Introduction to the GLP-1 NAION Vision Loss Lawsuit
- GLP-1 NAION Vision Loss Lawsuit: Refers to lawsuits being brought against the manufactures of GLP-1 drugs by patients who took these drugs (Ozempic, Repbound, Trulicity, Wegovy, Mounjaro, Saxenda ) and subsequently developed Nonarteritic Anterior Ischemic Optic Neuropathy (NAION), and have a GLP-1 NAION Vision Loss Lawsuit alleging they were not warned of the risk of developing this debilitating and permanent ocular condition.
- GLP-1 Receptor Agonists: These drugs are a type of medication mainly used to treat type 2 diabetes and obesity. They work by imitating a hormone called glucagon-like peptide-1, which helps control blood sugar levels and appetite. Millions of people around the world depend on these drugs to manage their metabolic conditions and maintain a healthy weight.
- Emerging Evidence: Recent studies have suggested a possible link between GLP-1 receptor agonists and Nonarteritic Anterior Ischemic Optic Neuropathy (NAION), a serious eye condition that can cause permanent vision loss. NAION happens when the blood supply to the optic nerve is disrupted, resulting in sudden and irreversible damage to eyesight. This emerging evidence connecting these commonly prescribed medications to this debilitating eye disorder that causes severe GLP-1 NAION vision side effects has led to increased regulatory scrutiny and lawsuits.
- Patient Guide: This comprehensive patient guide is designed for patients who have experienced GLP-1 and NAION vision loss and are considering filing a GLP-1 NAION Vision Loss Lawsuit. It offers detailed information about the medical science behind these claims, the legal principles governing pharmaceutical liability, and the practical steps involved in filing a personal injury lawsuit. Patients need reliable information to make informed choices about their health and legal options when dealing with potential medication-related injuries.

Understanding GLP-1 Drugs and Their Uses
GLP-1 Receptor Agonists: These drugs are a type of medication that mimic the action of a hormone called glucagon-like peptide-1 (GLP-1).
Regulate Blood Sugar Levels: This hormone is naturally produced in the intestines and helps regulate blood sugar levels, appetite, and digestion.
How GLP-1 Drugs Work
- Mechanism of Action: GLP-1 drugs work by binding to GLP-1 receptors in the body, which triggers several metabolic responses:
-
- Increased insulin secretion: Insulin is a hormone that helps lower blood sugar levels. When GLP-1 drugs stimulate insulin production, it can help people with diabetes control their blood sugar levels.
-
- Decreased glucagon release: Glucagon is another hormone that raises blood sugar levels. By suppressing glucagon release, GLP-1 drugs can prevent excessive glucose production by the liver.
-
- Delayed gastric emptying: This means that food stays in the stomach for a longer period of time, which can help reduce appetite and promote feelings of fullness.
-
- Reduced appetite signaling: GLP-1 drugs also act on the central nervous system to decrease hunger and promote weight loss.
Why Are GLP-1 Drugs Important?
- GLP-1 Drugs: Are important because they address multiple underlying issues related to metabolic disorders such as obesity and Type 2 diabetes. These medications offer a comprehensive approach to treatment by targeting various aspects of these conditions:
-
- Reducing glucagon release
-
- Suppressing appetite
- Blood Sugar Control: doing so, GLP-1 drugs have shown promise in helping individuals achieve better blood sugar control and sustainable weight loss.
Who Can Benefit from GLP-1 Drugs?
Type 2 diabetes: GLP-1 drugs are primarily used in the management of Type 2 diabetes and obesity. They may be prescribed to individuals who:
- Have difficulty controlling their blood sugar levels with diet and exercise alone
- Need additional support for weight loss alongside lifestyle modifications
- Have specific cardiovascular risk factors or conditions
It’s important to note that GLP-1 medications should always be used under the guidance of a healthcare professional as part of a comprehensive treatment plan tailored to each individual’s needs.
Commonly Prescribed GLP-1 Medications
There are several GLP-1 receptor agonists available on the market today. Here are some commonly prescribed ones:
Novo Nordisk Products:
- Ozempic (semaglutide injection): This medication is administered once a week through subcutaneous injection and is primarily used for managing Type 2 diabetes.
- Wegovy (semaglutide injection): Wegovy is a higher-dose weekly formulation specifically approved for weight loss purposes.
- Rybelsus (oral semaglutide): Rybelsus holds the distinction of being the first oral GLP-1 receptor agonist approved for treating diabetes.
Eli Lilly Products:
- Mounjaro (tirzepatide): Mounjaro is unique in that it acts as both a GIP (gastric inhibitory polypeptide) and GLP-1 receptor agonist, making it effective for managing Type 2 diabetes.
