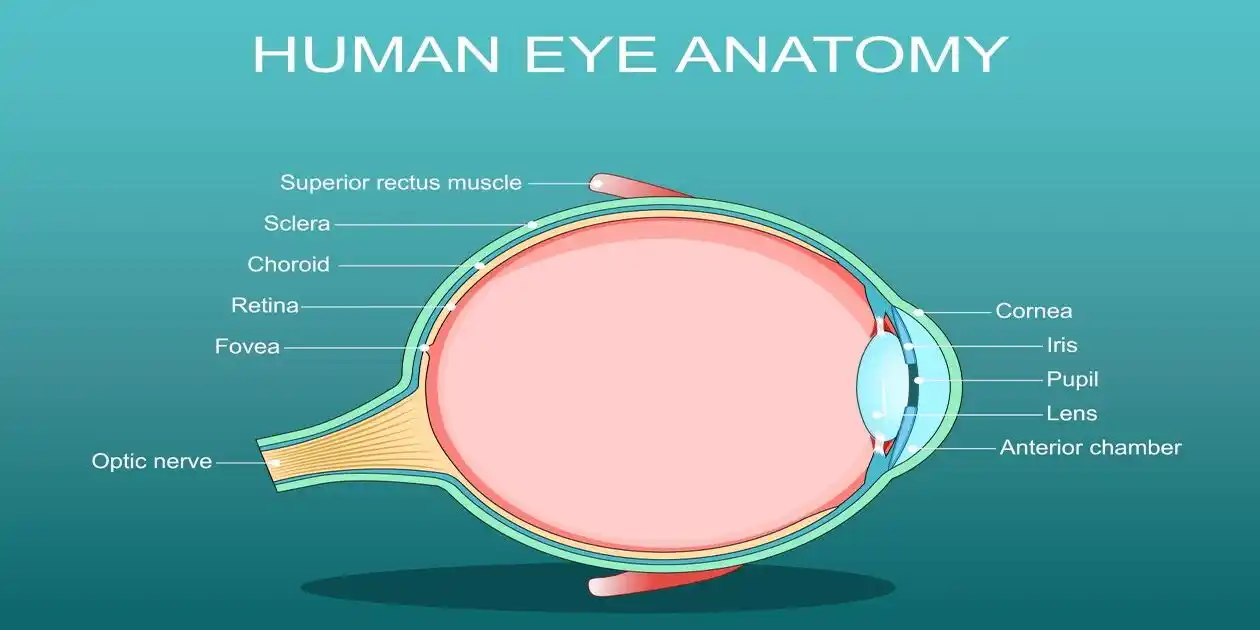
Introduction to Saxenda and NAION Update
Welcome to this authoritative Saxenda and NAION Update. As of April 2026, Saxenda (liraglutide) and other GLP-1 agonists are heavily involved in litigation regarding Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION), a serious, often permanent, vision loss condition.
Federal multidistrict litigation (MDL 3163) was established in late 2025 to consolidate lawsuits alleging that manufacturer Novo Nordisk failed to warn users about the risk of blindness.
Key Updates on Saxenda and NAION
- Legal Action (2026): A federal MDL is active, with a “Science Day” set for April 2, 2026, to educate the court on the scientific issues surrounding GLP-1 drugs and vision loss.
- Increased Risk Studies: Research, including studies published in JAMA Ophthalmology and by Epic Research, has indicated a significantly higher risk of developing NAION in patients taking liraglutide, with some findings suggesting a 179% higher likelihood compared to other medications.
- The Condition (NAION): Known as “eye stroke,” NAION results from blocked blood flow to the optic nerve, causing sudden, painless, and often permanent vision loss in one or both eyes.
- Saxenda Vision Loss lawsuit Claims: Plaintiffs claim that despite evidence linking these medications to NAION, warnings were not included on the labels, failing to inform patients of the risk of permanent blindness.
