Introduction to the Wegovy and NAION Update
Welcome to the authoritative Wegovy and NAION Update. Wegovy (semaglutide) has helped many people achieve clinically meaningful weight loss and improve cardiometabolic health. However, patients and clinicians have raised important questions about rare but serious eye events, including non-arteritic anterior ischemic optic neuropathy (NAION) and Wegovy and vision loss. associated with the use of this medication. If you are taking Wegovy, considering Wegovy, or using a related GLP‑1 medication, this guide explains what NAION is, what is currently known in 2026, what symptoms warrant urgent care, and how to approach risk reduction and monitoring with your clinicians.
This is a patient education article, not a substitute for individualized medical advice. If you experience sudden vision changes while on Wegovy, treat it as an emergency.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Wegovy Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.

1) What Wegovy is, and why it is widely used
Wegovy is a prescription medication containing semaglutide, a GLP‑1 receptor agonist (glucagon-like peptide‑1 receptor agonist). It is approved for chronic weight management in eligible adults and, in some regions, certain adolescents, as part of a comprehensive plan that includes nutrition, physical activity, and behavior change.
GLP‑1 receptor agonists work through several mechanisms that support weight loss and metabolic health:
- Appetite regulation via central satiety pathways
- Slower gastric emptying, which can reduce hunger and meal size
- Improved glucose regulation through effects on insulin and glucagon signaling
Because obesity is a chronic disease associated with diabetes, cardiovascular disease, fatty liver disease, sleep apnea, and more, Wegovy is often positioned as a long-term, proactive intervention. That long-term view also makes safety monitoring essential, particularly for rare events like NAION, that may appear only after broad real-world use.
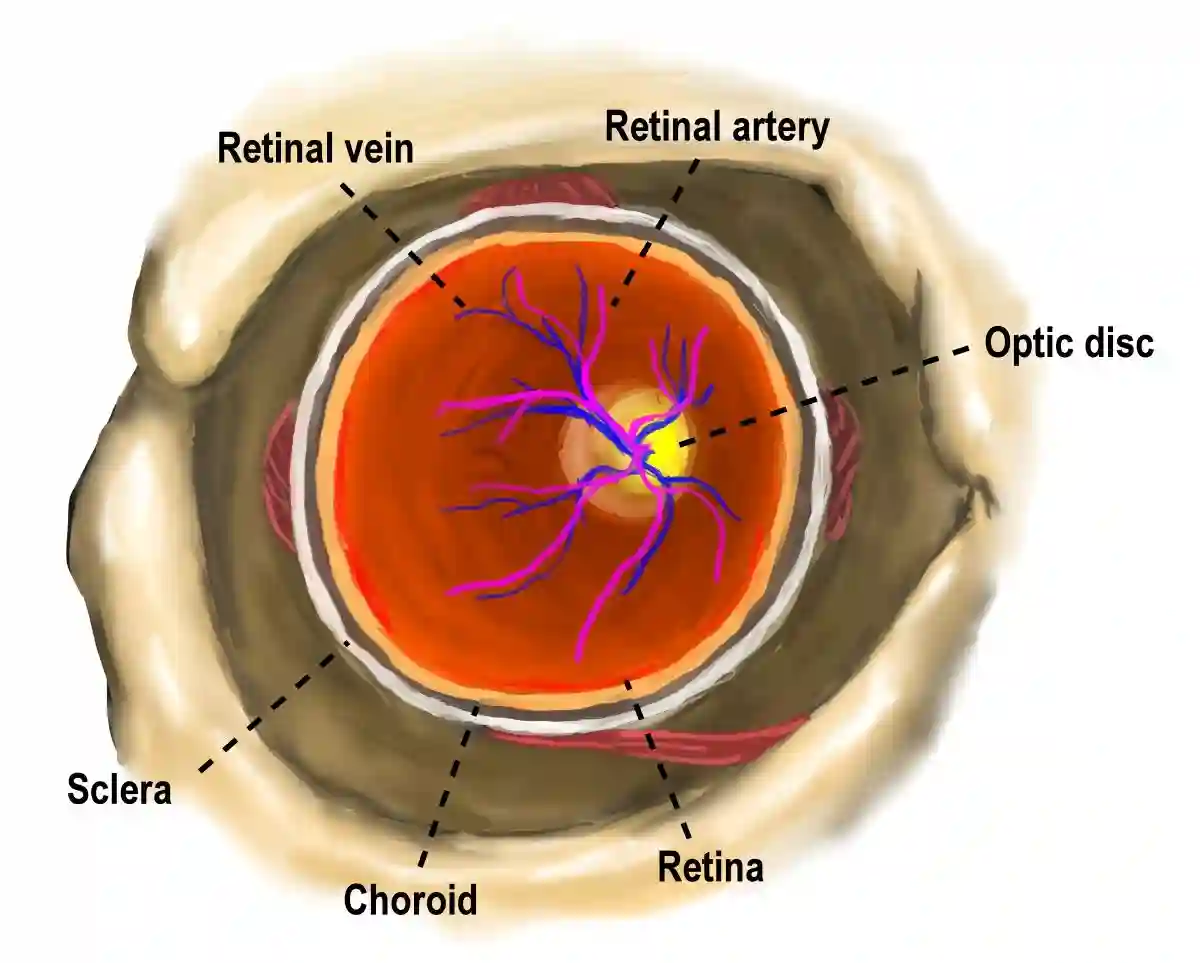
2) What NAION is (in clear terms)
NAION stands for Non-Arteritic Anterior Ischemic Optic Neuropathy.
- Non-arteritic means it is not caused by inflammation of arteries (as in giant cell arteritis).
- Anterior ischemic optic neuropathy means damage occurs at the front portion of the optic nerve due to reduced blood flow (ischemia).
What NAION typically feels like
NAION often presents as:
- Sudden, painless vision loss in one eye (commonly noticed on waking)
- Dark or missing area in the visual field (often the lower or upper half)