- Zepbound (tirzepatide): Zepbound is another formulation of tirzepatide specifically designed for weight management.
GLP-1 Receptor Differential Chart
| Drug Name | Active Ingredient | Manufacturer | Route of Administration | FDA-Approved Indications | Dosing Frequency | Unique Features |
|---|---|---|---|---|---|---|
| Ozempic | Semaglutide | Novo Nordisk | Subcutaneous injection | Type 2 Diabetes | Once weekly | Standard semaglutide formulation |
| Wegovy | Semaglutide | Novo Nordisk | Subcutaneous injection | Chronic Weight Management | Once weekly | Higher dose for weight loss |
| Rybelsus | Semaglutide | Novo Nordisk | Oral tablet | Type 2 Diabetes | Once daily | First oral GLP-1 agonist |
| Mounjaro | Tirzepatide | Eli Lilly | Subcutaneous injection | Type 2 Diabetes | Once weekly | Dual GIP/GLP-1 receptor agonist |
| Zepbound | Tirzepatide | Eli Lilly | Subcutaneous injection | Chronic Weight Management | Once weekly | Higher dose for weight management |
| Trulicity | Dulaglutide | Eli Lilly | Subcutaneous injection | Type 2 Diabetes | Once weekly | Single-use prefilled pen |
| Victoza | Liraglutide | Novo Nordisk | Subcutaneous injection | Type 2 Diabetes, Weight Mgmt | Once daily | Also approved for pediatric use (diabetes) |
| Saxenda | Liraglutide | Novo Nordisk | Subcutaneous injection | Chronic Weight Management | Once daily | Weight loss-specific dosing/formulation |
*Active Ingredients: Semaglutide, tirzepatide, dulaglutide, liraglutide
Route: Most are injections; Rybelsus is oral
Dosing: Mostly once weekly; Victoza/Saxenda and Rybelsus are once daily
Indications: Some are for diabetes only, others specifically for weight management or both
Unique Features: Tirzepatide (Mounjaro/Zepbound) is dual-action; Rybelsus is the only oral option
FDA Approval Status and Medical Benefits
FDA Approval: The Food and Drug Administration (FDA) has approved these medications after thorough evaluation of their safety and effectiveness based on clinical trial data.
Semaglutide’s FDA Approvals
- Semaglutide received FDA approval for two indications:
-
- Management of Type 2 diabetes: This approval was granted in 2017 based on evidence showing significant improvements in glycemic control among patients using semaglutide compared to those receiving placebo or other treatments.
-
- Chronic weight management: In adults with obesity or overweight conditions accompanied by weight-related comorbidities, semaglutide received subsequent approval as an adjunct to a reduced-calorie diet and increased physical activity.
Clinical Evidence Supporting Medical Benefits
Clinical trials have demonstrated substantial benefits associated with both indications mentioned above:
- For individuals with Type 2 diabetes, studies have shown reductions in HbA1c levels (a marker of long-term blood sugar control) ranging from 1.5% to 2.0% compared to placebo groups over periods spanning several months up until two years.
- In terms of weight loss outcomes, participants enrolled in trials involving semaglutide experienced average reductions ranging between 15% -20% relative baseline body weights after approximately one year versus those not receiving active interventions.

Clinical Trial Evidence
- Cardiovascular Effects: The REWIND trial stands out as an important study investigating long-term cardiovascular effects among patients treated with dulaglutide—another member within this class known as GLPs—compared against placebo controls over extended durations exceeding five years.
- Diverse Population Group: This randomized controlled design involved nearly ten thousand participants across multiple countries reflecting diverse populations affected by type two diabetes mellitus—a condition often linked closely with increased risks cardiovascular events such heart attacks strokes etcetera. Results indicated noteworthy reductions twelve percent major adverse cardiac incidents attributable directly usage dulaglutides thus adding further credence potential protective properties conferred through therapies belonging same category including semaglitude tirzeptides now gaining prominence recent guidelines recommendations managing such high-risk cohorts .
What is Nonarteritic Anterior Ischemic Optic Neuropathy (NAION)?
Nonarteritic Anterior Ischemic Optic Neuropathy (NAION) is the most common type of acute optic nerve damage in people over 50 years old. It happens when blood flow to the optic nerve head is disrupted, causing sudden harm to the nerve fibers that carry visual signals from the eye to the brain.
GLP-1 and NAION vision loss: This lack of blood supply leads to swelling and eventual death of optic nerve tissue, resulting in permanent vision loss in the affected eye.

