Results from several large epidemiological studies suggest that exposure to semaglutide in adults with type 2 diabetes is associated with an approximately two-fold increase in the risk of developing NAION compared with people not taking the medicine.
European Medicines Agency Clinical Review, June 6, 2015.
Introduction to a Wegovy and Vision Loss
- Wegovy and Vision Loss: However, recent clinical observations and scientific studies have raised serious concerns about a possible connection between Wegovy and vision loss, specifically Nonarteritic Anterior Ischemic Optic Neuropathy (NAION). NAION is a condition that causes significant damage to the optic nerve and can lead to sudden, permanent vision loss. As a result of this emerging evidence, affected patients are now filing lawsuits against the manufacturers of Wegovy, claiming that they were not adequately warned about these potential vision-related risks.
- Wegovy: Has become a popular medication for long-term weight management and type 2 diabetes treatment. It contains semaglutide, an active ingredient that helps control appetite and regulate blood sugar levels. This medication has gained significant attention from the medical community due to its effectiveness in helping obese patients lose a substantial amount of weight.
- Wegovy Vision Side Effects: It is crucial for patients, healthcare providers, and legal professionals to understand the potential vision side effects of semaglutide as they navigate this evolving optical situation. The relationship between GLP-1 receptor agonists and eye health needs careful investigation, especially since millions of people are using these medications to manage their weight.
- Patient Guide: This article aims to provide a thorough understanding of the documented Wegovy eye side effects, assess the scientific evidence linking the medication to permanent vision impairment, and outline the legal options available through Wegovy eye side effects lawsuits for individuals who have experienced Wegovy and vision loss. The goal is to equip patients with reliable information necessary for making informed decisions about their treatment plans and potential legal actions.
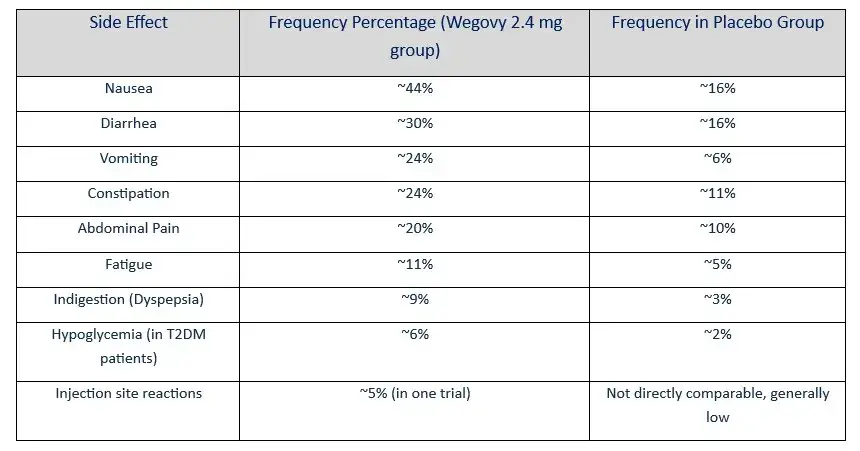
WEGOVY COMMON SIDE EFFECTS PROFILE
Understanding How these Diet Drugs Work
- What is Wegovy?: Wegovy (semaglutide 2.4 mg) is part of a group of medications called GLP-1 receptor agonists. The FDA has specifically approved it for long-term weight management in adults with obesity or overweight conditions, especially those with weight-related health issues. Made by Novo Nordisk, this injectable drug is a higher dose version of the active ingredient found in Ozempic, which is mainly used for managing type 2 diabetes.
- GLP-1 Receptor Agonists: There are other GLP-1-based therapies available besides semaglutide products. Mounjaro and Zepbound, both containing the active ingredient tirzepatide, work as dual GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptor agonists. Mounjaro received FDA approval for treating type 2 diabetes, while Zepbound got authorization specifically for weight management. Although these medications have different regulatory classifications and dosing protocols, they share similar goals in terms of treatment.
How Wegovy Works
- Mechanism of Action: This semaglutide medication works by acting like a hormone in the body called glucagon-like peptide-1 (GLP-1). GLP-1 helps control appetite and food intake.
- Receptor Agonist: When Wegovy is taken, it binds to GLP-1 receptors in the brain, specifically in areas that regulate hunger. This reduces feelings of hunger and increases the sensation of fullness after meals.
