Introduction to the Dupixent Lawsuit Update: A Complete Timeline of Events
- Dupixent Lawsuit Update: Dupixent and cancer lawsuits are in the early stages, with individual lawsuits being filed across the United States. There is currently no Dupixent class action lawsuit or multidistrict litigation (MDL) for Dupixent cases, as the damages are considered too severe and unique to each patient for a single class action. However, consolidation into an MDL is possible as more cases are filed.
- Dupixent (dupilumab): Is an injectable biologic medication that is used to treat particular types of inflammatory diseases. The medicine is currently the subject of a growing number of lawsuits alleging it causes or accelerates the development of cutaneous T-cell lymphoma (CTCL), a rare form of cancer. As of November 2025, the litigation is in early stages, with no settlements or jury verdicts yet.
TIMELINE OF EVENTS
November 2014: Dupilumab Receives Breakthrough Therapy Designation from the FDA
- The Preceding to the Dupixent Cancer Lawsuit: In November 2014, Dupilumab receives Breakthrough Therapy designation from U.S. Food and Drug Administration (FDA) for the treatment of adults with moderate-to-severe atopic dermatitis (AD) who are not adequately controlled with topical prescription therapy and/or for whom these treatments are not appropriate. Breakthrough Therapy designation was created by the FDA to expedite the development and review of drugs that target serious or life-threatening conditions.
- No Dupixent and Cancer Risks: A Breakthrough Therapy drug must show preliminary clinical evidence of a substantial improvement on a clinically significant endpoint over available therapies, or over placebo if there is no available therapy. The designation includes all of the Fast Track program features, as well as more intensive FDA guidance and discussion. The Breakthrough Therapy designation is distinct from both accelerated approval and priority review, which can also be granted to the same drug if relevant criteria are met. No prior studies had examined the link between Dupixent and cancer.
September: 2016: FDA Accepts Biologics License Application for Dupilumab in Atopic Dermatitis
- BLA Priority Review Granted: In September 2016, the FDA accepted a Biologics License Application (BLA) for a priority review of dupilumab for the treatment of moderate-to-severe atopic dermatitis in adults. The application, submitted by Sanofi and Regeneron, included data from three Phase III clinical trials and had a target action date of March 29, 2017.
- Clinical Research Results: Data from 3 phase 3 studies involving over 2,500 patients globally. The studies assessed dupilumab as monotherapy and in combination with topical corticosteroids for patients whose disease was not adequately controlled by existing therapies.
- Dupixent and Cancer Risks: At this time there had been no studies, patient reports or any association between Dupixent and Cancer.
FDA APPROVAL TIMELINE FOR DUPIXENT FOR THE TREATMENT OF ATOPIC DERMATITIS

March 2017: Dupilumab Is Approved for Eczema in the US
- Dupilumab Approved for Eczema in the US: The FDA first approved Dupixent for adults with moderate-to-severe eczema or atopic dermatitis that did not respond well to topical therapies.
- Safety and Efficacy of Dupilumab: Were confirmed via 3 placebo-controlled clinical trials with a total of 2,119 adult participants with moderate-to-severe atopic dermatitis that was not sufficiently controlled by topical applications. Overall, participants who received dupilumab attained a greater response, of clear or almost clear skin, and experienced a reduction in itch after 16 weeks of treatment. At this times there were still no reports on concerns about Dupixent and Cancer.
October 2018: Dupixent Was Approved to Help Treat Certain Types of Asthma
- Add-On Maintenance Treatment: In October 2018, the FDA approved Dupixent (dupilumab) as an add-on maintenance treatment for adults and adolescents with moderate-to-severe asthma that is either eosinophilic or oral corticosteroid-dependent..
- Clinical Trials: The approval was based on clinical trials showing it can improve lung function and reduce exacerbations, and the drug works by inhibiting the signaling proteins interleukin-4 (IL-4) and interleukin-13 (IL-13) that are involved in the inflammation that causes asthma.
-
- Approval date: The FDA approved Dupixent for asthma on October 19, 2018.
-
- Target patient group: The approval was for patients aged 12 and older with moderate-to-severe asthma who have an eosinophilic phenotype or rely on oral corticosteroids.
