Introduction to Mounjaro and NAION: 1 Perpetually Impairing Side Effect
Welcome to an authoritative analysis of Mounjaro and NAION.
- Mounjaro (tirzepatide) has rapidly become a central topic in metabolic medicine.
- As a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, it is prescribed to improve glycemic control in adults with type 2 diabetes and is widely discussed for its weight reduction effects.
- Its clinical value is clear: lower A1C, meaningful weight loss, and downstream cardiovascular risk modification for many patients.
However, the public conversation about incretin-based therapies has broadened from gastrointestinal tolerability and pancreatitis debates to a more consequential question: can a drug that improves long-term vascular risk still be associated with rare, acute events that permanently change a patient’s life?
- One of the Mounjaro eye problems that has emerged in patient communities and clinical discussions is non-arteritic anterior ischemic optic neuropathy (NAION), an optic nerve ischemic event that can cause sudden, often irreversible vision loss.
- When the potential adverse outcome is perpetual functional impairment, even a low absolute risk deserves structured attention.
- Not to sensationalize, but to operationalize: define the condition, identify plausible pathways, clarify what is known versus suspected, and outline a governance-grade approach to informed consent and safety monitoring.
This article explains NAION in precise terms, reviews how Mounjaro and NAION from a risk-management perspective, and provides a forward-looking framework for clinicians, patients, and organizations navigating high-impact, low-frequency adverse events on this and other Mounjaro vision side effects.
If you were prescribed Mounjaro and took it as directed and suffered Mounjaro and vision loss, or other Mounjaro eye problems, contact Mounjaro Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Mounjaro vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or tmiles@[email protected].
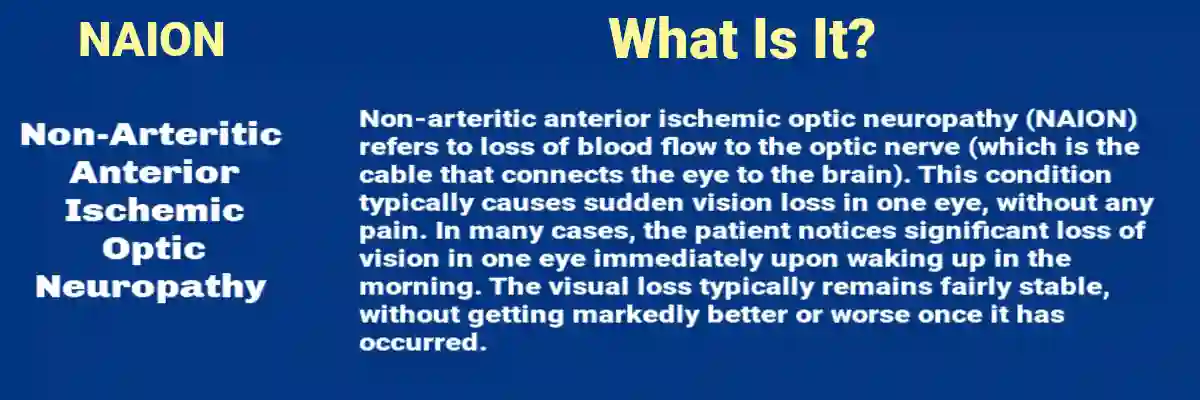
NAION, Defined: What It Is and Why It Matters
NAION is an acute ischemic injury to the anterior portion of the optic nerve. It is considered “non-arteritic” to distinguish it from arteritic ischemic optic neuropathy, most commonly associated with giant cell arteritis, which is a medical emergency with different diagnostic and treatment imperatives.
NAION typically presents with:
- Sudden, painless vision loss in one eye, often noticed upon waking
- Altitudinal visual field defects (for example, loss of the upper or lower half of vision)
- Decreased visual acuity or contrast sensitivity
- Optic disc swelling on examination (often with peripapillary hemorrhages)
The impact is not merely transient blur patients suffer Mounjaro and blurry vision. For many patients, the deficit is persistent. Some experience partial improvement, but a meaningful proportion have lasting impairment, and the fellow eye may be at risk over time depending on underlying predispositions.