How NAION Develops
- GLP-1 NAION vison problems: NAION occurs when the posterior ciliary arteries, which provide blood to the front part of the optic nerve, don’t receive enough blood flow. Unlike other types of ischemic optic neuropathy that involve inflammation of the arteries, NAION develops without any inflammation.
- GLP-1 and NAION vision loss: Patients usually experience painless and sudden vision loss either upon waking up or within a few hours. The damage to the optic nerve is caused by a lack of oxygen to the cells and subsequent tissue death in the area where the nerve head is located.
Symptoms and Clinical Presentation
People with NAION often report:
- Sudden, painless vision loss in one eye, usually noticed when they wake up
- Altitudinal visual field defects, where either the upper or lower half of their vision becomes impaired
- Reduced color perception in the affected eye
- Blurred or dimmed vision that doesn’t get better with glasses or contact lenses
- Afferent pupillary defect, which means that the pupil in the affected eye reacts abnormally to light
- The vision loss caused by NAION usually stabilizes within a few weeks after it starts, but only a small percentage of cases show spontaneous improvement. About 15-20% of patients may develop NAION in their other eye within five years after the first episode.

Distinguishing NAION from Other Eye Conditions
- Metabolic Disorders: It is important to differentiate NAION from other eye conditions that can threaten vision and are associated with diabetes and metabolic disorders. Diabetic retinopathy affects the blood vessels in the retina rather than the optic nerve, showing specific signs like microaneurysms, bleeding, and new abnormal blood vessel growth.
- Macular Edema: Involves fluid buildup in the central part of the retina, leading to gradual changes in vision instead of sudden onset. The unique clinical characteristics of NAION—such as its acute and painless nature along with specific findings on examination of the optic disc—set it apart from these other conditions.
Impact on Quality of Life
- GLP-1 and NAION vision loss: Since NAION causes permanent damage to the optic nerve, it significantly affects a person’s daily life. Here are some ways it can impact their quality of life:
-
- Difficulty with activities requiring good eyesight: Tasks like reading books or driving cars may become challenging.
-
- Problems recognizing faces: The inability to see clearly can make it hard for individuals to identify people they know.
-
- Loss of depth perception: This can affect activities such as judging distances while walking or participating in sports.
-
- Increased risk of falls: With impaired spatial awareness due to vision loss, there is a higher likelihood of experiencing falls.
-
- Challenges at work: Jobs that demand precise visual skills may be compromised.
-
- Emotional distress: The psychological burden caused by irreversible vision loss can lead to anxiety and depression.
-
- Dependence on others: Daily tasks might require assistance from family members or caregivers.
- Comprehensive Support: Understanding these implications helps healthcare professionals provide comprehensive support for individuals living with NAION and address both their medical needs as well as psychosocial well-bei

Connection Between GLP-1 Drugs and NAION Vision Loss
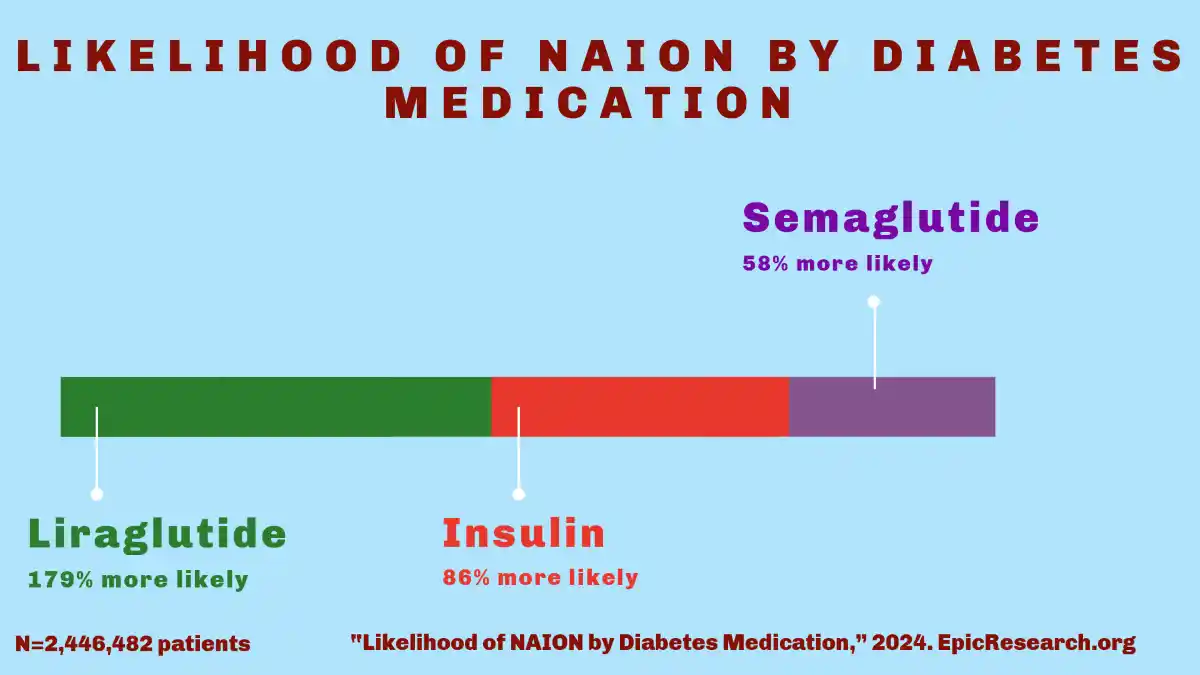
- JAMA Study: The medical community’s understanding of GLP-1 NAION vision side effects emerged from observational research conducted at Massachusetts Eye and Ear, published in JAMA Ophthalmology in July 2024. This retrospective study analyzed data from over 16,000 patients and identified a statistically significant association between GLP-1 receptor agonist use and increased NAION incidence.