- Blurred vision, reduced contrast, or a “shadow/curtain” effect
- Sometimes reduced color saturation
NAION is considered an ocular emergency. Even if vision improves later, early evaluation is crucial to confirm the diagnosis, rule out other causes, and address risk factors that may reduce the chance of additional injury.
3) Why patients are hearing about Wegovy and NAION
Patients are hearing about Wegovy and NAION for a straightforward reason: NAION is a rare condition, and any credible signal connecting a common medication to a rare but serious event deserves careful attention.
In 2026, the discussion is shaped by three realities:
- GLP‑1 medications are used at scale, including by people with multiple vascular risk factors.
- NAION has known baseline risk factors that overlap strongly with obesity, diabetes, and sleep apnea.
- The presence of reports does not automatically prove that a medication causes NAION. However, it does justify patient-facing guidance focused on symptom recognition and risk management.
This situation underscores the importance of robust pharmacovigilance systems, which are designed to detect rare events that may not appear clearly in pre-approval trials.
Notably, there have been instances where specific GLP-1 medications have been linked to NAION, as seen with Trulicity. Similar concerns have arisen regarding Saxenda, resulting in a lawsuit related to NAION. Additionally, Zepbound has also faced scrutiny in this regard, leading to a Zepbound NAION lawsuit. Mounjaro is another medication under investigation for its potential link to NAION, as highlighted in this Mounjaro NAION lawsuit. Furthermore, there have been updates regarding Zepbound’s association with blindness, which adds another layer of concern for patients using this medication ([Zepbound blindness lawsuit update
4) What is currently known (and not known) as of 2026
What is known
- NAION occurs in the general population and is more common in people with vascular and metabolic risk factors.
- Many people eligible for Wegovy have risk factors that also increase NAION risk, including hypertension, type 2 diabetes, hyperlipidemia, and obstructive sleep apnea.
- Patients and clinicians have reported NAION events in temporal association with semaglutide and other GLP‑1 drugs in real-world settings, prompting continued scientific and regulatory attention.
What is not definitively known
- Whether Wegovy causes NAION in a direct way, increases risk in a subset of patients, or whether reported cases reflect underlying baseline risk in the treated population.