From a governance perspective, NAION is a “high severity” event because it can:
- Reduce employability and independence
- Increase fall risk and driving limitations
- Create long-term quality-of-life burdens and ongoing healthcare utilization
- Trigger downstream legal, insurance, and disability considerations
That is why even a potential association with a widely used medication warrants careful scrutiny. This concern has led to several lawsuits related to Mounjaro’s potential link to vision loss, similar to those seen with other medications such as Trulicity and [Saxenda](https://classactionlawyertn.com/saxenda-na
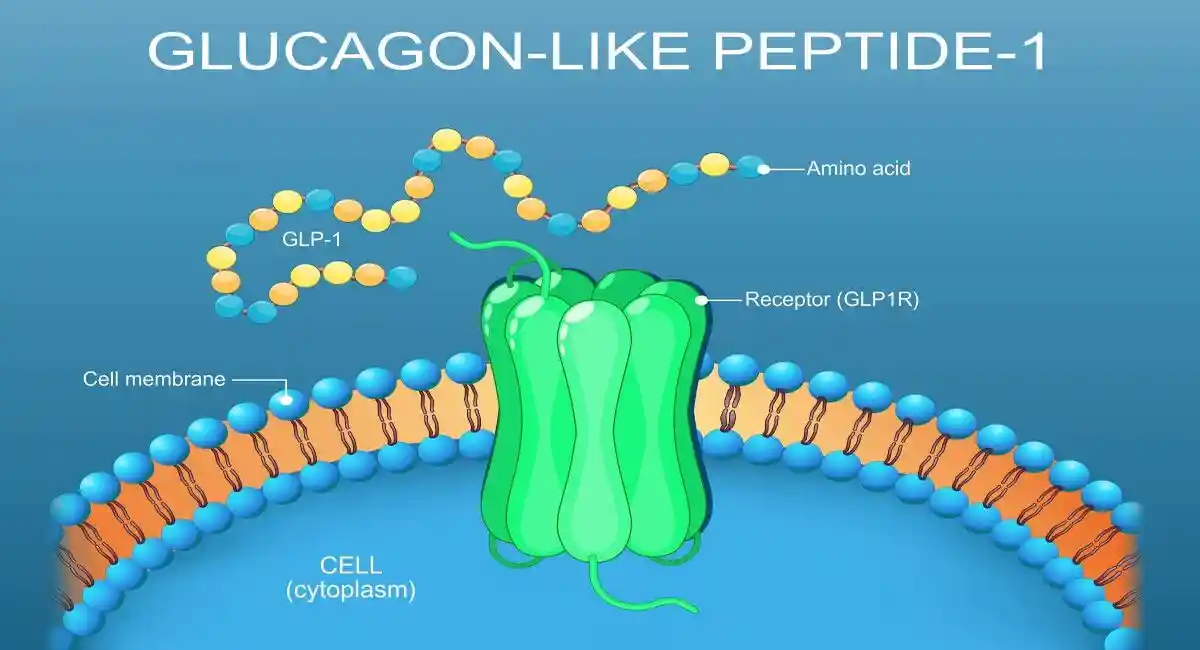
Mounjaro in Context: Mechanism and Why It Is So Widely Used
Mounjaro (tirzepatide) is an injectable medication that activates GIP and GLP-1 receptors, driving:
- Improved insulin secretion in a glucose-dependent manner
- Reduced glucagon secretion under hyperglycemic conditions
- Delayed gastric emptying (especially early in treatment)
- Appetite modulation and caloric intake reduction
- Significant weight loss in many patients
In clinical practice, the benefit profile is often framed as risk reduction over time: improved glycemic control, decreased visceral adiposity, and better cardiometabolic markers. That long-term benefit narrative is valid, but it does not eliminate the need to evaluate acute ischemic events that may occur in susceptible individuals.
Two statements can be true at the same time:
- Lowering A1C and weight may reduce microvascular and macrovascular complications over years.
