Introduction to Zepbound and Blurry Vision
- Zepbound and Blurry Visions: Recent developments have raised substantial concerns regarding the relationship between Zepbound and vision loss effects and have prompted increased scrutiny from medical professionals, regulatory bodies, and affected patients. Reports by patients and clinical research results have raised serious ocular complications, including conditions like Zepbound and Blurry Vision and more serious conditions that may result in permanent visual impairment, have catalyzed legal action against the manufacturer. Understanding these potential Zepbound eye side effects has become essential for current and prospective users of this medication.
- Tirzepatide: Zepbound, a weight-loss medication containing the active ingredient tirzepatide, received FDA approval in November 2023 as a treatment option for individuals struggling with obesity and weight-related health conditions. Manufactured by Eli Lilly and Company, this glucagon-like peptide-1 receptor agonist (GLP-1 RA) has gained significant attention within the medical community and among consumers seeking effective weight management solutions.
- GLP-1 Receptor Agonists: This medication belongs to a class of drugs known as GLP-1 receptor agonists, which also includes widely prescribed medications like semaglutide. What sets tirzepatide apart from other similar medications is its ability to target two receptors in the body: the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. This dual action mechanism makes it different from single-action alternatives and has shown promising results in achieving significant weight loss.
- Blurry Vision: These eye complications can vary in severity, ranging from temporary symptoms like blurred vision to more serious conditions that could potentially lead to permanent visual impairment.
- Extensive Guide: This comprehensive guide aims to provide evidence-based information on the potential effects of Zepbound on eye health. It is designed for patients who are considering or currently using this medication. The document explores the scientific evidence connecting tirzepatide to vision problems, identifies risk factors involved, outlines strategies for recognizing symptoms, and offers practical recommendations for monitoring and safeguarding eyesight during treatment with Zepbound.

The Zepbound Dual Mechanism of Action
- Divergent Mechanism of Action: Zepbound (tirzepatide) is a new type of weight loss and type 2 diabetes medication that works differently from other drugs in this category.
- Dual Methods of Operation: It does this by targeting two specific receptors in the body: the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. This sets it apart from traditional GLP-1 receptor agonists, which only affect one pathway.
How Zepbound Works
- Mechanism of Action: The unique way Zepbound works makes it more effective than other medications. Here’s how it functions:
-
- Increased Insulin Secretion: When blood sugar levels are high, Zepbound stimulates the release of insulin, which helps lower those levels.
- Reduced Glucagon Release: Glucagon is a hormone that raises blood sugar levels. Zepbound inhibits its release, preventing blood sugar from rising too much.
- Slowed Gastric Emptying: Zepbound slows down the rate at which food leaves the stomach, leading to a feeling of fullness for longer periods.
- Decreased Appetite: Through its effects on the central nervous system, Zepbound reduces appetite and cravings.
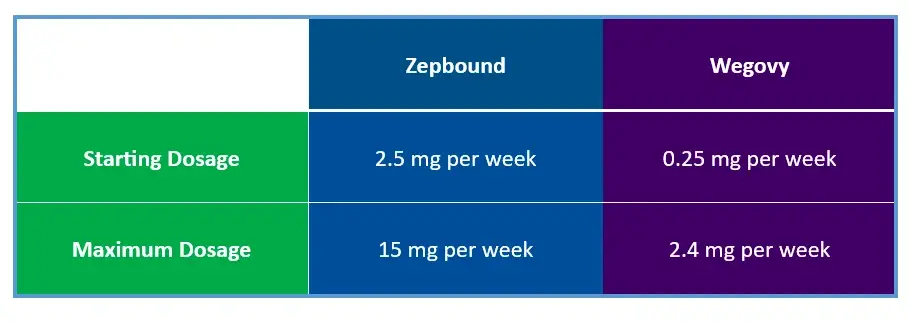
ZEPBOUND VS. WEGOVY DOSAGE COMPARISON
Differences Between Zepbound and Other GLP-1 Receptor Agonists
- GLP-1 Receptor: Other commonly prescribed GLP-1 receptor agonists like Ozempic (semaglutide) and Trulicity (dulaglutide) only target the GLP-1 receptor. While these medications can help with blood sugar control and modest weight loss, they may not be as effective as Zepbound.
