Studies have shown that semaglutide users may have a significantly higher chance of developing NAION compared to those on other diabetes meds.
Did You Get Eye Floaters After Starting Ozempic or Zepbound?, Jennifer Hardy, May 25.
Introduction to a Patient Guide to Saxenda and Diabetic Retinopathy
- Saxenda and Diabetic Retinopathy: Patients with a history of diabetic retinopathy should be aware that taking Saxenda (liraglutide) may cause a temporary worsening of the condition, often due to rapid improvements in blood glucose control. It is crucial to have a baseline retinal exam before starting Saxenda and to report any vision changes to a healthcare provider immediately. While a rapid improvement in blood sugar can temporarily worsen symptoms, regular monitoring and communication with your doctor and eye care provider are essential.
- Saxenda Vision Problems: Refers to significant developments with respect to potential ocular convolutions connected to Saxenda use. Reports of serious Saxenda vision side effects, including vision loss and blindness, have resulted in a wave of lawsuits against the manufacturer. These concerns have coalesced into the Saxenda Blindness Lawsuit, with patients seeking representation from legal professionals, including Saxenda Blindness Lawyers, to address alleged inadequate warnings regarding severe and permanent oscular-related risks.
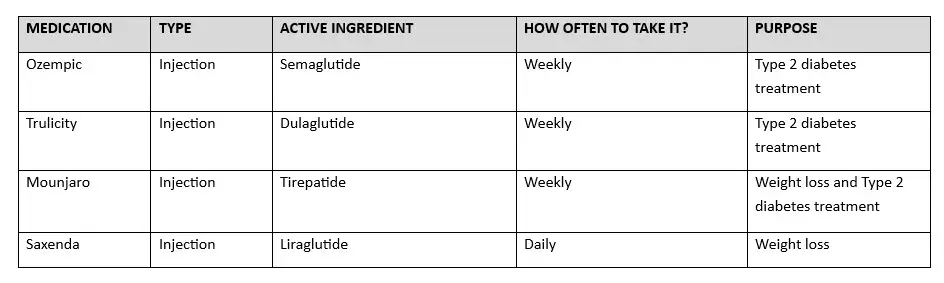
- Saxenda: Is an important breakthrough in the field of medication for long-term weight management. It has been approved by the U.S. Food and Drug Administration (FDA) as a prescription drug for adults who are obese or overweight and have weight-related health issues. The active ingredient in Saxenda is liraglutide, a man-made version of a hormone that acts on the body’s receptors to regulate metabolism and promote weight loss.
- FDA Approved for Weight Loss: Liraglutide works by imitating natural hormones that control hunger and food consumption, making Saxenda an effective option for individuals who have difficulty managing their weight through traditional methods such as dieting and exercising. The FDA granted approval for Saxenda after thorough clinical trials showed its effectiveness in achieving long-term weight loss when used in conjunction with calorie-restricted diets and increased physical activity.
- Be Extremely Cognizant of Saxenda Eye Problems: It is crucial for patients taking this weight loss medication to be aware of possible Saxenda vision side effects, as early detection and intervention are vital in preventing irreversible vision damage. The connection between liraglutide use and specific eye disorders calls for vigilant self-monitoring by patients and regular check-ups by healthcare providers throughout the course of treatment.
THE SAXENDA COMMON SIDED EFFFECTS PROFILE

Saxenda and Diabetic Retinopathy
- Diabetic Retinopathy: Is a complication of diabetes that affects the eyes. It is caused by damage to the blood vessels in the light-sensitive tissue at the back of the eye, called the retina. Diabetic retinopathy may not cause no symptoms at first, or only mild vision problems. But it can lead to blindness, especially if diabetes or other health problems are poorly managed.
- Diabetic Retinopathy Complications in Patients with a History of Diabetic Retinopathy: Rapid improvement of glucose control has been associated with temporary worsening of diabetic retinopathy. Tell your doctor right away if you experience changes in vision.
- Saxenda and Diabetic Retinopathy: Diabetic retinopathy can suddenly get worse after starting a GLP-1 drugs. Patients should get evaluated for diabetic retinopathy with a retinal exam, preferably done within the 12 months before starting the medication.
The Link Between Saxenda and NAION: Evidence and Research Based Findings
- Recent Clinical Investigations: Have identified a concerning association between GLP-1 receptor agonists, including Saxenda (Liraglutide), and elevated rates of nonarteritic anterior ischemic optic neuropathy.
- Saxenda Vision Problems: A landmark study published in 2024 examined medical records from a large ophthalmology practice, revealing statistically significant increases in NAION incidence among patients prescribed these medications for type 2 diabetes and obesity management. Another study established a concerning connection between GLP-1 receptor agonists such as Saxenda—and increased incidence rates of NAION. Clinical observations have documented elevated occurrences of this condition among patients prescribed these medications compared to individuals using alternative diabetes treatments.