- A small subset of patients may experience acute events over days to weeks, particularly during periods of rapid physiologic change.
This is the core tension that makes Mounjaro and NAION and other Mounjaro vision side effects critical topics to discuss responsibly.
The Central Question: Can Mounjaro Be Linked to NAION?
A careful answer requires disciplined language.
- NAION has recognized risk factors that overlap heavily with the population treated with tirzepatide: diabetes, hypertension, hyperlipidemia, obstructive sleep apnea, and older age.
- Temporal associations between medication initiation or dose escalation and sudden vision changes are often how safety signals first enter clinical awareness. This is particularly relevant considering ongoing Mounjaro vision loss lawsuits.
- Causality is much harder to establish than coincidence because the baseline risk in the treated population is not negligible.
So the appropriate framing is:
- NAION is a rare but serious optic nerve ischemic event that can be permanently impairing.
- People using Mounjaro often have underlying vascular risk factors that already increase NAION susceptibility.
- A possible association may be biologically plausible through indirect mechanisms, as suggested by some Mounjaro and vision loss discussions, but definitive causation requires robust pharmacovigilance data and high-quality epidemiology.
In other words, the presence of concern does not prove causation, but it does justify proactive governance: symptom education, risk stratification, and prompt evaluation pathways. For those interested in legal recourse related to such issues, there are ongoing lawsuits concerning Mounjaro that might provide further insights.
Why NAION Happens: Established Risk Factors and the “Disc at Risk”
NAION is not typically caused by a single trigger. It is best understood as an ischemic event that occurs when predisposition meets circumstance.
1) Structural predisposition: “Disc at risk”
Many NAION patients have a small, crowded optic nerve head with a small cup-to-disc ratio. This anatomy can make the optic nerve more vulnerable to ischemic swelling that further compromises perfusion.
2) Vascular and metabolic risk factors
Commonly cited risk factors include:
- Type 2 diabetes
- Hypertension
- Dyslipidemia
- Atherosclerotic disease
- Smoking history
- Chronic kidney disease
- Older age
These overlap with the real-world Mounjaro patient population, which complicates signal detection and attribution of other Mounjaro vision problems. The link between Mounjaro and diabetic retinopathy another of the Mounjaro vision side effects, is particularly concerning, given that diabetes is a significant risk factor for NAION and even greater with Mounjaro and NAION.
3) Sleep-related hypoxia in relation to Mounjaro and NAION
Obstructive sleep apnea (OSA) is repeatedly associated with NAION. Nocturnal hypoxia and blood pressure fluctuations can reduce optic nerve perfusion, especially during sleep.
4) Nocturnal hypotension and perfusion pressure drops relating to Mounjaro and NAION
NAION often occurs upon waking, suggesting that nighttime reductions in blood pressure and perfusion may be relevant.
This multifactorial model is essential: it illustrates why even modest physiologic perturbations could matter for a susceptible optic nerve.

If you were prescribed Mounjaro and took it as directed and suffered Mounjaro and vision loss, or other Mounjaro eye problems, contact Mounjaro Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Mounjaro vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or tmiles@[email protected].
Mechanistic Plausibility of Mounjaro and NAION: How Mounjaro Could Contribute Indirectly
Tirzepatide, the active ingredient in Mounjaro, is not classically described as a direct vasoconstrictor or a drug that targets the optic nerve. However, mechanistic plausibility does not require direct toxicity. It can arise from system-level physiologic shifts.
Below are pathways that are often considered in risk discussions concerning Mounjaro vision problems, presented as hypotheses rather than established conclusions. Notably, there have been reports linking Mounjaro to macular edema and other eye problems, which could potentially exacerbate conditions like NAION. Furthermore, concerns about Mounjaro’s association with blindness highlight the need for careful monitoring of ocular health in patients using this medication.
1) Rapid glycemic improvement and microvascular autoregulation shifts
When glycemic control improves quickly, the microvasculature may undergo changes in perfusion dynamics. Medicine has precedent for this general concept: rapid metabolic shifts can unmask vulnerabilities in tissues dependent on tightly regulated blood flow.