- Whether risk, if present, is tied to dose escalation, rapid weight loss, dehydration/hypotension, glycemic shifts, sleep apnea severity, or optic nerve anatomy.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Wegovy Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.

The practical takeaway for patients
Until the evidence base is fully settled, the best patient strategy is to focus on what is controllable and clinically sound:
- Know NAION warning signs.
- Get urgent evaluation for sudden vision changes.
- Manage modifiable risk factors (blood pressure, sleep apnea, hydration).
- Coordinate care between your prescribing clinician and your eye specialist.
5) Who is at higher baseline risk for NAION
NAION is often described as multifactorial. Risk is influenced by both systemic vascular factors and local optic nerve anatomy.
Common risk factors include:
- Age (more common in older adults)
- Hypertension
- Diabetes / insulin resistance
- High cholesterol / atherosclerotic disease
- Smoking
- Obstructive sleep apnea (OSA)
- Nocturnal hypotension (blood pressure dipping too low overnight)
- “Crowded” optic disc anatomy (often described as a “disc at risk”)
Many of these overlap with the clinical profile of patients treated for obesity. This overlap matters because it can complicate interpretation of any medication safety signal. For instance, similar to the concerns raised about Wegovy, there have been recent reports linking other weight loss medications like Mounjaro to potential health risks such as NAION, as discussed in this Mounjaro lawsuit update. Additionally, patients using Trulicity have also reported adverse effects that may warrant further investigation, akin to the ongoing inquiries surrounding Trulicity lawsuits.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Wegovy Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
6) Symptoms that require emergency evaluation
If you are taking Wegovy (or any GLP‑1 medicine) and experience the symptoms below, seek same-day emergency evaluation (emergency department or urgent ophthalmology):
- Sudden vision loss in one eye
- New blind spot, missing area, or “shadow” in vision
- Sudden decrease in visual clarity or contrast
- Sudden change in color perception
- Vision loss that is painless, especially on waking
Do not wait to “see if it improves.” Time matters because other serious diagnoses can mimic NAION, including:
- retinal detachment
- retinal artery occlusion
- optic neuritis
- stroke-related visual field loss
- giant cell arteritis (a medical emergency requiring immediate treatment)
If you are older than 50 and have symptoms like new headache, scalp tenderness, jaw pain with chewing, fever, or unexplained weight loss along with vision symptoms, clinicians must urgently evaluate for giant cell arteritis.
It’s crucial to note that NAION has been linked to the use of GLP-1 medications like Wegovy. If you suspect NAION as a result of taking Wegovy or another GLP-1 medicine, seek immediate medical attention, as this condition can lead to severe vision loss.
7) If you suspect NAION: what will happen at the eye exam
A typical urgent workup may include:
- Visual acuity and color vision testing
- Pupil exam (looking for a relative afferent pupillary defect)
- Dilated fundus exam (often shows optic disc swelling in acute NAION)
- Optical coherence tomography (OCT) to evaluate optic nerve head and retinal nerve fiber layer
- Visual field testing to map the defect pattern
- Sometimes blood tests (especially to rule out giant cell arteritis in older patients)
- In selected cases, MRI or additional imaging to exclude compressive or inflammatory causes
If NAION is diagnosed, your eye specialist will typically recommend close follow-up and coordinated management of vascular risk factors. It’s also important to be aware of potential legal actions related to Wegovy and vision loss, as there have been reports linking the drug with serious ocular side effects.
8) What to do about Wegovy if NAION is diagnosed or suspected
This decision must be individualized. There is no universal rule that applies to every patient because the benefit-risk balance differs substantially across patients.
A clinically structured approach often includes:
- Immediate medical evaluation first. Confirm diagnosis and rule out emergencies.
- Inform the prescribing clinician promptly. Provide ophthalmology documentation when available.
- Joint risk assessment. This often involves primary care, obesity medicine/endocrinology, and ophthalmology or neuro-ophthalmology.
Possible outcomes may include:
- Temporary hold of Wegovy while evaluation is underway
- Discontinuation if the treating team believes the medication may contribute to risk, or if risk tolerance is low
- Continuation with enhanced monitoring if the team concludes benefits outweigh risks and alternative explanations are more likely
Patients should not stop prescription medications abruptly without clinician guidance. The exception is when a clinician advises immediate discontinuation, or when emergency services instruct it as part of an acute evaluation.