Wegovy Approval Patient Population: Who Can use Wegovy?
- FDA Approval: The weight loss drug has been approved for two main groups:
-
- Adults with obesity (body mass index or BMI of 30 kg/m² or greater)
- Adults who are overweight (BMI of 27 kg/m² or greater) and have at least one weight-related condition such as high blood pressure, abnormal cholesterol levels, or type 2 diabetes
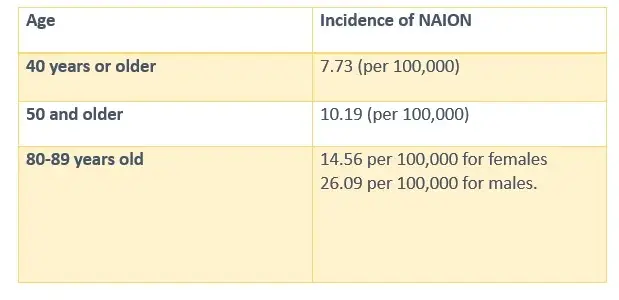
NAION INCIDENCE BY AGE
Wegovy Common Side Effects
- Side Effects of Wegovy: Patients who are prescribed Wegovy can expect to experience some side effects. These side effects are usually mild and include:
-
- Nausea
- Diarrhea
- Vomiting
- Constipation
- Abdominal discomfort
- Locked Stomach: These gastrointestinal issues are the most commonly reported side effects associated with Wegovy.
How GLP-1 Receptor Agonists Work
- GLP-1 Receptor Agonists: work by imitating the natural hormone called glucagon-like peptide-1 (GLP-1). This mechanism involves several processes in the body:
-
- Appetite control: Activation of GLP-1 receptors in the brain reduces feelings of hunger and increases the sensation of fullness
-
- Stomach emptying: Slowing down the emptying of the stomach prolongs the feeling of fullness aftermeals
-
- Insulin release: Enhancing the release of insulin from pancreatic beta cells in response to glucose
-
- Glucagon reduction: Decreasing the secretion of glucagon, which leads to reduced production of glucose by the liver
-
- Continuous Receptor Activation: By continuously activating these receptors through synthetic versions like semaglutide and tirzepatide, significant metabolic effects occur. Clinical trials have shown that patients using Wegovy achieved average weight losses of 15-20% of their initial body weight, which is much higher than what traditional weight loss methods can achieve.
FDA Approvals and Growing Market Demand
- FDA Approval: In June 2021, the FDA approved Wegovy for long-term weight management in adults with a body mass index (BMI) of 30 kg/m² or higher, or 27 kg/m² or higher with at least one weight-related health condition such as high blood pressure, type 2 diabetes, or abnormal lipid levels. In December 2022, this approval was extended to include adolescents aged 12 years and older with obesity.
- Social Media Testimonials: Since their launch, these medications have become extremely popular. Prescriptions for semaglutide-based products have skyrocketed due to impressive weight loss results reported in clinical studies and amplified through social media testimonials. This surge in demand has led to supply shortages throughout 2022 and 2023, prompting manufacturers to increase production capacity.
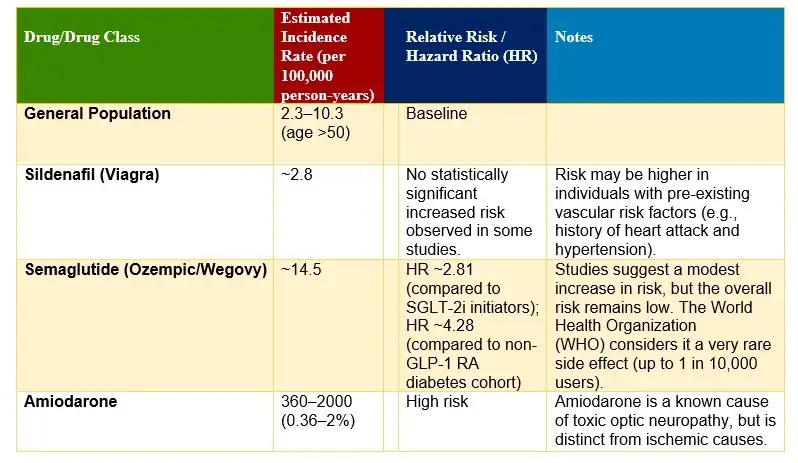
The Link Between Wegovy and NAION: Evidence and Research Findings
- Recent Clinical Investigations: Have identified a concerning association between GLP-1 receptor agonists, including Wegovy (semaglutide), and elevated rates of nonarteritic anterior ischemic optic neuropathy.