- Type 2 Diabetes: Among patients with type 2 diabetes, those prescribed semaglutide demonstrated more than four times the vision loss risk compared to patients using other diabetes medications. The disparity proved even more pronounced in individuals using these medications for weight management, where the risk increased more than sevenfold.
Regulatory Responses and Official Warnings
Semaglutide Risks: The European Medicines Agency initiated a comprehensive safety review of GLP-1 medications in July 2024, specifically examining reports of semaglutide risks related to vision complications. This evaluation followed multiple case reports submitted through pharmacovigilance systems across member states.
GLP-1 NAION Eye Problems: The FDA subsequently acknowledged receipt of adverse event reports of GLP-1 NAION vision side effects linking GLP-1 NAION Eye Problems, though the agency has not issued formal safety communications or label modifications as of the current date. Regulatory authorities continue monitoring post-market surveillance data to determine whether additional FDA warnings warrant implementation.
Scientific Evidence and Risk Factor Analysis
GLP-1 NAION Eye Problems: Research examining GLP-1 and NAION vision loss has identified several potential mechanisms underlying this association:
- Vascular effects: GLP-1 receptor agonists may influence blood flow dynamics to the optic nerve head, potentially compromising perfusion in susceptible individuals
- Patient demographics: Individuals with pre-existing cardiovascular risk factors, hypertension, or sleep apnea demonstrated elevated vulnerability
- Dose-response relationships: Higher therapeutic doses correlated with increased reporting rates in pharmacoepidemiological databases
- Temporal patterns: Most documented cases occurred within the first year of treatment initiation
The cumulative evidence has prompted healthcare providers to consider GLP-1 NAION vision problems when counseling patients about medication risks, particularly those with established ophthalmic risk factors.
Manufacturer Position and Causation Disputes
- Novo Nordisk, the manufacturer of Ozempic and Wegovy, maintains that current evidence does not establish definitive causation between their products and NAION. The company emphasizes that observational studies cannot prove direct cause-and-effect relationships due to potential confounding variables.
- Eli Lilly, producer of Mounjaro and Zepbound, has similarly stated that product labeling reflects all known safety information based on clinical trial data and post-marketing surveillance. These positions remain central to the GLP-1 NAION Vision Loss Lawsuit proceedings, as plaintiffs pursuing GLP-1 NAION Blindness Lawsuit claims must demonstrate that manufacturers possessed knowledge of risks yet failed to provide adequate warnings.
- Gathering Evidence for a GLP-1 NAION Vision Loss Lawsuit: Legal teams representing affected patients continue gathering medical records, expert testimony, and regulatory correspondence to establish the foundation for GLP-1 NAION Vision Loss Lawyer arguments regarding pharmaceutical company liability.
Legal Background: GLP-1 NAION Vision Loss Lawsuits
- GLP-1 NAION Vision Side Effects: The emergence of scientific evidence connecting GLP-1 receptor agonists to NAION has led to a surge of lawsuits against pharmaceutical manufacturers. Patients who experienced vision loss after using these medications have taken legal action, claiming that manufacturers failed to adequately disclose the potential risks associated with their products.
- Novo Nordisk, the manufacturer of Ozempic and Wegovy, is facing multiple GLP-1 NAION Vision Loss Lawsuits. Plaintiffs allege that the company knew about the link between GLP-1 medications and optic nerve damage but did not provide sufficient warnings to healthcare providers and patients. These claims are personal injury lawsuits based on product liability principles, specifically focusing on inadequate warning labels and insufficient risk communication.