-
- Mechanism of action: Dupixent is a monoclonal antibody that blocks IL-4 and IL-13, which are key drivers of Type 2 inflammation in asthma.
-
- Clinical results: Clinical trials showed Dupixent improved lung function and reduced the frequency of asthma attacks.
-
- Administration: The drug is administered via subcutaneous injection, initially by a healthcare professional, but can be self-administered at home after proper training.
-
- Not for acute attacks: It is important to note that Dupixent is not for treating sudden breathing problems.
-
- Dupixent and Cancer: The clinical trials did not report any link between Dupixent and cancer and again at this early stages no concerns were raised about Dupixent and cancer risks.
March 2019: Dupilumab Is Approved for Moderate-To-Severe AD In Adolescents in the US
- Moderate-To-Severe Atopic Dermatitis: In March 2019, the FDA approved dupilumab (Dupixent) for adolescents aged 12 to 17 with moderate-to-severe atopic dermatitis (AD) that is not adequately controlled by topical treatments. This approval provided a new biologic treatment option for this age group, which can be used alone or with topical corticosteroids or topical calcineurin inhibitors.
- Targeted patient group: Adolescents aged 12 to 17 with moderate-to-severe atopic dermatitis.
- Significance: This marked the first time a biologic treatment was approved for this specific adolescent population with moderate-to-severe AD, offering a new option for controlling chronic symptoms like itch and rash.
- Dupixent and Cancer Risks: There was again no patients from clinical trials showing signs or symptoms of Dupixent and cancer and no concerns were yet raised about Dupixent and cancer risks.
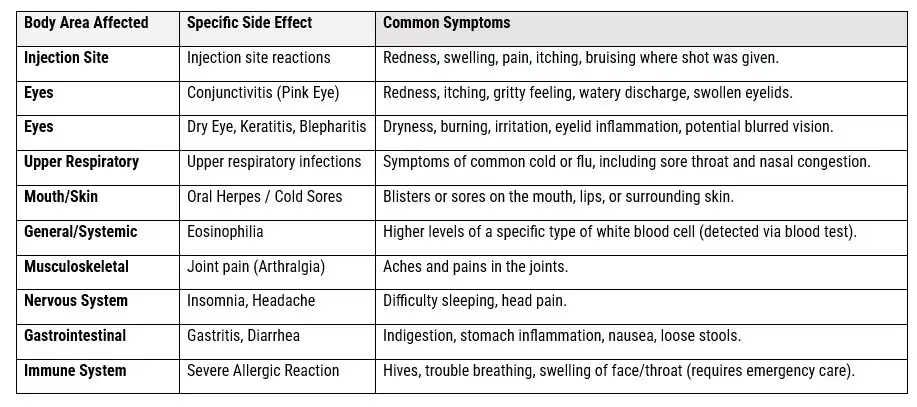
SAFETY AND EFFICACY DATA OF DUPIXENT SIDE EFFECTS FROM CLINICAL TRIALS
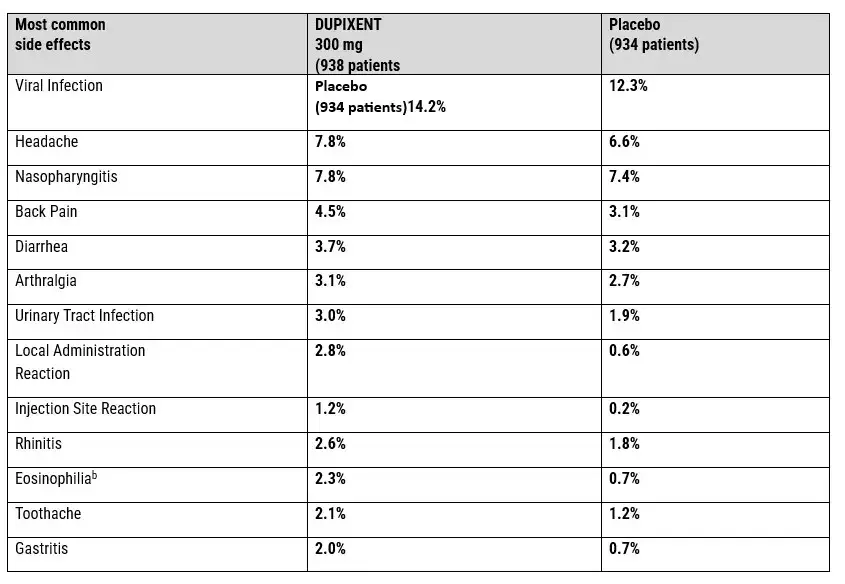
May 2020: Dupilumab is Approved for Moderate-To-Severe AD In Children 6-11 In The US
- Children Aged 6 To 11 Years: In May 2020, the FDA approved the use of Dupixent (dupilumab) for treating moderate-to-severe atopic dermatitis (AD) in children aged 6 to 11 years.