- More Saxenda Vision Loss Lawsuits involving drugs like Saxenda (Zepbound, Ozempic, Wegovy, Trulicity, Mounjaro) have now been filed in federal court. Over the last few weeks, more than 100 new cases have been filed in federal court by people who say that they experienced serious stomach issues or Saxenda and NAIONafter taking a GLP-1.

The “Gold Standard” Diagnostic Tests
- Gastric Emptying Scintigraphy: The primary “gold standard” test where a patient’s digestion is tracked via a radio-labeled meal.
- Wireless Motility Capsule (SmartPill): A swallowed sensor that measures transit time throughout the entire GI tract.
- 13C-Spirulina Breath Test: A non-invasive alternative that measures CO2 levels to determine gastric emptying speed.
Insufficient Evidence (What to Avoid)
- You should be advissed that common scans like CT scans, MRIs, and endoscopies are generally insufficient for the MDL. While these can rule out physical obstructions, they do not measure the actual speed of stomach motility, which is the core requirement for a gastroparesis diagnosis.
New Vision Loss (NAION) Track
- A dedicated “Vision Injury Track” was established in late 2025. This specifically addresses NAION (Non-Arteritic Anterior Ischemic Optic Neuropathy), an “eye stroke” linked to GLP-1 drugs like Trulicity and Saxenda.
Saxenda and Blurry Vision vs. NAION Update
- The Trap: Most Saxenda users are told Saxenda and blurry vision just a temporary side effect of blood sugar changes.
- The Legal Reality: New 2026 lawsuits (MDL No. 3163) argue that this “blurriness” can actually be an early sign of Saxenda and NAION, also called an “eye stroke,” which leads to permanent blindness.
- Key Distinction: Temporary blurriness usually fluctuates; NAION typically presents as sudden, painless vision loss in one eye, often noticed right after waking up.
Saxenda Eye Pain & Inflammation
- While not as common as Saxenda and blurry vision or Saxenda eye pain it is being cited in some of the roughly 3,363 GLP-1 lawsuits active as of March 2026.
- Calls continue to come in with Saxenda eye pain, blurry vision, Saxenda and floaters, and unfortunatley permanent loss of vision in one eye and a very painful condition, as opposed to diabetic retinopathy, which is usually painless.
Saxenda and Floaters and Retinal Issues
- Newer reports of Saxenda and floaters (spots or strings in vision) are being investigated as potential signs of retinal detachment or accelerated diabetic retinopathy complications.
- The Novo Nordisk Saxenda label does not mention retinopathy and does not explicitly warn about the sudden onset of floaters leading to permanent Saxenda eye damage.
High-Value Litigation Terms (Permanent Injury)
- Saxenda and NAION: Often called an “eye stroke,” this is the core of current Saxenda Vision Loss lawsuit and Zebpound vision loss lawsuitss.. It occurs when blood flow to the optic nerve is blocked, leading to sudden, irreversible blindness.
- Optic Nerve Damage: Damage to the “cable” connecting the eye to the brain. Unlike some vision issues, this damage typically does not regenerate, meaning the vision loss is permanent.
- Failure to Warn: The legal basis for most claims. Plaintiffs argue that manufacturers like Eli Lilly (Zepbound/Trulicity) failed to explicitly warn that these drugs could cause permanent Saxenda blindness or Saxenda and NAION.
Saxenda and Floaters
- Dark specks, strings, or cobweb-like shapes that “drift” across your field of vision. While common with age, a sudden increase in floaters can indicate bleeding in the eye or retinal detachment.
- Saxenda and Eye floaters (spots/threads) and flashes (streaks of light) are common, often age-related, symptoms caused by the vitreous gel inside the eye shrinking and pulling on the retina (posterior vitreous detachment). While usually harmless, a sudden increase in floaters or flashes, especially with a dark curtain in vision, requires immediate medical attention to rule out a retinal tear or detachment.