- Wegovy Vision Problems: A landmark study published in 2024 examined medical records from a large ophthalmology practice, revealing statistically significant increases in NAION incidence among patients prescribed these medications for type 2 diabetes and obesity management. Another study established a concerning connection between GLP-1 receptor agonists such as Wegovy—and increased incidence rates of NAION. Clinical observations have documented elevated occurrences of this condition among patients prescribed these medications compared to individuals using alternative diabetes treatments.
Epidemiological Evidence of Increased NAION Risk
- NAION Occurrence Rates: Clinical Results: The research documented NAION occurrence rates across distinct patient populations:
-
- Type 2 diabetes patients: Those prescribed semaglutide or tirzepatide (both in the same class of drugs as Wegovy) demonstrated a 4.28-fold increased risk of developing NAION compared to patients using alternative diabetes medications
-
- Linking Wegovy to Optic Nerve Problems: These findings represent the first substantial epidemiological evidence linking Wegovy vision problems to this severe optic nerve disorder. The magnitude of risk elevation has prompted calls for enhanced surveillance protocols and patient counseling regarding potential ocular complications.
Drug Safety Findings
- The Link Between Wegovy and Vision Loss: Reviews of post-marketing surveillance data have found scientific evidence linking Wegovy to vision loss through multiple reporting channels. Regulatory agencies have received notifications of eye-related side effects at rates higher than expected for the general population. These analyses have documented:
-
- Disproportionate reporting ratios for NAION among users of GLP-1 receptor agonists
- Increased progression of diabetic retinopathy during treatment intensification
- Reports of sudden visual disturbances requiring immediate eye care intervention
Other Types of Vision Problems Reported with Wegovy Use
Breaking Down the Wegovy Vision Problems: Wegovy use has been linked to various eye issues beyond just NAION. These problems involve different parts of the eye and include several specific conditions that require thorough assessment and supervision.
1. Diabetic Retinopathy
-
- Diabetic retinopathy is a major concern for patients using GLP-1 receptor agonists like Wegovy. It involves gradual harm to the blood vessels in the retina, which can be seen through signs such as microaneurysms, bleeding, and new blood vessel growth. Interestingly, while Wegovy treatment aims to control blood sugar levels quickly, it may unintentionally worsen retinopathy in susceptible individuals, especially those with existing microvascular issues. This is because a sudden drop in blood glucose levels can disturb the retinal blood vessels, causing them to become more permeable and leading to insufficient blood supply.
2. Macular Edema
-
- Another complication associated with Wegovy is macular edema, characterized by fluid buildup in the macula—the central part of the retina responsible for sharp vision. As a result, patients may experience blurred central vision, difficulty reading, and altered color perception. This condition occurs when the barrier between the blood vessels and retinal tissue is disrupted, allowing fluid leakage into the macula and compromising visual clarity.
3. Dry Eye Syndrome
-
- Wegovy Vision Side Effects and Dry Eyes: Multiple case reports have documented instances of dry eye syndrome among users of GLP-1 receptor agonists. Patients may feel discomfort on the surface of their eyes, experience burning sensations, and have intermittent blurry vision due to insufficient tear production or excessive evaporation of tears. The underlying mechanism could involve effects on the autonomic nervous system or inflammatory responses triggered by the medication.
4. Myodesopsias (Floaters)
-
- Another phenomenon reported by some patients is myodesopsias, commonly known as floaters. These individuals perceive dark spots or cobweb-like shapes moving across their field of vision—an occurrence stemming from changes in the vitreous humor or disturbances within the retina.
5. Blurred Vision from Hypoglycemia
-
- An acute complication that can arise when Wegovy’s glucose-lowering effects become too strong is blurred vision due to hypoglycemia. The visual cortex and retinal neurons rely on a steady supply of glucose for proper functioning; when this supply is compromised, it leads to impaired visual processing—resulting in temporary but potentially hazardous vision impairment.