9) Practical risk-reduction steps for patients on Wegovy (or considering it)
Even though NAION risk cannot be reduced to zero, many risk modifiers are aligned with good long-term health and good obesity care. These actions are proactive, measurable, and reasonable.
A) Optimize blood pressure, but avoid overly aggressive nighttime lows
- Track home blood pressure if advised.
- Ask your clinician whether you are at risk for nocturnal hypotension, especially if you take blood pressure medication at night.
- Do not change dosing times without medical direction.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Wegovy Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
B) Screen and treat obstructive sleep apnea
OSA is a recurring theme in NAION risk discussions. If you snore, wake unrefreshed, have witnessed apneas, or experience daytime sleepiness:
- Ask for a sleep study or validated screening.
- Use CPAP or other prescribed therapy consistently if diagnosed.
C) Prevent dehydration and severe gastrointestinal fluid loss
GLP‑1 medicines can cause nausea, vomiting, diarrhea, and reduced intake, especially during dose escalation.
- Maintain steady fluid intake.
- Treat persistent vomiting or diarrhea as a medical issue, not a routine side effect.
- Discuss anti-nausea strategies early, including slower titration when clinically appropriate.
D) Avoid abrupt, unsupervised dose escalation
Follow the prescribed titration schedule. If side effects are significant, discuss:
- holding at a lower dose longer
- stepping back temporarily
- targeted dietary adjustments to improve tolerance
E) Control diabetes and lipids using defined targets
If you have diabetes or prediabetes, avoid extremes:
- Prevent prolonged hyperglycemia.
- Avoid frequent hypoglycemia if on insulin or sulfonylureas.
- Follow lipid and cardiovascular prevention plans as recommended.
F) Do not ignore “minor” vision changes
Patients sometimes delay evaluation because symptoms are subtle. With NAION and other optic nerve disorders, delay can increase uncertainty and limit options.
10) Should you get a baseline eye exam before starting Wegovy?
A baseline eye exam is not universally mandated for every patient starting Wegovy. However, it is often a prudent step if you have:
- diabetes (especially with known retinopathy)
- prior optic nerve disease
- glaucoma or optic disc anomalies
- history of NAION in the other eye
- strong vascular risk factors plus sleep apnea symptoms
- new or unexplained visual complaints
A practical approach is to ask your clinician one question:
“Given my risk factors, should I have a baseline comprehensive eye exam, and should it include optic nerve evaluation and visual field testing?”
For many patients, a standard comprehensive exam with optic nerve documentation is sufficient. In higher-risk cases, the clinician may suggest neuro-ophthalmology input.
However, it’s important to be aware of potential Wegovy-related eye side effects, which could necessitate more frequent monitoring by an eye specialist.
11) Wegovy, diabetes eye disease, and why the distinction matters
Many patients understandably group all eye risks together, but there are important distinctions:
- Diabetic retinopathy involves damage to retinal blood vessels and can worsen with rapid changes in glucose control in certain circumstances.
- NAION involves the optic nerve and is not the same condition, even though both relate to vascular health.
If you have diabetes, your clinician may already recommend regular retinal screening. That screening does not automatically detect NAION risk, but it does strengthen continuity of eye care, which is beneficial.
However, it’s crucial to be aware of potential Wegovy-related eye damage, which can exacerbate existing conditions or introduce new risks.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Wegovy Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
12) Questions to ask your clinician (use this checklist)
Bring these questions to your next appointment if you are concerned:
- What is my baseline risk for NAION, given my age, blood pressure, diabetes status, cholesterol, and sleep apnea risk?
- Do you recommend a baseline eye exam before starting or escalating Wegovy?
- How should I handle vomiting, diarrhea, or inability to eat or drink, especially during dose increases?
- Do any of my medications increase the chance of nighttime low blood pressure?
- If I have sudden vision symptoms, where should I go immediately, and who should I notify?
- If an eye specialist suspects NAION, what is our plan regarding continuing, pausing, or stopping Wegovy?