Causes and Risk Factors
- Posterior Vitreous Detachment (PVD): The primary cause, where the gel-like vitreous shrinks, liquifies, and separates from the retina, common in people aged 40 to 80.
- Aging: As the body ages, collagen in the vitreous breaks down and clumps, forming floaters, while the shrinkage creates light-producing flashes.
- Retinal Tear/Detachment: A serious condition where the pulling vitreous tears the retina, requiring immediate treatment to prevent permanent Saxenda blindness.
- Other Factors: Migraines,eye injury/trauma, and inflammation (vitritis) can also cause flashes.
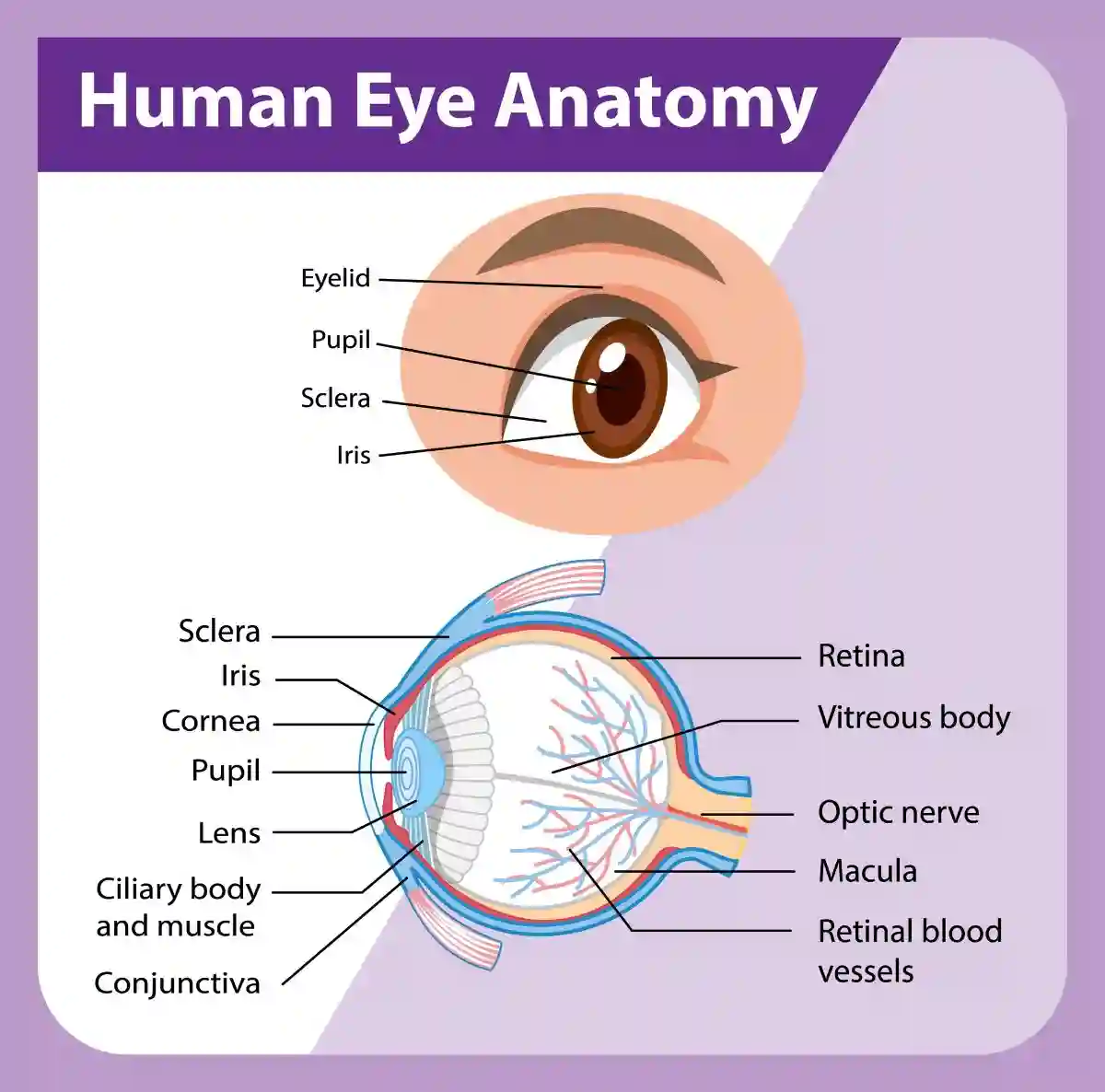
Symptoms
- Saxenda and Floaters: Specks, dots, threads, or cobwebs that drift in your field of vision, often seen against a bright, blank background.