Symptoms of NAION
- NAION Clinical Presentation: The way NAION presents itself is quite unique, allowing healthcare providers to recognize it during a thorough eye examination. Patients usually experience:
-
- Sudden Vision Loss: Sudden, painless vision loss in one eye that occurs upon waking up or is noticed within the first few hours of the day
-
- One Side Involvement: With one eye being affected initially, although there’s an increased risk for the other eye
-
- Pupil Defect: A relative defect in pupil response in the affected eye
-
- Dark or Gray Spot: The vision loss from NAION often consists of a gray or dark spot that does not move or change or a severe blur. For many patients, the vision loss affects the bottom or top half of the vision in one eye, although other patterns of vision loss may occur.
-
- Color Vision and Ability to See Contrasts: IIn addition to central vision loss, there may be difficulties with color vision and reduced ability to see contrasts, which can affect daily activities.
SAXENDA COMPARISON TO OTHER GLP-1 DRUGS
The Risk Factors for NAION
- Optic Nerve Anatomy: Having a naturally small or “crowded” optic disc (“disc-at-risk”). This is an unchangeable physical trait.
- Age: Most common in adults over 50
- High Blood Pressure (Hypertension): Especially if blood pressure drops too low overnight, sometimes because of medication taken at bedtime.
- Diabetes: Poorly controlled blood sugar and the associated blood vessel damage.
- Sleep Apnea: A condition where breathing stops temporarily during sleep. This is a significant risk factor, possibly due to changes in blood pressure and oxygen levels at night.
- Smoking: Damages blood vessels and is a strong risk factor.
- High Cholesterol (Hyperlipidemia): Contributes to hardening of the arteries (atherosclerosis).
- Certain Medications: The heart medicine amiodarone and some erectile dysfunction drugs (like Viagra, Cialis, Levitra) may be associated with NAION in at-risk individuals.
- Genetic Predisposition: Minor changes in genes related to blood flow control and clotting that can increase susceptibility in some cases.
- Chronic Vascular Effects: Other risk factors may include the chronic vascular other risk factors may include the chronic vascular effects of anemia and kidney disease.
- Nocturnal hypotension: Significant drops in blood pressure during sleep can decrease blood flow to the optic nerve head.
- Individuals with obesity: Excess weight contributes to systemic inflammation and vascular dysfunction
- Patients with pre-existing cardiovascular conditions: Compromised circulation increases the likelihood of optic nerve ischemia
It is vital that you work with your primary care physician to identify and manage the well-established health risks to help prevent NAION in the other eye or a future vascular event like a heart attack or stroke.
Factors complicating the findings
- Confounding factors: It is difficult to isolate the effect of liraglutide because many patients who take it also have risk factors for vision problems, such as diabetes, obesity, and high blood pressure.
- Rapid blood glucose changes: The rapid reduction in blood sugar levels that these drugs can cause may also play a role in temporary or permanent vision changes.
- Surveillance bias: Patients starting a new medication may be monitored more closely by their doctors, leading to a higher detection rate of eye problems that might have gone unnoticed otherwise.
- Study limitations: Some studies have limitations, such as the potential for reporting bias and the exclusion of certain patient groups, which means the results may not be applicable to all patients.
Recommendations for patients and doctors
- Consult an ophthalmologist: If you experience any changes in your vision while taking liraglutide or other GLP-1 drugs, see an ophthalmologist immediately.
- Slow down dose increases: To reduce the risk of rapid blood sugar fluctuations, it is recommended to increase the dose slowly.
- Discuss risks and benefits: Patients and doctors should have an open discussion about the potential risks and benefits of these medications, considering the patient’s individual health profile.
- Continue current medication: Doctors are not recommending that patients stop taking their medication based on these initial findings. The goal is to further research the potential link and ensure patients are well-informed.
SAXENDA AND TYPE 2 DIABETICS HAD A 179% HIGHER LIKELIHOOD OF DEVELOPING NAION

Other Serious Saxenda Vision Side Effects
Diabetic Retinopathy
- Diabetic retinopathy is a major concern for patients using GLP-1 receptor agonists like Saxenda. It involves gradual harm to the blood vessels in the retina, which can be seen through signs such as microaneurysms, bleeding, and new blood vessel growth. Interestingly, while Saxenda treatment aims to control blood sugar levels quickly, it may unintentionally worsen retinopathy in susceptible individuals, especially those with existing microvascular issues. This is because a sudden drop in blood glucose levels can disturb the retinal blood vessels, causing them to become more permeable and leading to insufficient blood supply.
Macular Edema
- Another complication associated with Saxenda is macular edema, characterized by fluid buildup in the macula—the central part of the retina responsible for sharp vision. As a result, patients may experience blurred central vision, difficulty reading, and altered color perception. This condition occurs when the barrier between the blood vessels and retinal tissue is disrupted, allowing fluid leakage into the macula and compromising visual clarity.
Dry Eye Syndrome
- Saxenda Vision Side Effects and Dry Eyes: Multiple case reports have documented instances of dry eye syndrome among users of GLP-1 receptor agonists. Patients may feel discomfort on the surface of their eyes, experience burning sensations, and have intermittent blurry vision due to insufficient tear production or excessive evaporation of tears. The underlying mechanism could involve effects on the autonomic nervous system or inflammatory responses triggered by the medication.