- Clinical trials: Have shown that patients taking tirzepatide experience greater weight loss and better control of blood sugar levels compared to those on single-receptor medications.
Dosage and Administration Guidelines
- Administration: Zepbound is administered through injections under the skin (subcutaneous) once a week. The dosage may vary depending on individual needs and how well the patient responds to treatment.
- Guidelines: Here are the specific guidelines for dosage and administration:
-
- Initial Dose: Start with 2.5 mg once weekly for four weeks.
- Maintenance Escalation: If tolerated, increase the dose by 2.5 mg every four weeks until reaching the maximum dose of 15 mg.
- Injection Sites: Rotate between different injection sites such as the abdomen, thigh, or upper arm to prevent any potential side effects like lipodystrophy (abnormal distribution of fat).
- Storage: It is important to note that Zepbound should be stored in a refrigerator before first use but can be kept at room temperature for up to 21 days after opening. To maintain consistent levels of the medication in your body, make sure to administer your injections on the same day each week.
Overview of Vision Loss and Eye Problems Linked to Zepbound
- Zepbound Eye Problems: Reports from patients and healthcare providers have documented various Zepbound eye problems following the administration of tirzepatide.
- Zepbound Eye Side Effects: These eye issues can vary in severity and how they show up, which is why it’s important for both doctors prescribing the medication and patients receiving treatment to pay close attention.
Common Eye Side Effects of Zepbound
- Here are some specific ways that Zepbound eye side effects may show up:
-
- Blurred vision: Patients have reported temporary or persistent difficulty focusing, which may affect one or both eyes
-
- Eye pain: Discomfort ranging from mild irritation to severe ocular pain requiring immediate medical evaluation
-
- Visual field disturbances: Alterations in peripheral or central vision that interfere with daily activities
-
- Photophobia: Increased sensitivity to light sources
-
- Diplopia: Double vision episodes occurring spontaneously during treatment
- The rise in cases of Zepbound and vision loss has caught the attention of the medical community. Doctors have noticed that these negative effects might happen at different points during treatment, starting from the first dose and continuing through maintenance therapy. Some patients experience temporary symptoms that go away with ongoing use, while others face worsening problems that require stopping the medication.
Zepbound and Blurry Vision
- Surge in Blurry Vision Causes Concern: With the surge in use of these medications, researchers and medical experts are paying closer attention to the potential side effects of GLP-1 drugs, due to their effect the eyes. Rapid blood sugar control has been associated with a temporary worsening of diabetic retinopathy, which may lead to vision disturbances, most notably blurry vison.
- Bo Cognizant of Suppen Changes to Eyesight Including Blurry Vision: Watch for any sudden changes, such as blurriness, altered color perception, or loss of peripheral vision. Zepbound may cause your vision to worsen when you start using the medicine or start a higher dose. Tell your healthcare provider if you develop new vision problems, including blurry vision. Previous studies have shown that Zebound or other GLP-1 Drugs causes blurry vision.
Serious Eye Conditions Associated with Zepbound Use
In addition to common vision problems, Zepbound has been linked to several serious eye conditions that require immediate medical attention.
1. Nonarteritic Anterior Ischemic Optic Neuropathy (NAION)
- Zepbound and Vision Loss: Nonarteritic anterior ischemic optic neuropathy (NAION): Is one of the most concerning complications associated with tirzepatide therapy.
- Zepbound Eye Pain: This condition occurs when blood flow to the optic nerve is compromised, leading to sudden and painless vision loss that may be permanent. NAION usually affects one eye initially, but it can also involve both eyes in subsequent episodes.
2. Bilateral Papillitis
- Bilateral papillitis: Is characterized by inflammation of both optic nerve heads and has been reported in patients receiving Zepbound treatment. Symptoms of this condition include reduced visual acuity, visual field defects, and swelling of the optic disc that can be observed during a fundoscopic examination.
- Zepbound Eye Problems: The bilateral presentation distinguishes this condition from unilateral optic neuritis and necessitates a thorough evaluation by an ophthalmologist.
3. Paracentral Acute Middle Maculopathy (PAMM)
- Paracentral acute middle maculopathy (PAMM) is another serious complication associated with tirzepatide use.