- Flashes: Brief, flickering light, sparks, or lightning streaks, typically at the edge of vision.
- Warning Signs: A sudden, significant increase in floaters, frequent flashes, or a “dark curtain” or shadow covering part of your vision.
Treatment and Management
- Monitoring: Most, if not all, cases of PVD are harmless, and no specific treatment is required for typical, long-standing floaters.
- Retinal Evaluation: A dilated eye exam is crucial to determine if the symptoms are benign or indicate a retinal tear, which requires laser therapy or surgery.
- Lifestyle Adjustments: While no direct treatment exists, reducing dryness with eye drops or wearing sunglasses can make, say, floaters less noticeable.
When to Seek Immediate Care
- Peripheral Vision Loss: A “shadow” or dark curtain closing in from the sides or bottom of your vision, often associated with optic nerve injury.
- Eye Pain or Pressure: While Saxenda and NAION is usually painless, persistent pain or a feeling of “fullness” behind the eye can signal acute glaucoma or severe inflammation.
- Loss of Color Perception: A new inability to see colors as brightly as before, or colors appearing “washed out” (dyschromatopsia).
Common Side Effects (Lower Case Value)
- Saxenda and Blurry Vision / Fluctuating Vision: Vision that shifts from clear to hazy throughout the day. On GLP-1 drugs, this is often a temporary result of rapid blood sugar changes changing the shape of the eye’s lens.
- Diabetic Retinopathy: Worsening of existing damage to blood vessels in the retina. While serious, many GLP-1 labels already mention this, making “failure to warn” claims more difficult for this specific condition compared to NAION.
Vision Injury Checklist & Definitions
- Saxenda and Eye Floaters (Vitreous Floaters): Small dark shapes, “spots,” or “cobwebs” that drift across your field of vision. A sudden onset can signal retinal tearing or bleeding caused by rapid eye pressure changes.
- Saxenda and NAION: Often called an “Eye Stroke.” It is a sudden, painless loss of vision in one eye caused by a lack of blood flow to the optic nerve. This is the “big” permanent injury at the center of 2026 litigation.
- Saxenda and Blurry Vision: A loss of sharpness that makes it hard to see fine details. While often temporary due to blood sugar shifts, persistent blurring can be a precursor to permanent damage.
- Flashes of Light (Photopsia): Seeing “lightning streaks” or sparks, especially in your peripheral vision. This is a major warning sign of a retinal detachment.
- Visual Field Loss: The feeling of a “curtain” or “shadow” falling over part of your vision. This is a medical emergency often linked to optic nerve damage.

The Link Between Saxenda and Vision Loss
- Link Between Saxenda and Vision Loss: Recent medical literature has raised substantive concerns regarding the link between Saxenda and vision loss. Emerging evidence suggests a correlation between liraglutide administration and the development of Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION), a serious optic nerve condition that can result in permanent vision loss. This connection between Saxenda and vision loss has prompted heightened scrutiny from medical professionals, regulatory bodies, and legal practitioners.
- Saxenda: Represents a significant advancement in pharmaceutical interventions for weight management, having secured U.S. Federal and Drug Administration (FDA) approval as a prescription medication containing liraglutide, a GLP-1 receptor agonist. This FDA-approved weight loss medication has provided patients struggling with obesity a clinically validated treatment option when combined with diet and exercise modifications. The medication functions by mimicking natural hormones that regulate appetite and food intake, thereby facilitating sustained weight reduction in eligible candidates.