Scientific Studies Investigating the Correlation Between Wegovy and Vision Loss Incidents
- Investigating the Link to GLP-1 Receptors: The medical community has initiated rigorous investigations into the potential association between GLP-1 receptor agonists and adverse ocular outcomes. Research published in JAMA Ophthalmology has provided critical insights into the relationship between these medications and vision-threatening conditions, particularly nonarter
Insights on Mechanisms Behind Adverse Effects
- Wegovy Eye Problems Due to Reduced Blood Flow to the Optic Nerve: Research emphasizes the role of rapid glycemic fluctuations in compromising optic nerve perfusion. The sudden normalization of blood glucose levels, while therapeutically beneficial for metabolic control, may paradoxically create conditions that reduce blood flow to the optic nerve head.
- Normalization of Ichemia (Oxygen Supply to Blood): This phenomenon, termed “normalization ischemia,” represents a critical pathway through which GLP-1 medications may precipitate vision loss.
Clinical Monitoring Protocols
- Establishing a Bassline Assessment: Physicians recommend establishing baseline ophthalmologic assessments before initiating Wegovy therapy. This initial evaluation creates a reference point for detecting subsequent changes in ocular health. Patients should receive clear instructions to report any visual disturbances immediately, including:
-
- Sudden onset of blurred vision or visual field defects
- Unexplained eye pain or pressure
- Changes in color perception
- Appearance of floaters or flashing lights
- Difficulty with night vision or contrast sensitivity
- FDA Warnings: The FDA warnings on GLP-1 drugs underscore the necessity for heightened vigilance during the first several months of treatment, when metabolic changes occur most rapidly. Healthcare teams must educate patients about the distinction between temporary visual fluctuations related to blood sugar stabilization and potentially serious complications requiring urgent evaluation.
NAION RATE OF INCIDENCE BY DRUG
Legal Landscape: The Wegovy Blindness Lawsuit
- Wegovy Blindness Lawsuit Legal Landscape: The emergence of scientific evidence connecting semaglutide-based medications to vision complications has led to a surge of legal action against weight loss drug manufacturers.
- Failure to Warn: Multiple lawsuits have been filed against Novo Nordisk, the pharmaceutical company responsible for manufacturing Wegovy and Ozempic, with plaintiffs claiming the company failed to adequately warn patients and healthcare providers about the risk of developing NAION.
Primary Legal Claims
Plaintiffs pursuing legal action against Novo Nordisk have centered their claims on several key allegations:
- Failure to warn: The manufacturer did not provide sufficient information about the potential risk of NAION and other serious vision complications on product labeling or through direct communication with medical professionals
- Inadequate testing: Claims that pre-market clinical trials failed to identify or properly investigate the connection between semaglutide use and optic nerve damage
- Negligent misrepresentation: Allegations that the company promoted Wegovy’s safety profile without disclosing known or discoverable risks to ocular health
- Design defect: Arguments that the medication’s formulation presents unreasonable dangers that outweigh its therapeutic benefits for certain patient populations
Legal Representation and Case Development
- Wegovy Blindness Lawsuit: Law firms have begun accepting cases from individuals who experienced Wegovy and Vision Loss or similar GLP-1 receptor agonists. The Law Offices of Timothy L. Miles and other firms focusing on pharmaceutical litigation have established dedicated practices to represent affected patients. These Wegovy blindness lawyers are building cases by collecting medical records, ophthalmologic evaluations, and documentation of the temporal relationship between medication use and vision deterioration.
- Law Offices of Timothy L. Miles: If you suffered Wegovy vision problems, contact the Law Offices of Timothy L. Miles for a free case evaluation as you may be eligible for a Wegovy Blindness Lawsuit and possibly entitled to significant compensation. (855) 846–6529 or [email protected].
- The Legal Proceedings: Involve patients who suffered varying degrees of Wegovy vision problems, from partial visual field defects to complete blindness in one or both eyes. Many plaintiffs report that their vision loss occurred suddenly and without prior warning, fundamentally altering their ability to work, drive, and maintain independence in daily activities.
Patient Experiences and Testimonials with Wegovy Vision Problems
- Patient Testimonials Vision Loss Weight Loss Drugs: Reveal the profound personal toll of unexpected Wegovy vision side effects. Individuals who started treatment with semaglutide-based medications for weight management report experiencing sudden and alarming changes to their vision, often without any warning or proper information about potential risks.
- One patient described waking one morning to discover complete vision loss in their left eye after several months of Wegovy treatment. The sudden onset occurred without pain, leaving the individual disoriented and frightened. Despite immediate medical intervention, eye specialists confirmed permanent damage to the optic nerve consistent with NAION, effectively ending the patient’s career as a graphic designer and fundamentally altering their independence.