- Zepbound Eye Problems: It affects the intermediate and deep capillary plexuses of the retina, resulting in distinctive hyperreflective band-like lesions visible on optical coherence tomography imaging. Patients with PAMM typically experience paracentral scotomas that may persist despite treatment intervention.
4. Diabetic Retinopathy Progression and Macular Edema Development
- Zepbound and Vision Loss: The relationship between Zepbound and vision loss also includes the progression of diabetic retinopathy and the development of macular edema, particularly in patients with pre-existing diabetic eye disease.
- Increased Permeability of Blood Vessels: Rapid control of blood sugar levels achieved through tirzepatide therapy may paradoxically speed up the progression of retinopathy through mechanisms involving changes in retinal blood flow and increased permeability of blood vessels.
Scientific Evidence Linking Zepbound to Vision Issues
- Zepbound Eye Problems: Research published in JAMA Ophthalmology has established a concerning association between GLP-1 receptor agonists, including tirzepatide (Zepbound), and increased risk of vision-threatening complications. A landmark study examined electronic health records from multiple healthcare systems and identified a statistically significant elevation in the incidence of nonarteritic anterior ischemic optic neuropathy among patients prescribed these medications compared to those receiving alternative diabetes or weight management therapies.
- Heightened Risk for Zepbound and Vision Loss: The study documented that patients using GLP-1 receptor agonists demonstrated a two to four-fold increased risk of developing NAION, with the association remaining consistent across different patient populations and geographic locations.
- JAMA Ophthalmology studies have provided critical insights through rigorous methodological approaches:
-
- Retrospective cohort analyses involving tens of thousands of patients revealed temporal relationships between medication initiation and onset of visual symptoms
-
- Case-control studies demonstrated dose-dependent relationships, with higher cumulative exposures correlating with elevated risk profiles
-
- Subgroup analyses identified specific patient characteristics that modified risk magnitudes
- Pharmacovigilance Analysis: Conducted through the FDA Adverse Event Reporting System (FAERS) database has corroborated these findings through real-world evidence. These post-marketing surveillance systems have documented hundreds of adverse event reports describing vision loss, optic nerve disorders, and retinal complications temporally associated with tirzepatide use. The disproportionality analyses—statistical methods used to detect safety signals—have generated elevated reporting odds ratios for several ocular conditions, suggesting a potential causal relationship warranting continued investigation.
- Observational Studies: From international registries have expanded upon these findings by documenting cases across diverse healthcare settings. Data from European pharmacovigilance networks and Asian patient registries have reported similar patterns of ocular adverse events, strengthening the evidence base through geographic and ethnic diversity. These studies have documented median onset times ranging from three to nine months following treatment initiation, though cases have been reported across the entire treatment duration spectrum.
Keeping Track of Symptoms
- Symptom Substantiation: The connection between Zepbound and vision loss makes it important for patients to document their symptoms. Here’s what patients should record:
-
- The specific date when symptoms began
- Whether the symptoms affect one or both eyes
- Any associated symptoms such as headache, nausea, or neurological changes
- Diagnostic Evaluations: This information will be valuable for healthcare providers when conducting diagnostic evaluations and determining appropriate interventions.
Recommendations for Patients Using Zepbound Regarding Eye Health Monitoring Guidelines
- Monitoring Zepbound Eye Problems: Establishing comprehensive patient monitoring guidelines is a critical part of safe Zepbound therapy.
- Detecting Zepbound Eye Problems Early: Patients starting treatment with tirzepatide must have structured surveillance protocols in place to detect eye complications as early as possible.
Baseline Eye Examination
- Eye Examination: Before starting Zepbound therapy, patients should have a thorough eye examination conducted by a qualified eye doctor (ophthalmologist). This evaluation should include:
-
- Visual acuity testing to establish reference measurements for future comparison
- Intraocular pressure measurement to identify pre-existing glaucoma risk
- Dilated fundus examination to assess optic nerve health and retinal integrity
- Optical coherence tomography (OCT) when clinically indicated to document macular and optic nerve structure
- Visual field testing for patients with diabetes or other vascular risk factors
Dose Escalation Protocol
- Increase Dosage Gradually: Following a gradual dose escalation plan may help reduce the risk of sudden metabolic changes that could affect blood flow to the eyes (ocular perfusion). Patients should work closely with their prescribing doctor to follow the manufacturer’s recommended titration schedule, allowing enough time for the body to adjust at each dose level before moving up.