- Approval Basis: The FDA approval was based on data that included pivotal phase 3 results on the efficacy and safety of dupilumab combined with topical corticosteroids (TCS) compared to TCS alone in children with severe AD. In the trial, children treated with dupilumab and TCS experienced significant improvements in overall disease severity, skin clearance, and itch. There were no concerns about Dupixent and cancer risks.
June 2022: Dupilumab Is Approved for Moderate-To-Severe AD In Children 6 Months-5 Years in the US
- First Biologic Medicine: In June 2022, the FDA approved dupilumab as the first biologic medicine for children aged 6 months to 5 years with moderate-to-severe AD. Dupilumab became the sole biologic medicine authorized for treating moderate-to-severe AD from infancy to adulthood.
- Pivotal Phase 3 Results: Children treated with dupilumab alongside TCS attained clearer skin and experienced significantly reduced itch compared to those treated with TCS alone in a phase 3 trial conducted over 16 weeks. Long-term safety data from a 52-week open-label extension trial in this age group reinforced the well-established safety profile of dupilumab observed across all other approved age groups. There were reports of Dupixent and cancer and still no concerns about Dupixent and cancer risks.
January 2024: US Label Is Updated with Data Supporting Use in Hand/Foot AD Involvement
- FDA Updated the Label for Dupilumab in AD: The FDA updated the label for dupilumab in AD, incorporating efficacy and safety data for patients aged 12 years and older with AD with uncontrolled moderate-to-severe hand and/or foot involvement.
- Phase 3 LIBERTY-AD-HAFT Trial: Data from the Phase 3 LIBERTY-AD-HAFT trial showed that more than twice as many patients treated with dupilumab achieved clear or almost clear skin on hands and feet, and nearly 4 times as many experienced improvement in itch compared to those on placebo. At 16 weeks, significant improvements were observed in both clear skin and reduction in itch for patients treated with dupilumab.
- Dupixent and Cancer Risks: There were reports of Dupixent and cancer and still no concerns about Dupixent and cancer risks.
The Link Between Dupixent and Cancer
April 2024: Dupixent and Cancer Risks Among Atopic Dermatitis and Cutaneous T Cell Lymphoma
-
- Study Reveals Dupixent and Cancer Risks: A study published on April 6, 2024 (Hasan et al., 2024), became the first to establish a link between Dupixent and cancer. The study assessed whether people with atopic dermatitis (AD) who were treated with the drug dupilumab had a higher risk of developing cutaneous T-cell lymphoma (CTCL) compared to those who did not take the medication.
-
- Adjusted Study Shows Higher Risk for Dupixent and Cancer. In their first model, which adjusted for age only, they found that people who took dupilumab had a 300% higher risk of getting CTCL compared to those who didn’t (OR 4.10). Even after they adjusted for more factors—like sex, ethnicity, and race, and removed people who had taken certain other immune-suppressing drugs—the risk of Dupixent and cancer was still more than two times higher (OR 3.20).