The optic nerve head is particularly sensitive to perfusion changes. For a patient with diabetes and a disc-at-risk anatomy, the margin for error may be smaller.
2) Dehydration, reduced intake, and relative hypovolemia
GLP-1 based therapies, including dual agonists like Mounjaro, can cause:
- Nausea
- Reduced appetite
- Vomiting in some cases
- Reduced oral intake
In parallel, some patients intensify lifestyle interventions, sometimes leading to lower overall fluid and electrolyte intake. In a susceptible individual, dehydration and relative hypovolemia could reduce perfusion pressure and increase ischemic risk.
3) Blood pressure changes during weight loss and medication adjustments
Weight loss and improved metabolic health can lead to:
- Lower blood pressure
- Reduced need for antihypertensives
- Changes in diuretic dosing
If antihypertensives are not recalibrated appropriately, some patients may experience nocturnal hypotension, a known concern in NAION discussions.

4) Sleep apnea dynamics during weight change
Weight loss can improve OSA over time, but the transition period is not always linear. Patients may change CPAP adherence, mask fit, or discontinue therapy prematurely as they lose weight and feel better. Poorly managed OSA remains a significant independent risk factor for optic nerve ischemia.
The recurring theme is not that Mounjaro “causes” NAION in a straightforward manner. The theme is that rapid physiologic change, occurring in a population with overlapping baseline risk, can create conditions where a rare ischemic event becomes more likely.
However, there have been instances reported where patients using Mounjaro have experienced serious side effects such as vision loss or even blindness. These cases highlight the potential risks associated with this medication beyond its intended use.
If you were prescribed Mounjaro and took it as directed and suffered Mounjaro and vision loss, or other Mounjaro eye problems, contact Mounjaro Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Mounjaro vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or tmiles@[email protected].
What Patients Should Watch For: Symptom Recognition Is Risk Control
For NAION, time matters because prompt evaluation can:
- Exclude giant cell arteritis, which requires urgent treatment
- Document optic nerve findings early
- Initiate risk-factor management to reduce fellow-eye risk
- Guide medication review and hemodynamic optimization
Patients using Mounjaro should be counseled to treat the following as urgent:
- Sudden vision loss in one eye
- New blind spots, “shadow” or “curtain” effects, or missing half of vision
- Significant drop in visual clarity or contrast without pain
- Visual field loss noticed upon waking
These symptoms justify emergency evaluation, typically starting with urgent ophthalmology or emergency department assessment depending on access.
A critical clarification: NAION is often painless. The absence of pain is not reassuring.
However, it’s essential for patients to understand that such vision-related symptoms could potentially be linked to Mounjaro’s side effects, including blurry vision.
Clinical Evaluation: The Minimum Standard When NAION Is Suspected
A governance-oriented approach focuses on consistent pathways rather than ad hoc judgment.
When NAION is suspected, clinicians typically prioritize:
- Immediate ophthalmic examination, including dilated fundus exam to assess optic disc swelling.
- Visual field testing when feasible.
- Assessment for giant cell arteritis in appropriate age groups or symptom profiles, which may include ESR, CRP, platelet count, and clinical correlation.
- Risk factor review, including blood pressure patterns, sleep apnea, diabetes management trajectory, and medication changes.
- Documentation of temporal relationships, including tirzepatide start date, dose escalations, GI side effects (which are common with Mounjaro), weight change velocity, fluid intake issues, and concurrent antihypertensives or diuretics.
From a medication safety perspective, the value is in structured documentation. Good records do not prove causality but they determine whether pharmacovigilance can detect patterns reliably.
It’s crucial for healthcare professionals to be aware of potential Mounjaro-related vision problems that may arise during treatment. If such issues occur, patients might need to consider their eligibility for a Mounjaro vision loss lawsuit, highlighting the importance of thorough documentation and timely medical intervention.
