Introduction to the Mounjaro Eye Problems
- Mounjaro Eye Problems: Refers to recent developments which have brought significant attention to potential ocular convolutions associated with Mounjaro use. Reports of serious Mounjaro vision side effects, including vision loss and blindness, have engendered legal action against the manufacturer. These concerns have crystallized into the Mounjaro Eye Side Effects Lawsuit, with patients seeking representation from legal professionals, including Mounjaro Blindness Lawyers, to address alleged inadequate warnings regarding vision-related risks.
- Mounjaro (tirzepatide): Is a significant advancement in prescription treatments for type 2 diabetes. It offers patients improved blood sugar control through its innovative mechanism as a dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptor agonist. Approved by the U.S. Food and Drug Administration (FDA) in 2022, this medication has gained widespread adoption among healthcare providers seeking effective solutions for managing blood sugar levels in diabetic patients.
- Mounjaro Vision Problems: The benefits of Mounjaro have made it an important option in diabetes treatment. However, new evidence suggests that patients need to be aware of potential Mounjaro vision side effects that may come with using this medication.
MOUNJARO APPROVAL TIIMELINE
|
DATE |
EVENT |
|
May 13, 2022 |
The U.S. Food and Drug Administration (FDA) grants initial approval for Mounjaro (tirzepatide) as a once-weekly therapy to improve blood sugar (glycemic) control in adults with Type 2 diabetes, as an adjunct to diet and exercise. This was the first in a new class of dual GIP and GLP-1 receptor agonist medications. |
|
December 2022 |
Mounjaro is added to the FDA drug shortage list due to an unexpected and significant increase in demand. |
|
November 8, 2023 |
The FDA approves the same active ingredient, tirzepatide, under a new brand name, Zepbound, for chronic weight management in adults with obesity or overweight and at least one weight-related condition. |
|
April 2024 |
Zepbound is also listed as being in shortage due to high demand. |
|
October 2024 |
The FDA removes both Mounjaro and Zepbound from the drug shortage list, determining that the manufacturer, Eli Lilly, can now meet market demand. |
| December 2024 |
The FDA upholds its decision that the tirzepatide shortage is resolved. The FDA also expands Zepbound’s indication, approving it as the first and only prescription medication for adults with moderate-to-severe obstructive sleep apnea and obesity. |
|
May 2025 |
Court rulings uphold the FDA’s decision to end the temporary permission for pharmacies to sell compounded (custom-made) versions of tirzepatide, enforcing that only FDA-approved Mounjaro and Zepbound remain the legitimate ways to access the medication. |
| Present (as of late 2024/early 2025) |
All doses of Mounjaro and Zepbound are available in the U.S. as supply issues have been resolved. |
The Importance of Understanding Mounjaro’s Potential Eye Risks
- Mounjaro Eye Problems: The connection between Mounjaro and eye problems requires careful examination, especially as reports of vision-related issues continue to emerge among users of GLP-1 receptor agonists.
- Mounjaro Vision Side Effects: Patients prescribed this medication must understand that while Mounjaro effectively controls blood sugar levels, the rapid metabolic changes it causes may pose unexpected risks to their eye health. By being aware of these possible side effects, patients can make informed decisions about their treatment options and implement appropriate monitoring strategies.

What This Guide Covers
This comprehensive guide addresses:
- Common and serious eye problems documented in Mounjaro users
- Underlying mechanisms and risk factors contributing to ocular complications
- Evidence-based strategies for vision protection during treatment
- Clinical indicators requiring immediate medical intervention
- Legal developments surrounding Mounjaro vision problems
The Legal Implications of Mounjaro’s Potential Eye Risks
- Mounjaro Vision Side Effects: The seriousness of these concerns goes beyond clinical observations. Ongoing lawsuits against Eli Lilly, the manufacturer of Mounjaro, have brought increased attention to allegations of insufficient warnings about eye-related side effects.
- Mounjaro Eye Problems: These legal actions highlight the significance of potential vision complications and the need for patient awareness. Individuals experiencing Mounjaro eye problems may benefit from consulting a Mounjaro blindness lawyer to understand their legal options and rights.