- Another account details a 52-year-old individual who noticed progressive blurring in their peripheral vision while using the medication for diabetes management and weight reduction. Within weeks, the condition worsened to significant visual field defects that made driving impossible. The patient reports experiencing:
-
- Inability to perform routine tasks requiring depth perception
- Loss of employment due to vision-dependent job requirements
- Psychological distress including anxiety and depression
- Financial strain from medical expenses and lost income
- Dependence on family members for daily activities
- Wegovy Vision Problems: Extend beyond complete blindness to include various degrees of visual impairment that significantly reduce quality of life. Patients describe struggling with reading, recognizing faces, and navigating unfamiliar places. The permanence of NAION-related damage adds to the emotional burden, as affected individuals face irreversible changes to their eyesight.
- Failure to Warn: Medical records submitted in legal proceedings document cases where patients specifically asked about potential side effects before starting treatment but received no information regarding eye risks. These documented exchanges highlight allegations that healthcare providers and pharmaceutical manufacturers failed to properly communicate the possibility of vision-threatening complications associated with GLP-1 receptor agonist therapy.
Regulatory Responses to Emerging Evidence Linking GLP-1 Receptor Agonists with Eye Disorders
- Regulatory Monitoring: The FDA has been actively monitoring reports of adverse events related to semaglutide-based medications since they were approved.
- Safety Data Review: After research was published suggesting a possible link between GLP-1 receptor agonists and NAION, the FDA began conducting thorough reviews of safety data collected after the drugs were on the market. The goal is to understand the extent and seriousness of reported eye problems.
FDA Drug Safety Communications and Monitoring
- FDA’s Adverse Event Reporting System (FAERS) Database: Has recorded many cases of Wegovy vision side effects as well as Ozempic, and similar medications. These reports have led the agency to take several actions:
-
- Conduct detailed analyses of temporal relationships between medication initiation and vision loss onset
- Evaluate the biological plausibility of proposed mechanisms linking GLP-1 receptor agonists to optic nerve damage
- Coordinate with pharmaceutical manufacturers to assess clinical trial data for previously unidentified safety signals
- Consider potential label modifications to reflect emerging safety information
Pharmaceutical Manufacturer Obligations
- Novo Nordisk, the manufacturer of Wegovy and Ozempic, is facing increased regulatory scrutiny regarding whether it adequately warns healthcare professionals and patients about potential risks. The company has submitted additional safety data to regulatory authorities while also defending its position in the Wegovy Blindness Lawsuit. Regulatory expectations require manufacturers to:
-
- Report serious adverse events within specified timeframes
- Update prescribing information when new safety data emerges
- Conduct post-marketing surveillance studies to identify rare complications
- Implement Risk Evaluation and Mitigation Strategies (REMS) when necessary
International Regulatory Coordination
- International Regulatory Bodies: The European Medicines Agency (EMA) and Health Canada are also reviewing the safety profiles of GLP-1 receptor agonists. These agencies work together through international networks focused on monitoring drug safety in order to share information about risks associated with these medications.
- FDA Drug Safety Alerts: When significant dangers are discovered, the FDA drug safety alerts system allows for quick communication of important details directly to healthcare providers.
 How to Identify Symptoms and Seek Medical Help for NAION
How to Identify Symptoms and Seek Medical Help for NAION
- Recognizing The Symptoms of NAION: Is crucial in minimizing permanent vision damage for patients undergoing treatment with semaglutide-based medications. The condition usually presents itself through specific eye warning signs that require immediate medical attention.
Primary Symptoms Requiring Urgent Evaluation
- Patients using Wegovy or related GLP-1 receptor agonists should be vigilant about certain visual disturbances:
-
- Sudden, painless vision loss in one eye, occurring upon waking or developing rapidly throughout the day
-
- Blurred vision or clouding that does not improve with blinking or rubbing the eyes
-
- Visual field defects, especially loss of peripheral or central vision creating blind spots
-
- Change in color perception, where colors appear washed out or less vibrant
-
- Difficulty with depth perception affecting daily activities such as driving or reading
- The Painless Nature of NAION: Sets it apart from other eye emergencies. Patients often describe the experience as “a curtain coming down” over their vision or noticing significant changes immediately upon opening their eyes in the morning.