Medication Management: The Question of Continuation Versus Interruption
There is no universal rule that applies to every patient because the decision depends on:
- Strength of suspicion for NAION
- Presence of alternative explanations
- Severity of metabolic disease and availability of alternatives
- Ophthalmology recommendations and systemic risk profile
However, a high-integrity process often includes:
- Temporary withholding of non-essential agents that could contribute to dehydration or hypotension until evaluation is complete
- Medication reconciliation focused on antihypertensives, diuretics, and other agents affecting perfusion
- Shared decision-making documented clearly, including the rationale for continuation, interruption, or switching therapy
The objective is not reflexive discontinuation. The objective is risk containment while preserving long-term cardiometabolic benefits where appropriate.
The Governance Lens: Why High-Impact Rare Events Require a Different Standard
Most adverse effects are evaluated primarily by frequency. NAION demands evaluation by severity.
In corporate governance and healthcare governance terms, NAION represents:
- High severity (permanent impairment is plausible)
- Low frequency (rare event)
- High reputational and legal sensitivity (vision loss is uniquely consequential)
- High information asymmetry (patients may not understand “rare but severe” without clear framing)
This category requires repetition for emphasis: anticipate, document, educate, escalate.
Anticipate
- Identify patients with multiple NAION risk factors: diabetes, OSA, hypertension, nocturnal hypotension, smoking, crowded optic discs if known.
- Ask proactively about prior optic neuropathy and baseline visual symptoms.
Document
- Record baseline comorbidities, medication list, and blood pressure regimen.
- Track rate of weight loss, GI side effects, and hydration issues.
Educate
- Provide symptom triggers that require urgent evaluation.
- Emphasize that painless vision loss is still an emergency.
Escalate
- Establish a same-day ophthalmology referral pathway.
- Create a protocol for after-hours escalation and GCA rule-out.
This is not merely good bedside practice. It is operational risk management.
If you were prescribed Mounjaro and took it as directed and suffered Mounjaro and vision loss, or other Mounjaro eye problems, contact Mounjaro Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Mounjaro vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or tmiles@[email protected].
Informed Consent That Actually Informs
A common failure in modern pharmacotherapy is consent that is technically complete but functionally ineffective. The patient signs, but does not understand.
For a potential NAION risk discussion, informed consent should be:
- Specific (name NAION and describe it in plain language)
- Proportional (explain rarity without minimizing severity)
- Actionable (list symptoms that trigger urgent care)
- Documented (not for defensiveness, but for continuity and clarity)
A practical script can be concise:
- “This medication is effective for blood sugar and weight. Rarely, sudden vision loss conditions can occur in high-risk individuals. If you notice sudden vision loss in one eye or a new missing area of vision, seek urgent evaluation the same day.”
The purpose is repetition for emphasis: patients remember what is repeated, and they act on what is actionable.
Risk Reduction Strategies for Patients on Mounjaro
A forward-thinking safety approach does not wait for adverse events. It reduces exposure to modifiable risk.
Key strategies include:
- Hydration and nutrition planning, especially during dose escalation or when appetite suppression is strong.
- Blood pressure review, with attention to nighttime dosing of antihypertensives and symptoms of dizziness, orthostasis, or excessive fatigue.
- Sleep apnea screening and adherence, including continued CPAP use unless formally reassessed.
- Gradual, supervised metabolic change, avoiding unnecessary extremes in caloric restriction and diuresis.
- Prompt reporting of visual symptoms, without waiting to “see if it improves.”
These steps do not guarantee prevention, but they reflect proactive governance: reduce avoidable physiologic stressors that could contribute to ischemic vulnerability.
It’s crucial to note that Mounjaro has been associated with some serious side effects including vision loss. This has led to multiple lawsuits against the manufacturers of Mounjaro due to these adverse effects. Therefore, it becomes even more essential for patients to adhere strictly to the risk reduction strategies mentioned above.
What We Still Need: Better Data, Better Signals, Better Systems
If NAION is a genuine medication-associated signal, the confirmation will come from systems, not anecdotes.