Empowering Patients Through Education
Mounjaro Vision Side Effects: Proactive education about these risks empowers patients to weigh the metabolic benefits of Mounjaro against potential threats to their vision. This ensures comprehensive care that addresses both blood sugar control and preservation of eye health.
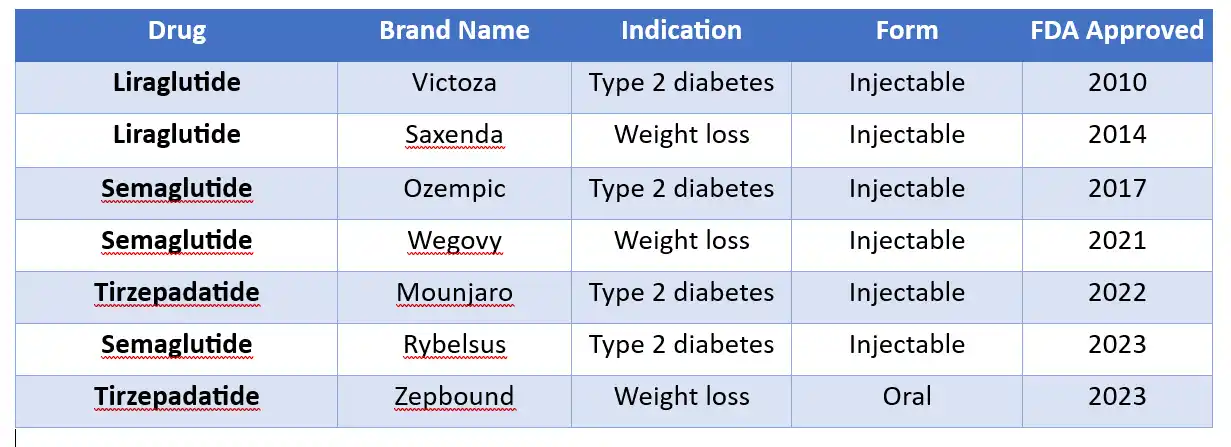
LIRAGLUTIDE VS. SEMAGLUTIDE VS. TIREPADATIDE
How Mounjaro Works in Managing Blood Sugar Levels, and Why It May Affect Your Eyesight
- Mounjaro (tirzepatide): Is a new medication developed by Eli Lilly and Company. It was approved by the FDA in May 2022 for the treatment of type 2 diabetes in adults.
- Mounjaro Eye Problems: What makes Mounjaro different from other medications is that it works in two ways: it stimulates insulin secretion and also activates GLP-1 receptors, which are involved in glucose regulation. This dual action mechanism is why Mounjaro is the first medication approved in this class.
How Mounjaro Works
- Mounjaro Vision Side Effects: Mounjaro works by activating specific receptors in the body that control insulin secretion and glucose metabolism. When taken, tirzepatide binds to GLP-1 receptors throughout the body, leading to several physiological responses that improve blood sugar control:
-
- Increased Insulin Release: Mounjaro stimulates the release of insulin from pancreatic beta cells when blood glucose levels are high.
- Decreased Glucagon Secretion: The medication suppresses the secretion of glucagon, a hormone that promotes the production of glucose by the liver.
- Slower Digestion: Mounjaro slows down the emptying of the stomach, resulting in a slower absorption of nutrients into the bloodstream.
- Reduced Appetite: The medication acts on pathways in the central nervous system to decrease appetite and food intake.

- Mounjaro and Vision Loss: These combined effects lead to significant reductions in hemoglobin A1C levels, which is a measure of long-term blood sugar control. Clinical trials have shown that Mounjaro can achieve A1C reductions ranging from 1.8% to 2.4%, depending on the dosage strength used.
Potential Impact on Eyesight
- Mounjaro Vision Side Effects: The connection between Mounjaro’s positive effects on blood sugar levels and potential Mounjaro and Vision Loss lies in how quickly and significantly glycemic improvement occurs. When blood glucose levels drop rapidly over a short period of time, it can affect delicate structures in the eye, particularly the lens and retina.