Critical Timeline for Medical Intervention
- Seek Immediate Medical Help: The opportunity for potential intervention becomes much smaller within hours of symptom onset. Patients experiencing any sudden Wegovy vision problems must contact an ophthalmologist or go to an emergency department without delay.
- Consult Retinal Specialist: Regular eye care providers may not have the specialized equipment needed for diagnosing NAION, making it necessary to consult with a neuro-ophthalmologist or retinal specialist for accurate assessment.
Diagnostic Procedures and Documentation
- Get a Comprehensive Medical Evaluation for NAION: Which includes comprehensive eye examination, optical coherence tomography (OCT), visual field testing, and fluorescein angiography. Patients should inform healthcare providers about their current use of semaglutide medications, dosage information, and treatment duration.
- Document Everything Related to Wegovy and Vision Loss: Documenting the timing relationship between medication use and symptom onset strengthens both medical records and a potential Wegovy Blindness Lawsuit. Keeping detailed records of all medical appointments, diagnostic test results, and treatment recommendations provides essential evidence for understanding the progression of vision impairment.
Legal Options for Affected Patientss: Wegovy Blindness Lawsuit
- Medical Timeline Documentation: Patients who have experienced vision complications following Wegovy use have specific legal options to seek compensation for their injuries.
- Wegovy Blindness Lawsuit: The process of filing a Wegovy Blindness Lawsuit begins with documenting the medical timeline connecting semaglutide administration to the onset of visual symptoms.
Initial Steps for Potential Claimants to file a Wegovy Blindness Lawsuit
Affected individuals should gather comprehensive medical records demonstrating:
- Prescription history for Wegovy or related GLP-1 receptor agonists
- Ophthalmologic evaluations confirming NAION diagnosis or other vision impairments
- Documentation of symptom onset dates relative to medication use
- Records of all treatments attempted to address Wegovy vision problems
- Evidence of functional limitations resulting from vision loss
Consultation with Experienced Legal Counsel
- Contact an Experienced Wegovy Blindness Lawyer: Engaging attorneys experienced in pharmaceutical litigation provides essential guidance through complex legal proceedings. Law firms, such as the Law Offices of Timothy L. Miles practicing in defective drug cases evaluate the strength of individual claims by examining medical evidence, assessing damages, and determining liability theories.
- Contingency Fee Agreements: The Law Offices of Timothy L. Miles takes all cases on a contingency fee basis so it does not cost anything to hire a Wegovy Blindness Lawyer if you are eligible for a Wegovy Blindness Lawsuit. Call today for a free case evaluation. (855) 846–6529 or [email protected].
Individual Lawsuits Versus Multidistrict Litigation
- Wegovy Blindness Lawsuit: Patients may pursue individual claims or participate in consolidated legal actions. Individual lawsuits allow for personalized case development addressing specific circumstances and damages.
- Multidistrict litigation (MDL): consolidates similar claims for pretrial proceedings, streamlining discovery and potentially expediting resolution while preserving individual trial rights.
Potential Compensation Categories for a Wegovy Blindness Lawsuit
Successful claims may recover damages for:
- Medical expenses including emergency care, ongoing treatment, and vision rehabilitation
- Lost wages and diminished earning capacity
- Pain and suffering associated with permanent vision impairment
- Loss of quality of life and inability to perform daily activities
- Punitive damages if evidence demonstrates willful misconduct by manufacturers
Conclusion
- Wegovy Blindness Lawsuit: Highlights the critical need for comprehensive Wegovy vision problems awareness among patients and healthcare providers. As evidence continues to emerge regarding Wegovy vision side effects and potential Wegovy eye damage, vigilance remains paramount for all individuals using semaglutide-based medications.
- Remain Proactive: Patients must maintain proactive communication with their ophthalmologists and prescribing physicians, reporting any visual disturbances immediately. The connection between GLP-1 receptor agonists and NAION demands heightened scrutiny from both medical professionals and regulatory agencies. Staying informed about evolving research, FDA updates, and legal developments empowers patients to make educated decisions about their treatment options.
- Lack of Transparency: The pharmaceutical industry bears responsibility for transparent disclosure of all potential adverse effects, including rare but serious complications affecting vision. As litigation progresses and scientific understanding deepens, the medical community must prioritize patient safety through rigorous monitoring protocols and comprehensive informed consent processes. Knowledge serves as the most effective tool for protecting vision health while pursuing weight management goals with these medications.
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com