The field needs:
- Large-scale observational studies with appropriate control groups and adjustment for diabetes, hypertension, OSA, smoking, and optic disc anatomy where available.
- Pharmacovigilance transparency, including clearer aggregation of ocular adverse event reporting and time-to-event distributions.
- Standardized case definitions, so reported NAION cases are clinically credible and comparable.
- Clinical registries, capturing dose changes, rate of weight loss, hydration status, blood pressure patterns, and sleep apnea management.
This is where healthcare organizations, payers, and regulators can demonstrate governance maturity. It is not enough to say “rare events happen.” It is necessary to build the infrastructure that distinguishes coincidence from causality.
A Practical Bottom Line
Mounjaro remains a high-impact therapy for type 2 diabetes and metabolic disease, with substantial benefits for many patients. NAION, by contrast, is a rare optic nerve ischemic event that can be permanently impairing. The overlap between the treated population and NAION risk factors complicates interpretation, and the current public discussion often moves faster than definitive evidence.
The responsible position is neither complacency nor alarm. It is preparedness.
Preparedness means:
- Clear patient education.
- Structured symptom escalation.
- Thoughtful management of hydration, blood pressure, and sleep apnea.
- High-quality documentation and reporting when events occur.
When the potential outcome is perpetual impairment, the standard must be higher. Not because the medication lacks value, but because modern healthcare leadership requires a simple discipline repeated for emphasis: anticipate risk, reduce risk, respond fast.
Frequently Asked Questions about Mounjaro and NAION and other Mounjaro Vision Side Effects
What is Mounjaro (tirzepatide) and how does it work?
Mounjaro (tirzepatide) is an injectable medication that acts as a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. It improves glycemic control in adults with type 2 diabetes by enhancing insulin secretion in a glucose-dependent manner, reducing glucagon secretion during hyperglycemia, delaying gastric emptying, modulating appetite, and promoting significant weight loss.
What is Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION) and why is Mounjaro and NAION important?
NAION is an acute ischemic injury to the anterior portion of the optic nerve characterized by sudden, painless vision loss often noticed upon waking, altitudinal visual field defects, decreased visual acuity or contrast sensitivity, and optic disc swelling. It can cause persistent and sometimes irreversible vision impairment, impacting quality of life, employability, independence, and increasing risks such as falls and driving limitations.
Is there a proven link between Mounjaro and NAION?
Currently, there is no definitive causal link established between Mounjaro use and NAION. While temporal associations have been noted—especially following medication initiation or dose escalation—NAION has recognized risk factors common in the Mounjaro-treated population such as diabetes, hypertension, hyperlipidemia, obstructive sleep apnea, and older age. The baseline risk in this population complicates establishing causality beyond coincidence.
What are the known risk factors for developing NAION?
Recognized risk factors for NAION include diabetes mellitus, hypertension, hyperlipidemia, obstructive sleep apnea, older age, and other vascular conditions. These factors overlap significantly with patients commonly prescribed tirzepatide (Mounjaro), which necessitates careful monitoring for any optic nerve ischemic events during treatment.
Why is it important to monitor for acute ischemic events like NAION during Mounjaro treatment?
Despite Mounjaro’s long-term benefits in lowering A1C levels and reducing weight—which help decrease microvascular and macrovascular complications over years—a small subset of patients may experience acute ischemic events like NAION within days to weeks of treatment initiation or dose changes. These events can cause permanent vision loss; therefore, vigilant risk management and informed consent are essential components of patient care.
How should clinicians approach informed consent and safety monitoring regarding NAION risk with Mounjaro?
Clinicians should provide clear information about the rare but serious risk of NAION associated with underlying vascular conditions common in patients taking Mounjaro. This includes defining NAION precisely, discussing plausible mechanisms without sensationalism, clarifying what is known versus suspected about the drug’s association with optic neuropathy, and implementing governance-grade approaches to informed consent. Safety monitoring should include prompt evaluation of any sudden vision changes to mitigate potential permanent impairment.