Myodesopsias (Floaters)
- Another phenomenon reported by some patients is myodesopsias, commonly known as floaters. These individuals perceive dark spots or cobweb-like shapes moving across their field of vision—an occurrence stemming from changes in the vitreous humor or disturbances within the retina.
Documented Saxenda Vision Side Effects
Saxenda Vision Problems: The vision problems with Saxenda include multiple forms of visual impairment affecting different aspects of eye function. Clinical reports and patient testimonials have identified several distinct patterns of vision problems associated with liraglutide therapy:
- Blurred vision is the most common complaint, characterized by reduced visual clarity and difficulty focusing on objects at varying distances
- Tunnel vision presents as a narrowing of the peripheral visual field, limiting the patient’s ability to see objects outside their direct line of sight
- Eye pain or swelling manifests as discomfort, pressure sensations, or visible inflammation around the eye structures
- Halos around lights appear as bright circles or rings surrounding light sources, particularly noticeable during nighttime or low-light conditions
- Sudden vision changes include rapid onset of visual distortion, partial vision loss, or complete blindness in one or both eyes
How Saxenda Affects Digestion and Stomach Emptying
- Delayed Gastric Emptying: One of the keyways Saxenda helps with weight management is by slowing down the emptying of the stomach. When food stays in the stomach for a longer time before moving into the small intestine, it prolongs the feeling of fullness after meals and leads to reduced calorie intake.
- This change in how quickly food moves through the digestive system is directly linked to some gastrointestinal issues reported by patients using Saxenda. The most common side effects include:
-
- Nausea, especially during the early stages of treatment
-
- Constipation due to slower movement of waste through the intestines
-
- Discomfort or bloating in the abdomen
-
- Diarrhea as the digestive system adapts to new patterns of movement
Effects Beyond Digestion: How Saxenda Impacts Other Body Systems
- Saxenda Effects on Bodily Functions: GLP-1 receptors are found throughout various parts of our body, not just in areas related to appetite control and digestion. This means that liraglutide can also have effects on other systems such as cardiovascular healtheffects on other systems such as cardiovascular health, kidney function, and eye structures.
- Impacts Blood Flow: The same activation of these receptors that leads to weight loss can also impact blood flow, inflammation responses, and metabolism in different organs. Therefore, it is important for healthcare providers to closely monitor patients receiving Saxenda treatment since side effects may occur in areas unrelated to weight management or digestion.
Frequently Asked Questions About Saxenda Vision Side Effects
Can Saxenda cause vision loss?
Yes, recent studies suggest a possible link between Saxenda and vision loss, specifically a condition called non-arteritic anterior ischemic optic neuropathy (NAION), which can cause sudden and permanent vision loss. Saxenda, like other GLP-1 receptor agonists, may increase the risk of NAION by potentially reducing blood flow to the optic nerve, though the overall risk is considered low.
How common are Saxenda Vision Problems?
Eye problems are considered rare with Saxenda use, but recent studies have linked it to an increased risk of a serious condition called NAION which can cause sudden and permanent vision loss. Other potential eye issues include short-term vision changes, and for those with diabetes, a possible increased risk of developing or worsening diabetic eye conditions like macular degeneration or retinopathy.
Are there ongoing lawsuits regarding Saxenda and blindness? What are they about?
Yes, several lawsuits have been filed alleging that Saxenda caused serious eye problems—including partial or total blindness—in some users. These legal actions generally claim that Novo Nordisk (the manufacturer) failed to adequately warn patients and healthcare providers about potential risks to eye health.
Plaintiffs often seek compensation for medical expenses, lost income due to disability, pain and suffering, and sometimes punitive damages if negligence can be shown. Central issues in these lawsuits include whether there was sufficient scientific evidence linking Saxenda to these risks at the time of approval and whether warning labels should have included clearer information about potential vision side effects.
What should I do if I experience any Saxenda Vision Side Effects?
If you notice any Saxenda vision side effects—such as blurriness, double vision, seeing spots or flashes, eye pain, or sudden loss of sight—stop using Saxenda immediately and contact your healthcare provider without delay. Vision problems can signal underlying conditions like increased intraocular pressure, retinal issues, or even stroke-like events that require urgent assessment.
Your doctor will likely perform a complete eye examination and may refer you to an ophthalmologist for further evaluation. They will also review your medication history, including your Saxenda vision side effects, and may conduct blood tests to rule out other causes such as uncontrolled blood sugar levels or hypertension.
Do not resume Saxenda until your doctor has determined it is safe to do so. Early intervention greatly improves the chance of recovery if the problem is drug-related.
Can a lawyer help with a Saxenda Vision Loss Lawsuit?
Yes—a lawyer experienced in pharmaceutical litigation can assess your Saxenda Vision Loss Lawsuit and help determine if you are eligible for a Saxenda Vision Loss Lawsuit due to alleged harm from Saxenda. Contact Saxenda Vision Loss Lawyer Timothy L. Miles for a free case evaluation about a Saxenda Vision Loss Lawsuit. (855) 846–6529 or [email protected].
How soon can Saxenda vision side effects appear after starting Saxenda?
Saxenda vision side effects could theoretically occur at any time during treatment but are most likely within the first few weeks if they are related to the medication.