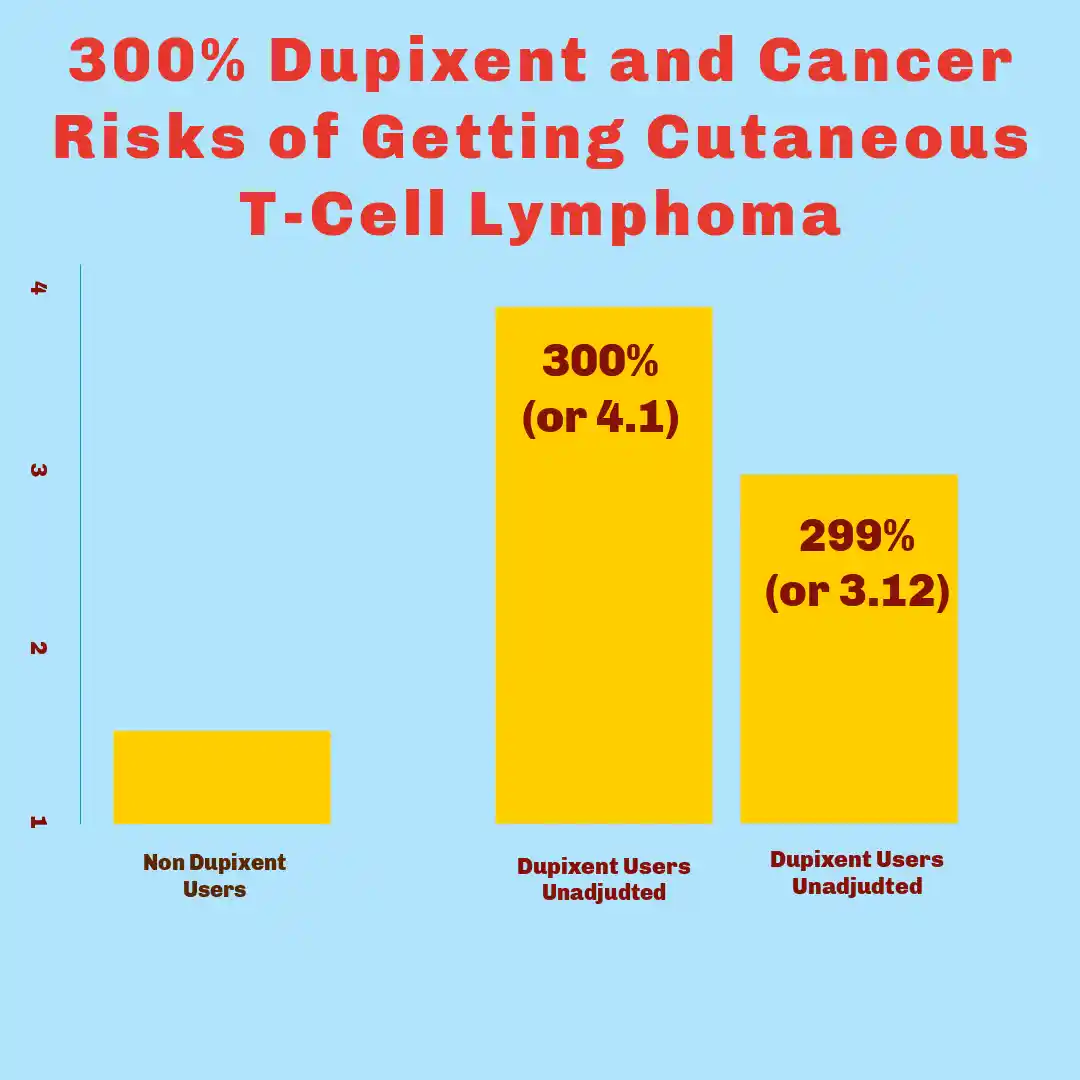
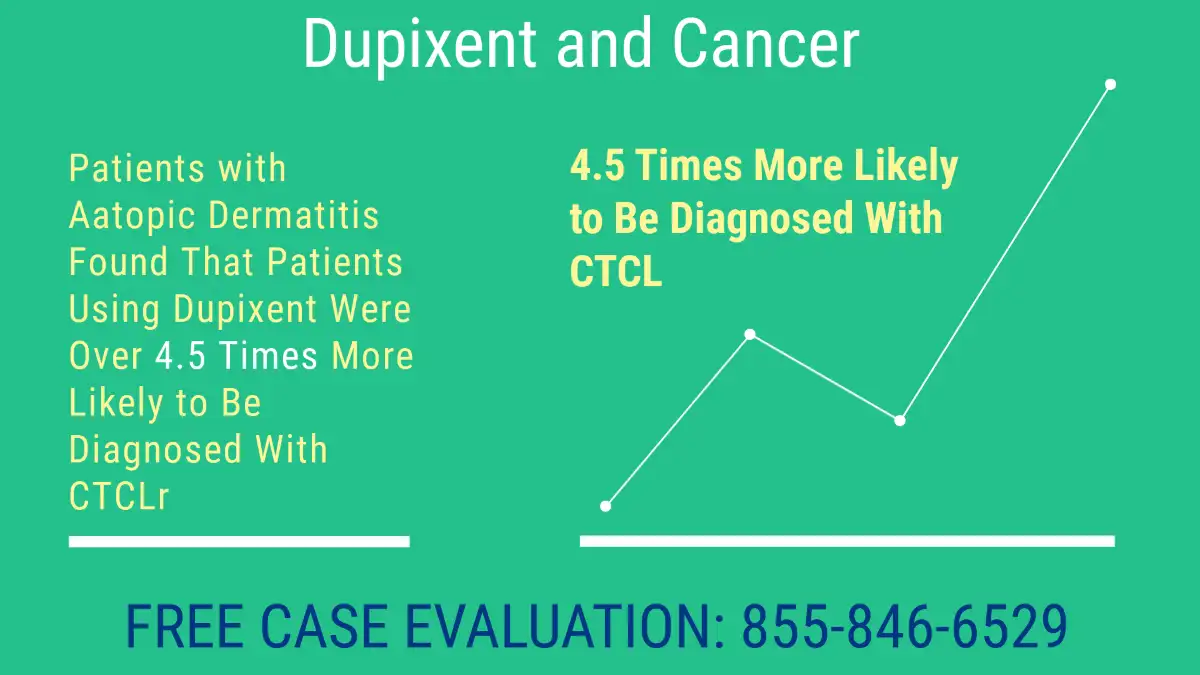
August 2024: Study Of 19,612 Patients with Aatopic Dermatitis Found That Patients Using Dupixent Were Over 4.5 Times More Likely to Be Diagnosed With CTCL
-
- Dupixent and Cancer Risks: A second study using TriNetX data (Mandel et al., 2024) was published in August 2024 and followed a method similar to the first study by Hasan et al. The researchers excluded patients who had other inflammatory diseases or had taken biologic drugs that might be linked to lymphoma.
-
- Dupixent and Cancer:: After matching patients by age, race, and sex, they found that people with atopic dermatitis (AD) who were treated with dupilumab had a 350% higher risk of developing D U Uupixent and Cancer, specifically cutaneous T-cell lymphoma (CTCL) compared to those who were noy treated with the drug (RR 4.59).
March 2025: The FDA Placed Dupixent on A List of Medications with Potentially Serious Risks Due To Reports Of CTCL. The Agency Is Evaluating the Need for Regulatory Action
- Dupixent and Cancer: In March 2025, the FD) placed Dupixent (dupilumab) on its quarterly “Potential Signals of Serious Risks/New Safety Information” list due to reports of a link link between Dupixent and cancer, specifically cutaneous T-cell lymphoma (CTCL).
- Regulatory Action Over Dupixent and Cancer Risks: The agency is actively evaluating the need for regulatory action, which could include mandating stronger warnings or label changes.
Key Information
- Safety Signal: The FDA identified a safety signal after analyzing data from its Adverse Event Reporting System (FAERS), which included more than 300 reports of lymphoma diagnoses submitted by patients and healthcare providers as of September 2025.
FAERS DATABASE
- Regulatory Status: Placement on the “Potential Signals” list indicates that the FDA has seen enough reports to warrant a formal safety review, but it is not a final conclusion that the drug caused the events. As of late 2025, no label changes or recalls had been issued, though the investigation is ongoing.