Ongoing Eye Monitoring Requirements
Regular eye check-ups throughout Zepbound treatment enable early detection of vision-threatening complications. The recommended monitoring schedule includes:
- Comprehensive eye examinations every 6-12 months for patients without pre-existing eye disease
- Quarterly assessments for individuals with diabetes, hypertension, or known retinal problems
- Immediate evaluation upon experiencing any visual symptoms, regardless of scheduled appointment timing
Selection of Appropriate Eye Care Expertise
Patients benefit from establishing care with ophthalmologists who have specific expertise in medication-related vision complications. These specialists understand how drug-induced eye problems typically present and stay updated on new evidence regarding GLP-1 receptor agonist-associated vision changes. During the initial consultation, patients should ask about their ophthalmologist’s experience managing patients on similar medications.
The Litigation Landscape
- Zepbound Eye Side Effects Lawsuit: The emergence of Zepbound eye side effects has prompted legal action from affected individuals seeking accountability and compensation. Multiple Zepbound Eye Side Effects lawsuit have been filed in various jurisdictions, alleging that patients developed serious ocular conditions, including nonarteritic anterior ischemic optic neuropathy and other debilitating eye problems, following their use of tirzepatide.
- Failue to Warn: These legal proceedings center on claims that the manufacturer failed to adequately warn patients and healthcare providers about the potential risk of severe vision impairment.
What Affected Patients Should Know About Their Rights
- Proving Causation: Patients who have experienced vision loss or significant eye damage while taking Zepbound possess specific legal rights that warrant careful consideration. The foundation of any successful claim requires establishing a clear causal relationship between the medication and the ocular injury through comprehensive medical documentation. This evidence typically includes:
-
- Detailed medical records documenting the timeline of Zepbound use and symptom onset
- Ophthalmological examinations and diagnostic imaging confirming the nature and extent of eye damage
- Expert medical testimony linking the vision complications to tirzepatide exposure
- Documentation of functional impairment and its impact on quality of life
Importance of Timely Action
The statute of limitations for filing such claims varies by jurisdiction, making prompt consultation with legal counsel essential for preserving one’s rights to seek compensation for medical expenses, lost income, and diminished quality of life resulting from Zepbound-related vision complications.
FREQUENTLY ASKED QUESTIONS ABOUT ZEPBOUND AND VISION LOSS
1. Is there scientific evidence connecting Zepbound and vision loss like NAION?
Yes. Recent studies indicate patients using tirzepatide-containing medications have an increased risk of developing NAION compared to non-users.
2. What are the reported Zepbound eye side effects?
Reported Zepbound eye side effects include blurry vision, dry eye syndrome, eye floaters, diabetic retinopathy progression, eye twitching, ocular inflammation, visual disturbances, allergic reactions around the eyes, and sudden blindness.
3. What should I do if I experience Zepbound eye side effects?
Seek immediate medical attention if you notice any new or sudden changes in your vision while taking Zepbound.
4. What kind of compensation could I receive through a Zepbound Eye Side Effects lawsuit?
Potential compensation includes reimbursement for medical expenses, lost wages, pain and suffering, diminished quality of life, and possibly punitive damages.
Conclusion
Zepbound Vision Side Effects: The emerging evidence surrounding Zepbound Vision Problems necessitates heightened awareness among patients and healthcare professionals. A comprehensive summary of Zepbound vision risks reveals potential associations with nonarteritic anterior ischemic optic neuropathy, diabetic retinopathy exacerbation, and other serious ocular complications that demand immediate attention.
Patient Vigilance Remains Paramount: In mitigating these risks. Individuals prescribed Zepbound must maintain proactive communication with healthcare providers, reporting any visual disturbances—no matter how subtle—without delay. Regular ophthalmologic examinations should constitute a non-negotiable component of treatment protocols, particularly for patients presenting with diabetes or pre-existing vascular conditions.
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com