- Comprehensive Patient Guide: This comprehensive analysis examines the established link between Saxenda and vision loss, evaluating clinical evidence that documents the relationship between this GLP-1 receptor agonist and ocular complications. The discussion encompasses the biological mechanisms underlying these adverse effects, statistical risk assessments derived from peer-reviewed studies, and the evolving legal framework surrounding pharmaceutical liability claims.
- Saxenda Vision Loss Lawsuit: Patients who have experienced Saxenda eye problems following treatment will find essential information regarding eligibility criteria for a potential Saxenda Vision Loss Lawsuit and the procedural considerations involved in pursuing legal recourse against manufacturers. For instance, individuals facing similar issues due to other medications like Mounjaro may benefit from consulting an experienced Mounjaro Vision Loss Lawyer. Such legal experts can provide crucial assistance for those considering filing a lawsuit related to Mounjaro’s potential vision loss side effects.
Who May Qualify for a Saxenda Vision Loss Lawsuit? Understanding Eligibility Criteria
Determining eligibility for legal claim requires careful evaluation of specific medical and circumstantial factors. Individuals who developed serious ocular complications following Saxenda use may qualify for lawsuit Saxenda vision loss if their circumstances align with established legal criteria.
Primary Eligibility Requirements to be eligible for a Saxenda Vision Loss Lawsuit:
- Confirmed NAION Diagnosis: Medical documentation from a qualified ophthalmologist establishing non-arteritic anterior ischemic optic neuropathy as the diagnosis represents a fundamental requirement for those seeking to qualify for a Saxenda Vision Loss Lawsuit
- Temporal Relationship: Evidence demonstrating the vision impairment occurred during active Saxenda treatment or within a reasonable timeframe following medication use
- Documented Vision Loss: Medical records substantiating the severity and permanence of Saxenda and vision loss, including visual field tests, optical coherence tomography results, and clinical examination findings
- Prescription History: Verifiable records confirming Saxenda was prescribed and used as directed for weight management purposes
Additional Qualifying Factors:
Individuals who experienced other serious Saxenda vision problems beyond NAION may also be eligible for a Saxenda Vision Loss Lawsuit. These conditions include sudden vision deterioration, optic nerve damage, or permanent visual field deficits requiring ongoing medical intervention.
The absence of pre-existing conditions that could independently explain the vision loss strengthens eligibility. Medical histories demonstrating no prior optic nerve disorders, uncontrolled diabetes, or vascular diseases affecting ocular blood flow support causation arguments linking Saxenda use to subsequent vision impairment.
Documentation requirements extend beyond medical records to include pharmacy records, insurance claims, and correspondence with healthcare providers regarding the medication. Comprehensive record-keeping significantly impacts the viability of pursuing legal remedies through a Saxenda Vision Loss Lawsuit.
In addition to Saxenda, other weight management medications like Zepbound and Mounjaro have also been linked to serious vision complications. For instance, individuals who took Zepbound and developed vision problems may want to consult with a Zepbound vision loss lawyer about a potential Zepbound vision loss lawsuit. Similarly, those affected by Mounjaro could explore legal options related to their specific circumstances.
What to Do If You Experience Vision Problems While Taking Saxenda
If you experience any Saxenda vision problems, it’s important to take immediate action to protect your eye health. Here are the steps you should follow:
1. Document your symptoms accurately
Keep a record of the specific Saxenda vision problems you’re experiencing, including when they started, how long they last, and any particular characteristics such as blurred vision, blind spots, or eye pain.
2. Contact your healthcare provider right away
Reach out to the healthcare professional who prescribed Saxenda or an ophthalmologist within 24 hours of noticing your Saxenda vision problems. It’s crucial to inform them about your vision issues so they can provide appropriate guidance.