- Clinical Concerns: The primary concern is that Dupixent may either increase the risk of developing CTCL, or “unmask” a pre-existing, undiagnosed lymphoma, potentially delaying proper cancer treatment.
- Current Warnings: The current Dupixent label does not include a warning about CTCL risk.
- Dupixent Cancer Lawsuit: Multiple law firms are investigating or filing Dupixent and Cancer lawsuits against the manufacturers, Sanofi and Regeneron, alleging they failed to adequately warn patients and doctors about the potential risk.
- Who Is Eligible for a Dupixent Cancer Lawsuit? If you who were prescribed Dupixent and were subsequently diagnosed with T-Cell Lymphoma or experienced other severe Dupixent side effects, call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation as you may qualify for a Dupixent Cancer Lawsuit and possibly entitled to substantial compensation. (855) 846-6529 or [email protected].
May 2025: Study Finds Dupixent Has 30 Times the Reporting Rate of the Average Drug
- May 2025 FDA Analysis on Dupixent and Cancer: An analysis of FDA adverse-event data found 181,575 reports involving Dupixent. The rate of reports involving Dupixent and Cancer (cutaneous T-cell lymphoma} was about 30 times higher for Dupixent users compared to all other medications.
- Source: The finding was part of an analysis of data from the FDA Adverse Event Reporting System (FAERS).
- Context: Adverse event reporting systems rely on voluntary self-reports of Dupixent and Cancer or other servere Dupixent side effects, which can be influenced by media coverage and increased awareness, and do not definitively prove that the drug caused the events. However, the signal was significant enough to prompt attention.
June 2025: A Study Published in the European Respiratory Journal Found That Patients Who Use Dupixent to Treat Asthma Face a Greater Risk of CTCL Than Patients Treated With Other Medications
- Dupixent and Cancer: A 2025 study in the European Respiratory Journal found that asthma patients using Dupixent (dupilumab) had a significantly higher risk of lymphoma, especially T-cell and NK-cell lymphomas, compared to those using other asthma medications.
- Study Overview: Dupilumab, an interleukin (IL)-4α receptor monoclonal antibody, was at first approved as a treatment for atopic dermatitis and later used to treat asthma, where it has been displayed improved lung function, reduce reliance on oral corticosteroids, and decrease severe exacerbations. However, recent studies have shown an increased risk of lymphoma, particularly cutaneous T-cell lymphoma (CTCL), in patients with atopic dermatitis receiving dupilumab.
- Dupixent and Cancer Risks: Overall, this study found that patients treated with dupilumab were at an increased risk of lymphoma, especially T-cell and natural killer-cell lymphomas.

October 2025: Wrongful Dealth
- Dupixent and Cancer: In October 2025: Dupixent wrongful death lawsuit was filed in Tennessee by Chandra Richardson. Her mother, who began using Dupixent in June 2024 and was later diagnosed with T-cell lymphoma, passed away in October 2024.
- Allegations: According to the complaint, Cynthia Marie Hyde was treated for adult-onset atopic dermatitis in 2019, and was prescribed Dupixent in May 2024. She received injections of Dupixent in June and July 2024. After the injections, her skin condition reportedly worsened, with raised scars and widespread darkened, swollen patches.
- Dupixent and Cancer Diagnosis: In September 2024, a biopsy indicated Dupixent and Cancer, specifically peripheral T-cell lymphoma (PTCL), and she died on October 28, 2024. The lawsuit contends Dupixent triggered or accelerated an underlying T-cell lymphoma process that led to her rapid decline.
Current Status of Dupixent and Cancer Lawsuits
- Individual Lawsuits: Dupixent and cancer lawsuits are in the early stages, with individual lawsuits being filed across the United States.
- No Dupixent Class Action Lawsuit or MDL (Yet): There is currently no class action lawsuit or multidistrict litigation (MDL) for Dupixent cases, as the damages are considered too severe and unique to each patient for a single class action. However, consolidation into an MDL is possible as more cases are filed.
- No Recalls or Settlements: As of November 2025, the FDA has not recalled Dupixent, and no settlements or jury verdicts have been reached.