- Changes to Water in Eye Lens: The lens relies on glucose metabolism to maintain its transparency and ability to bend light properly. During times when blood sugar levels fluctuate, such as when starting a new medication like Mounjaro, there may be changes in how much water is present in the lens and how its proteins are arranged.
- Diabetic Retinopathy: Patients who already have diabetic retinopathy (an eye condition caused by diabetes) may be more susceptible during this adjustment phase. The sudden breakdown of stored glucose byproducts and restoration of normal blood flow patterns can trigger inflammation within retinal tissues, potentially worsening vision problems even though overall blood sugar control improves.
- Regular Monitoring: It is important for individuals using Mounjaro or any other diabetes medication to regularly monitor their eyesight and communicate any changes with their healthcare provider.
COMPARISON OF GLP-1 DRUG PROFILES
|
Drug |
Brand Name | Indication | Form | FDA Approved |
|
Liraglutide |
Victoza | Type 2 diabetes | Injectable | 2010 |
|
Liraglutide |
Saxenda | Weight loss | Injectable | 2014 |
| Semaglutide | Ozempic | Type 2 diabetes | Injectable |
2017 |
|
Semaglutide |
Wegovy | Weight loss | Injectable | 2021 |
| Tirzepadatide | Mounjaro | Type 2 diabetes | Injectable |
2022 |
|
Semaglutide |
Rybelsus | Type 2 diabetes | Injectable | 2023 |
|
Tirzepadatide |
Zepbound | Weight loss | Oral |
2023 |
Common Eye Problems Linked to Mounjaro Use
- Mounjaro Vision Problems: Patients starting treatment with tirzepatide often experience various eye issues. While these problems are usually not severe, they still need careful attention and documentation.
- Early Stages of Treatment: Both clinical observations and patient reports have shown a pattern of Mounjaro eye problems that occur during the early stages of treatment or after dosage changes.
Dry Eye Syndrome
One of the most common eye complaints among people receiving Mounjaro treatment is dry eye syndrome. This condition shows up through several signs:
- A constant feeling of grittiness or something foreign in the eyes
- Increased sensitivity to things like wind, air conditioning, or looking at screens
- Unusual tearing as the eye tries to make up for lack of moisture
- Discomfort when wearing contact lenses or trouble getting used to them
- Redness and swelling of the surface of the eyes
The exact reason why dry eyes develop in Mounjaro users is still being studied. However, changes in how the body metabolizes substances and inflammation markers throughout the body might play a role in reducing tear film quality and stability.
Blurred Vision
- Blurred Vision: Another commonly reported side effect is blurred vision, which includes:
-
- Occasional difficulty focusing on things at different distances
- Changes in how clear vision is throughout the day, especially when the medication is most active
- Trouble reading small text or doing tasks that require detailed vision
- Temporary distortion of how things look that goes away on its own
- Osmotic Transfigurations: These visual disturbances usually happen during times when blood sugar levels are changing significantly. The eye’s ability to focus is affected by fluctuations in glucose levels in the fluid inside the eye (aqueous humor) and lens. When blood sugar levels rise or fall quickly, it causes osmotic changes in the lens, temporarily altering its shape and focusing ability.
- Keep Track of Changes: Even though these symptoms generally go away on their own, it’s important not to downplay their impact on daily life. Patients have reported difficulties with driving, especially at night, reduced productivity in jobs that require precise vision, and a lower quality of life during times when symptoms are present. By keeping track of when symptoms start, how long they last, and how severe they are, healthcare providers can better differentiate between expected effects of the medication and potentially serious complications that need immediate attention.
Serious Vision Complications Associated with Mounjaro: What You Need to Know
- Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION): Represents the most severe ocular complication associated with Mounjaro use. This condition occurs when blood flow to the optic nerve becomes suddenly restricted, resulting in irreversible damage to the nerve tissue responsible for transmitting visual information from the eye to the brain.
- Mounjaro and Vision Loss: Patients experience sudden, painless vision loss—typically upon waking—that may affect one or both eyes. The vision impairment manifests as a dark area in the visual field, reduced color perception, or complete blindness in the affected eye.

