed during or following Dupixent treatment
- Temporal Relationship: Evidence establishing a reasonable timeframe between the medications administration Dupixent and Cancer development that supports potential causation
- Absence of Pre-existing Malignancy: Documentation demonstrating no prior cancer diagnosis before initiating Dupixent therapy
Medical Documentation Requirements
The importance of having thorough medical documentation supporting both the use of Dupixent and a diagnosis of CTCL or a related malignancy cannot be overstated in establishing a viable claim. Comprehensive medical records serve as the foundational evidence upon which a Dupixent class action lawsuit cases are built and evaluated.
Essential documentation includes:
- Complete prescription history detailing Dupixent treatment initiation dates, dosing schedules, and duration of therapy
- Pathology reports confirming CTCL or lymphoma diagnosis through biopsy results and histological examination
- Oncology records documenting cancer staging, progression, and treatment protocols implemented
- Dermatology or immunology records showing the original condition for which Dupixent was prescribed
- Imaging studies (CT scans, PET scans, MRI) demonstrating disease extent and progression
- Laboratory results including blood work and immunological markers relevant to both the treated condition and subsequent malignancy
Additional Considerations for Eligibility
Statutes of Limitation: Patients must also consider jurisdictional requirements and statutes of limitations that vary by state, as these legal parameters directly impact the viability of pursuing compensation through a Dupixent Cancer Lawsuit.
Through Documentation: The strength of individual claims depends substantially upon the completeness of medical evidence, the clarity of the temporal relationship between drug exposure and cancer development, and the ability to demonstrate that manufacturers failed to provide adequate warnings regarding potential malignancy risks associated with Dupixent therapy.

The Role of a Experienced Dupixent Cancer Lawyer in Navigating a Dupixent Cancer Lawsuit
- Extensive Legal Experience: Pharmaceutical litigation involving Dupixent requires extensive legal expertise due to the complex intersection of medical science, regulatory frameworks, and product liability law.
- A Dupixent Cancer Lawyer: Possesses the technical knowledge and litigation experience necessary to build compelling cases against pharmaceutical manufacturers who may have failed to adequately warn patients and healthcare providers about potential cancer risks.
Comprehensive Case Evaluation and Strategic Planning
- Dupixent Cancer Lawyer: Experience attorneys conduct thorough assessments of potential claims by examining medical records, treatment histories, and diagnostic reports to establish causation between Dupixent use and subsequent cancer development. This evaluation process involves:
-
- Analyzing temporal relationships between medication administration and cancer diagnosis
- Reviewing prescribing information and manufacturer communications
- Consulting with medical experts specializing in dermatology, oncology, and immunology
- Assessing the strength of scientific evidence linking Dupixent to cutaneous T-cell lymphoma
- Experienced Legal Counsel: In pharmaceutical litigation understands the evidentiary standards required to demonstrate that manufacturers knew or should have known about cancer risks associated with their product. These professionals evaluate whether adequate warnings were provided to prescribing physicians and patients, examining promotional materials, clinical trial data, and internal company documents.
Litigation Services and Legal Representation
- Dupixent Cancer Lawyer: How experienced lawyers assist patients in navigating complex pharmaceutical litigation cases involving Dupixent extends beyond initial case evaluation to encompass comprehensive legal services throughout the litigation lifecycle:
-
- Claims preparation and filing: Drafting detailed complaints that articulate legal theories of liability, including failure to warn, design defect, and negligent misrepresentation
- Discovery management: Conducting depositions of corporate representatives, medical experts, and treating physicians while obtaining critical internal documents through formal discovery requests
- Expert witness coordination: Retaining qualified medical professionals and pharmacologists who can testify regarding causation, standard of care, and manufacturer knowledge
- Settlement negotiations: Engaging with defense counsel and corporate representatives to secure fair compensation without the uncertainty of trial
- Trial advocacy: Presenting evidence to judges and juries when settlement negotiations fail to produce adequate results
- Experience: Attorneys experienced in pharmaceutical litigation maintain relationships with medical experts who understand the immunological mechanisms by which biologics like Dupixent may influence cancer development. This network of professional resources strengthens case presentations and enhances credibility during settlement discussions or trial proceedings.
- Complex Litigation: The complexity of establishing pharmaceutical liability necessitates legal counsel with specific experience in mass tort litigation, regulatory compliance issues, and the scientific principles underlying biologic drug development and approval processes.
Compensation Available for Victims in a Successful Dupixent Cancer Lawsuit
- Compensation in a Dupixent Lawsuit: Individuals who successfully establish liability in their legal claims against Sanofi and Regeneron may pursue various types of damages that may be awarded to individuals who prevail in their Dupixent Cancer Lawsuit against Dupixent’s manufacturers based on its alleged link to cancer development.
- Compensation: The scope and magnitude of compensation in a Dupixent cancer lawsuit depend on the specific circumstances of each case, the severity of the diagnosed condition, and the demonstrable impact on the plaintiff’s life.
Economic Damages
- Economic Damages: Represent quantifiable financial losses directly attributable to the alleged harm caused by Dupixent use. These damages encompass:
-
- Medical Expenses: Reimbursement for all healthcare costs associated with the diagnosis, treatment, and management of cutaneous T-cell lymphoma or other malignancies allegedly linked to Dupixent. This category includes hospitalization fees, chemotherapy treatments, radiation therapy, surgical procedures, diagnostic testing, prescription medications, rehabilitation services, and ongoing monitoring costs. Future medical expenses anticipated for continued treatment and care may also be recoverable.