FREQUENTLY ASKED QUESTIONS THE DUPIXENT LAWSUIT
What are the potential benefits of a Dupixent Cancer Lawsuit?
If you are eligible for a Dupixent Lawsuit, you may be entitled to substantial compensation for your medical treatment and for your pain and suffering.
How long will the Dupixent Cancer Lawsuit take to resolve?
The duration of lawsuits can vary significantly, depending on the complexity of the case, legal strategies, and whether settlements are reached. It could take several months to years to resolve the Dupixent Cancer Lawsuit.
What Is Cutaneous T-cell lymphoma?
Cutaneous T-cell lymphoma (CTCL) is a rare, slow-growing cancer of the T-cells (a type of white blood cell) that affects the skin. Symptoms often include itchy, scaly rashes, red patches, plaques, or tumors, and can mimic other skin conditions like eczema, which can delay diagnosis. In some cases, the cancer can spread from the skin to the lymph nodes or other organs. phagitis.
Key characteristics
-
What it is: A type of non-Hodgkin lymphoma that starts in T-cells in the skin.
-
Common symptoms:
-
-
Itchy, scaly patches or plaques
-
-
-
Redness on the skin
-
-
-
Thicker, raised lesions or tumors
-
-
-
Sometimes, the skin redness can cover a large portion of the body, a condition called erythroderma
-
-
Progression: CTCL is often slow-growing, but some types can be aggressive and spread to other parts of the body, such as the lymph nodes, blood, or organs like the liver or lungs.
Common types: The two most common types are mycosis fungoides and Sézary syndrome.
-
Diagnosis: Diagnosis can be challenging due to similar-looking symptoms and often involves a skin biopsy, physical exam, blood tests, and imaging.
-
Treatment: Treatments are chosen based on the stage of the cancer and can include creams, light therapy, radiation, or systemic treatments like chemotherapy or other medications.
How can I join the Dupixent Lawsuit?
Contact Dupixent Lawyer Timothy L. Miles: Today who can determine of you qualify for a Dupixent Cancer Lawsuit and can further explain the process. 855/846-6529 or via e-mail at [email protected]. (24/7/365).
Is There a Dupixent Class Action Lawsuit?
As of now, there is no Dupixent class action lawsuit, with almost all Dupixent lawsuits being filed product liability cases on their behalf only, rather than a single class action. In an MDL, the plaintiff maintains his own case and potential compensation, but legal proceedings share a concerted strategy for judicial efficiency. Furthermore, it gives patients across the country to present consistent evidence about Dupixent and cancer risks while keeping their individual right to compensation intact.
What is cutaneous T-cell lymphoma (CTCL)?
Cutaneous T-cell lymphomas (CTCL) are a wide range and group of extremely rare cancers. Some forms grow slowly and stay in the skin. Less frequently, the cancer can spread from the skin to the blood or a lymph node. Dermatologists often work closely with oncologists (doctors who treat cancer) and other specialists to care for patients who have one of these cancers.
How many different types of CTCL are there?
There are several types. The most common types of cutaneous T-cell lymphoma, also known as CTCL cancer or CTCL, are:
-
Mycosis Fungoide (MF) – The most common type, characterized by patches, plaques, and tumors on the skin.
-
Sézary Syndrome (SS) – A more aggressive form of MF that affects the blood as well as the skin.
Other more rare types of CTCL include:
-
Lymphomatoid papulosis – A benign form of CTCL that causes small, itchy bumps on the skin.
-
Granulomatous slack skin – A rare, indolent form of CTCL that causes loose, pendulous folds of skin.
-
Anaplastic large cell lymphoma – An aggressive form of CTCL that can spread to other organs.
-
Extranodal NK/T-cell lymphoma: A rare type of CTCL that affects the skin and lymph nodes.
-
Extranodal NK/T-cell lymphoma, nasal type – A rare, aggressive lymphoma that can affect the skin, among other places.
-
Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) – A rare, slow-growing type that causes nodules deep in the fatty layer of the skin, most often on the legs.
-
Primary cutaneous peripheral T-cell lymphoma (unspecified) – This is a category for rare entities that do not fit into other classifications.