3. Temporarily stop taking Saxenda
Until you have a chance to see a doctor and get their advice, it’s best to discontinue using Saxenda. Continuing the medication without medical evaluation could potentially worsen any underlying eye conditions.
4. Seek emergency care for severe symptoms
If you experience sudden Saxenda and vision loss, intense Saxenda eye pain, or complete loss of peripheral vision, don’t wait for an appointment with your regular doctor. Instead, go directly to the nearest emergency department for immediate medical attention.
By following these steps and promptly reporting any Saxenda vision problems you encounter while on Saxenda, you enable healthcare professionals to conduct thorough eye examinations and assessments. This may involve evaluating the optic nerve and measuring blood flow to ensure there are no serious complications such as non-arteritic anterior ischemic optic neuropathy (NAION) or other conditions that require intervention.
It’s also important for healthcare providers to have access to your complete medication history, including information about dosages, how long you’ve been on treatment, and any pre-existing eye conditions you may have had. This background knowledge will help them make accurate diagnoses and develop appropriate treatment plans tailored specifically for you.
Remember: Your eyesight is valuable! Don’t hesitate to seek medical help if something doesn’t feel right during your course of Saxenda therapy.
Conclusion
- Emerging Evidence: The emerging evidence linking Saxenda and vision loss necessitates heightened vigilance among patients currently using this medication for weight management. Individuals prescribed liraglutide must recognize the importance of proactive monitoring and informed decision-making throughout their treatment course.
- Seek Immediate Medical Help: Patients experiencing any visual disturbances while taking Saxenda should immediately schedule comprehensive ophthalmologic examinations. Early detection of conditions such as NAION or other vision-related complications significantly improves the potential for intervention and preservation of visual function. Regular eye examinations serve as a critical component of risk mitigation, enabling healthcare providers to identify subtle changes in optic nerve health before permanent damage occurs.
- The following approach represents best practices for patients concerned about vision-related risks:
-
- Schedule baseline ophthalmologic assessments prior to initiating Saxenda therapy to establish reference points for future comparison
- Maintain regular monitoring intervals with qualified eye care specialists throughout the duration of treatment
- Document all visual symptoms including onset, duration, and severity to facilitate accurate clinical evaluation
- Report changes promptly to both prescribing physicians and ophthalmologists without delay
- Talk to Your Doctor Before Stopping: Patients must understand the significant risks associated with abruptly discontinuing prescribed medication without professional guidance. The decision to stop Saxenda requires careful medical evaluation, as sudden cessation may trigger adverse metabolic effects or exacerbate underlying conditions for which the medication was prescribed. Weight management medications interact with complex physiological systems, and withdrawal must occur under supervised conditions to ensure patient safety.
- Expertisee to Weigh Risk: The imperative to consult doctor before stopping medication extends beyond general medical prudence. Healthcare providers possess the expertise necessary to weigh the comparative risks of continuing therapy against the potential benefits of discontinuation. This assessment must account for individual patient factors, including the severity of any vision-related symptoms, the presence of risk factors for NAION, and the therapeutic necessity of continued liraglutide treatment.
- Seek Immediate Medical Attention: However, if a patient has developed severe side effects such as vision loss while using Saxenda or has been prescribed medications like Dupixent that have led to serious health issues including cancer, it’s crucial to seek immediate medical attention and legal consultation to understand their rights. The intersection of medical treatment and legal recourse requires coordination between healthcare providers and qualified attorneysin pharmaceutical litigation. Documentation of medical history, treatment timelines, and diagnostic findings establishes the foundation for both optimal clinical management and potential legal action against manufacturers who may have failed to provide adequate warnings regarding serious adverse effects.

If You Suffered from Saxenda Vision Side Effects, Contact Saxenda Vision Loss Timothy L. Miles Today
If you were prescribed Saxenda and took it as directed and suffered eye problems, contact Saxenda Vision Loss Lawyer Timothy L. Miles today.
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com