- Lost Wages and Earning Capacity: Compensation for income lost during periods of illness, treatment, and recovery when the plaintiff was unable to work. Cases involving permanent disability or diminished capacity to perform occupational duties may warrant damages for reduced future earning potential. Documentation of employment history, salary records, and expert testimony regarding career trajectory support these claims.
- Out-of-Pocket Expenses: Recovery of miscellaneous costs incurred as a direct consequence of the illness, including transportation to medical appointments, home healthcare services, medical equipment, modifications to living spaces for accessibility, and other necessary expenditures related to managing the condition.
Non-Economic Damages
Non-Economic Damages: Address intangible losses that lack precise monetary valuation but represent significant harm to the plaintiff’s quality of life:
- Pain and Suffering: Compensation for physical pain, discomfort, and emotional distress endured as a result of developing cancer allegedly caused by Dupixent. This encompasses both past suffering and anticipated future pain related to the condition and its treatment.
- Loss of Enjoyment of Life: Damages awarded when the illness substantially diminishes the plaintiff’s ability to participate in activities, hobbies, and experiences previously enjoyed. The diagnosis and treatment of serious conditions such as cutaneous T-cell lymphoma often impose severe limitations on daily functioning and personal fulfillment.
- Emotional Distress: Recognition of psychological trauma, anxiety, depression, and mental anguish resulting from the cancer diagnosis and the knowledge that a prescribed medication may have contributed to the development of a life-threatening condition.
Punitive Damages
- Punitive Damages: Certain jurisdictions permit punitive damages when defendants demonstrate gross negligence, willful misconduct, or deliberate disregard for patient safety.
- Deterrent to Future Conduct: These damages serve to punish egregious behavior and deter similar conduct by pharmaceutical manufacturers in the future.
Current Status and Future Outlook: Ongoing Research on Dupixent’s Safety Profile and Legal Proceedings Related to Its Alleged Cancer Risks
- The Regulatory Landscape: Currently surrounding Dupixent and cancer continues to evolve as scientific evidence accumulates and legal proceedings advance.
- Monitoring FAERS: The FDA maintains active surveillance of adverse event reports through its FAERS database, analyzing patterns that may indicate previously unrecognized safety signals related to cutaneous T-cell lymphoma development in patients receiving dupilumab therapy.
FDA Regulatory Actions and Investigations
- Ongoing Investigation: The agency’s ongoing investigation into the potential association between Dupixent and Cancer represents a critical component of post-market drug safety monitoring. Regulatory authorities examine multiple data sources to assess causality, including:
-
- Spontaneous adverse event reports from healthcare providers and patients
- Post-marketing surveillance data from real-world clinical settings
- Comparative analyses of malignancy rates in treated versus untreated populations
- Temporal relationships between drug exposure and cancer diagnosis
- Transparancy: The FDA’s commitment to transparent communication regarding emerging safety concerns ensures that healthcare professionals and patients receive timely updates as new information becomes available. This regulatory oversight serves as an essential mechanism for identifying risks that may not manifest during pre-approval clinical trials with limited patient populations and shorter observation periods.
Clinical Research Initiatives
- Ongoing Dupixent and Cancer Updates: Latest updates regarding ongoing clinical trials assessing long-term safety profile of dupilumab (the active ingredient in Dupixent) include extended follow-up studies designed to capture late-onset adverse events. These investigations employ rigorous methodologies to evaluate malignancy incidence across diverse patient populations receiving prolonged treatment courses.
- Independent Analysis: Research institutions worldwide conduct independent analyses examining the biological mechanisms through which IL-4 and IL-13 pathway inhibition might influence cancer development or progression. These mechanistic studies provide crucial insights into whether observed associations represent true causal relationships or reflect confounding factors inherent to the underlying disease states being treated.
Dupixent Lawsuit Update
Dupixent Lawsuit Update: The trajectory of litigation against Sanofi and Regeneron depends substantially upon emerging scientific evidence and regulatory determinations. Plaintiff attorneys continue gathering medical records and expert testimony to establish causation between Dupixent exposure and subsequent CTCL diagnoses. The consolidation of individual cases into multidistrict litigation remains a possibility should the volume of claims reach sufficient levels to warrant coordinated pretrial proceedings.
Establishing Causation: Defendants’ legal strategies likely emphasize the complexity of establishing causation in cases involving diseases with multifactorial etiologies. The outcome of bellwether trials, should they proceed, will significantly influence settlement negotiations and the resolution of remaining claims within any coordinated litigation framework.
FREQUENTLY ASKED QUESTIONS THE DUPIXENT LAWSUIT
What are the potential benefits of a Lawsuit?
How long will the Dupixent Cancer Lawsuit take to resolve?
What Is Cutaneous T-cell lymphoma?
-
What it is: A type of non-Hodgkin lymphoma that starts in T-cells in the skin.
-
Common symptoms:
-
-
Itchy, scaly patches or plaques
-
-
-
Redness on the skin
-
-
-
Thicker, raised lesions or tumors
-
-
-
Sometimes, the skin redness can cover a large portion of the body, a condition called erythroderma
-
-
Progression: CTCL is often slow-growing, but some types can be aggressive and spread to other parts of the body, such as the lymph nodes, blood, or organs like the liver or lungs.
-
Diagnosis: Diagnosis can be challenging due to similar-looking symptoms and often involves a skin biopsy, physical exam, blood tests, and imaging.
-
Treatment: Treatments are chosen based on the stage of the cancer and can include creams, light therapy, radiation, or systemic treatments like chemotherapy or other medications.
How can I join the Dupixent Lawsuit?
Is There a Dupixent Class Action Lawsuit?
What is cutaneous T-cell lymphoma (CTCL)?