Alarming Data on Mounjaro and Vision Loss Risks
- NAION Incidence: Recent epidemiological data reveals alarming patterns regarding Mounjaro and Vision Loss risks. Studies examining GLP-1 receptor agonist users demonstrate a statistically significant elevation in NAION incidence compared to patients using alternative diabetes medications.
- Severe Mounjaro Eye Problems: Specifically, research published in medical journals indicates that individuals prescribed semaglutide or tirzepatide face approximately four to seven times higher risk of developing NAION than those receiving other glycemic control agents. For patients with pre-existing optic nerve conditions or cardiovascular risk factors, this probability increases substantially.
Diabetic Retinopathy Exacerbation: A Critical Concern
Diabetic Retinopathy Exacerbation: Constitutes another critical concern for patients managing type 2 diabetes with Mounjaro. The rapid reduction in blood glucose levels—while therapeutically beneficial—can paradoxically accelerate retinal damage in individuals with established diabetic eye disease. The mechanism involves sudden changes in retinal blood vessel permeability and fluid dynamics, potentially leading to:
- Increased macular edema
- Proliferative retinopathy progression
- Vitreous hemorrhage
- Retinal detachment
Heightened Vulnerability to Complications
- Mounjaro Vision Side Effects: Clinical observations suggest that patients experiencing aggressive glycemic improvement within the first months of Mounjaro treatment face elevated vulnerability to these complications.
- Heightened Surveillance: The correlation between treatment initiation and Mounjaro Eye Problems necessitate heightened surveillance protocols, particularly for individuals with documented retinal pathology at baseline assessment.

Identifying Symptoms and Seeking Help for Mounjaro-Related Eye Problems
- Self-Monitoring: Recognizing Mounjaro vision side effects, requires careful self-monitoring and awareness of specific visual disturbances that may indicate serious complications.
- Be Vigilant of Warning Signs: Patients prescribed tirzepatide must understand the difference between minor eye effects and potentially sight-threatening conditions requiring urgent intervention.
Critical Warning Signs Requiring Immediate Medical Attention:
- Sudden vision loss in one or both eyes, particularly if painless and occurring without trauma
- Persistent blurred vision that does not resolve within 24-48 hours or progressively worsens
- New visual field defects, including dark spots, blind spots, or peripheral vision loss
- Altitudinal visual field loss, characterized by vision loss in the upper or lower half of the visual field
- Difficulty distinguishing colors or perceiving color saturation changes
- Seeing halos or rings around lights, especially in low-light conditions
- Double vision (diplopia) that persists beyond brief episodes

Additional Symptoms Warranting Professional Evaluation:
- Mounjaro Vision Side Effects: Patients experiencing eye pain accompanied by visual changes, severe headaches localized around the eyes, or sudden onset of floaters and flashes should contact their healthcare provider promptly. Visual disturbances occurring immediately after dose escalation or within the first months of treatment initiation deserve particular attention, as rapid glycemic changes may precipitate retinal complications in susceptible individuals.
- Keep Track of Any Eye Changes: The timing between when symptoms start and when medication is taken provides important diagnostic information. Keeping track of the exact timing, duration, and characteristics of visual symptoms helps eye doctors distinguish between medication-induced effects and unrelated eye problems.
Emergency Response Protocol:
- Mounjaro and Vision Loss: Contact emergency services or go to the nearest emergency department if experiencing sudden, complete vision loss in one eye. This situation may indicate NAION or other acute vascular events requiring immediate ophthalmologic assessment.
- Act Promptly: Delays in evaluation can result in irreversible vision impairment, making prompt recognition and response essential for optimal outcomes.

Protecting Your Vision While Using Mounjaro: Monitoring, Legal Considerations, and Final Thoughts
- Monitoring vision health during Mounjaro treatment: Requires a structured approach to safeguard ocular function while maintaining glycemic control.
- Baseline Examination: Patients initiating tirzepatide therapy should establish baseline eye examinations prior to treatment commencement, particularly individuals with pre-existing diabetic retinopathy, cardiovascular disease, or other risk factors associated with Mounjaro Eye Problems.