- What alternatives exist for weight management if Wegovy is not appropriate for me?
This is governance in personal healthcare: define roles, define escalation paths, and document decisions before a crisis occurs.
Do not stop solely based on online discussion. Instead, evaluate your personal risk profile and your symptom status:
- If you have acute vision symptoms, seek emergency evaluation.
- If you are asymptomatic but concerned, schedule a focused discussion with your prescribing clinician and consider an eye exam based on your risk factors.
13) The forward-looking patient strategy for 2026
In modern obesity care, success requires more than initiating therapy. It requires sustained safety monitoring, transparent decision-making, and early escalation when warning signs appear. The NAION conversation reinforces three principles:
- Clarity over speculation: focus on symptoms and risk factors you can measure.
- Coordination over fragmentation: align your prescriber and your eye clinician.
- Preparedness over reaction: know what to do before an urgent event occurs.
Wegovy can be a valuable tool. Vision is also non-negotiable. A responsible plan respects both.
It’s important to stay informed about potential vision loss issues related to other weight loss medications, such as Zepbound or Mounjaro which also have ongoing vision loss lawsuits. Similarly, those using Saxenda should be aware of the vision loss lawsuit updates associated with that drug as well.
14) Summary (what to remember)
- NAION is a rare, serious optic nerve event that can cause sudden, painless vision loss, often in one eye.
- In 2026, there is ongoing attention to NAION reports in patients using semaglutide and related drugs, but causality and mechanism are not definitively established for every scenario.
- If you have sudden vision changes, seek same-day emergency evaluation.
- Reduce risk by addressing sleep apnea, blood pressure patterns (including nighttime lows), dehydration, and cardiometabolic risk factors.
- Ask your clinician whether you need a baseline eye exam and what your emergency plan is if symptoms occur.
If you want, share your age range, whether you have diabetes, hypertension, sleep apnea symptoms, and whether you have any prior eye disease. I can help you draft a concise question list to take to your clinician and suggest what information to request from an eye exam report.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Wegovy Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
Frequently Asked Questions about Wegovy and NAION
What is Wegovy and how does it help with weight management?
Wegovy is a prescription medication containing semaglutide, a GLP-1 receptor agonist approved for chronic weight management in eligible adults and some adolescents. It supports weight loss through appetite regulation, slower gastric emptying, and improved glucose regulation, making it part of a comprehensive plan including nutrition, physical activity, and behavior change.
What is Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION)?
NAION is a condition involving sudden, painless vision loss in one eye due to reduced blood flow at the front portion of the optic nerve. It often presents with dark or missing areas in the visual field, blurred vision, or a shadow effect. NAION is an ocular emergency requiring immediate medical evaluation.
Why are patients concerned about the link between Wegovy and NAION?
Patients are concerned because NAION is a rare but serious eye event that has been reported in association with GLP-1 medications like Wegovy. Given the widespread use of these drugs among people with overlapping vascular risk factors, credible signals connecting them to NAION warrant careful attention and patient education on symptom recognition and risk management.
Are other GLP-1 medications also linked to NAION?
Yes, other GLP-1 receptor agonists such as Trulicity, Saxenda, Zepbound, and Mounjaro have faced scrutiny or lawsuits related to potential links with NAION or blindness. This highlights the importance of ongoing pharmacovigilance to monitor rare but serious adverse events associated with this class of medications.
What symptoms should prompt urgent care if I am taking Wegovy or similar GLP-1 drugs?
If you experience sudden vision changes such as painless vision loss in one eye, dark or missing areas in your visual field, blurred vision, reduced color saturation, or a shadow/curtain effect while on Wegovy or related GLP-1 medications, you should seek emergency medical evaluation immediately as these may indicate NAION.
What is currently known about the risk of NAION for people using Wegovy as of 2026?
As of 2026, it is known that NAION occurs more commonly in individuals with vascular and metabolic risk factors—many of whom are eligible for Wegovy. While reports exist linking GLP-1 medications to NAION, causation has not been definitively established. Ongoing monitoring and patient-clinician communication are essential for managing potential risks.