Essential Monitoring Protocols
- Baseline Assessment: High-risk patients warrant enhanced surveillance through comprehensive ophthalmologic evaluations at the following intervals:
-
- Baseline assessment: Complete dilated eye examination before initiating Mounjaro
- Three-month follow-up: Early detection of acute changes during initial treatment phase
- Six-month intervals: Ongoing monitoring for patients with stable vision
- Immediate evaluation: Any occurrence of visual disturbances regardless of scheduled appointments
- Annual Eye Examination: Patients without pre-existing ocular conditions should maintain annual comprehensive eye examinations, with frequency adjusted based on individual risk profiles and emerging symptoms.
Legal Landscape and Patient Rights
- Mounjaro Eye Side Effects Lawsuit: The Mounjaro eye side effects lawsuit landscape reflects growing concerns regarding inadequate warnings about vision-threatening complications. Multiple plaintiffs have initiated legal action against Eli Lilly, alleging insufficient disclosure of NAION risks and other serious ocular adverse events. These proceedings underscore the gravity of vision complications associated with GLP-1 receptor agonists and the pharmaceutical industry’s responsibility to provide transparent risk communication.
- Legal Recourse: Patients experiencing significant Mounjaro eye problems while using Mounjaro may possess legal recourse, particularly when adequate informed consent regarding ocular risks was not obtained prior to treatment initiation.
Balancing Therapeutic Benefits Against Ocular Risks
- Benefits Versus Risk Assessment: The decision to continue or discontinue Mounjaro requires careful evaluation of individual circumstances. Patients must weigh substantial glycemic benefits—including HbA1c reduction and weight management—against documented risks of vision complications.
- Comprehensive Understanding: This assessment demands collaborative decision-making between patients, endocrinologists, and ophthalmologists, ensuring comprehensive understanding of both therapeutic advantages and potential ocular consequences.
- Document Everything: Documentation of all visual symptoms, maintenance of regular monitoring schedules, and open communication with healthcare providers constitute essential elements of responsible Mounjaro use.
- Balanced Assessment: Patients should be informed of the potential ocular risks associated with Mounjaro, including the possibility of vision complications. However, it is important not to overlook the significant therapeutic benefits this medication can provide. A balanced assessment of these factors is crucial for making informed treatment decisions.
FREQUENTLY ASKED QUESTIONS ABOUT MOUNJARO EYE PROBLEMS
1. What is the Mounjaro Eye Side Effects Lawsuit
Answer:
The Mounjaro Eye Side Effects Lawsuit refers to legal claims filed by individuals who allege that taking Mounjaro, a medication prescribed for type 2 diabetes, caused them to suffer vision loss or significant eye problems. These lawsuits typically claim that the manufacturer did not adequately warn patients and healthcare providers about the potential risk of vision side effects associated with Mounjaro.
2. What kinds of Mounjaro eye problems have been linked to the medication?
Answer:
Some users of Mounjaro have reported experiencing a range of Mounjaro eye problems, including blurred vision, changes in eyesight, and in rare cases, more serious conditions such as partial or complete vision loss. While these side effects do not affect everyone, they are concerning enough to have prompted legal action and increased medical scrutiny.
3. What are the reported vision side effects of Mounjaro?
Answer:
Reported vision side effects of Mounjaro include blurry vision, difficulty focusing, sudden changes in sight, and in some instances, progressive loss of eyesight. If you notice any changes in your vision while taking this medication, it’s important to contact your healthcare provider immediately.
4. Is there a proven connection between Mounjaro and vision loss?
Answer:
Currently, research is ongoing regarding the link between Mounjaro and vision loss. While some patients have reported experiencing vision problems after starting the medication, definitive scientific evidence directly connecting Mounjaro eye problems, is still being investigated. However, these reports have been significant enough to lead to lawsuits and further clinical evaluation.

![futuristic eye with white waves accross blue eye used in Zepbound Vision Loss Lawsuit Update: An Authoritative Patient Reference Guide [2026]](https://classactionlawyertn.com/wp-content/uploads/2025/11/mounjaro-eye-problems-feature-picture-1024x512.webp)