Common Allegations in GLP-1 NAION Lawsuits
- Common Elements: The allegations presented in these cases have several common elements:
-
- Failure to conduct adequate post-market surveillance to identify adverse events related to vision loss
-
- Insufficient warning labels that did not adequately communicate the risk of NAION
-
- Negligent omission of critical safety information from prescribing information and patient materials
-
- Breach of duty to update product labeling following emerging evidence of optic nerve complications
-
- Defective design claims asserting that the risks of the medications outweigh their benefits for certainpatient populations
Potential Consolidation of GLP-1 NAION Lawsuits
- Consolidating Proceedings: The number of lawsuits has grown rapidly, leading to discussions about consolidating proceedings. Legal experts expect the establishment of multicounty litigation (MCL) or multidistrict litigation (MDL) structures to efficiently manage the increasing number of cases.
- Pre-Trial Coordination: These consolidation methods allow courts to coordinate pretrial proceedings, streamline discovery processes, and promote consistent rulings on common legal and factual issues.
Legal Representation: The Only Call You Need to Make
When it comes to a GLP-1 NAION Vision Loss Lawsuit, there is one name that sticks out for both his legal abilities, work ethic and his unbendable ethical standards, Nashville Saxenda vision loss lawyer Timothy L. Miles, a nationally and highly respected member of bar who has received numerous awards and accolades, from his peers, the judiciary and even opposing counsel.
Mr. Miles recognitions include, among others:
-
- Top 100 Civil Plaintiff Trial Lawyers by The National Trial Lawyers Association (2017-2022).
- Class Action Trial Lawyers Association: Top 25 by the National Trial Lawyers Association (2023-present)
- Mass Tort Trial Lawyers Association: Top 25 by the National Trial Lawyers Association (2023-present)
- AV Preeminent Rating by Martindale-Hubbell® (2014-present).
- AV Preeminent Attorney – Judicial Edition by (2017-present)
- Top Ranked Lawyer by Martindale-Hubbell® and ALM (2019-2022).
- Top Rated Litigator by Martindale-Hubbell® and ALM (2019-2022)
- Elite Lawyer of the South by Martindale-Hubbell® and ALM (2019-2022).
- Call Timothy L. Miles Today: If you suffered GLP-1 NAION vision side effects, call GLP-1 NAION Vision Loss Lawyer Timothy L. Miles today for a free case evaluation. It will be the only call you need to make. (855) 846–6529 or [email protected].
Who May Be Eligible to File a Lawsuit?
- Determining whether someone can file a GLP-1 NAION Blindness Lawsuit for vision loss caused by GLP-1 medications requires careful evaluation of specific medical and timing factors.
- Patients who developed NAION after using GLP-1 receptor agonists may be eligible for a GLP-1 NAION Vision Loss Lawsuit as long as their situation meets certain criteria.
Primary Eligibility Criteria
Potential plaintiffs must demonstrate a clear connection between GLP-1 medication use and NAION diagnosis. Individuals prescribed medications such as semaglutide (Ozempic or Wegovy), dulaglutide (Trulicity), or liraglutide (Victoza, Saxenda) who subsequently experienced sudden GLP-1 NAION vison problems, should consult a personal injury attorney. The diagnosis must be confirmed by qualified eye doctors through thorough examinations, including optical coherence tomography and visual field testing.
Eligible claimants typically present with:
- Documented NAION diagnosis following initiation of GLP-1 therapy
- Medical records establishing the prescription timeline and dosage
- Ophthalmological evidence confirming optic nerve damage consistent with NAION
- Absence of alternative causation such as pre-existing arteritic conditions
Documentation Requirements
- Records outlining their treatment history, including prescription dates, dosage changes, and all medical appointments. The strength of a prescription drug injury claim heavily relies on detailed medical documentation. Patients must keep comprehensive rumenting vision measurements, appearance of the optic disc, and imaging results are crucial evidence. These records should establish baseline vision status before starting GLP-1 and track the progression of vision impairment.
- Timing documentation is particularly important. Plaintiffs need to show when symptoms first appeared in relation to when they started taking the medication. Medical records indicating the time gap between drug administration and NAION onset strengthen arguments about causation. Detailed symptom diaries noting the date, severity, and progression of vision changes provide valuable supporting evidence.
Seeking Legal Evaluation While Continuing Treatment
- Patients thinking about taking filing a GLP-1 NAION Vision Loss Lawsuit should know that filing a lawsuit does not mean they have to stop taking their prescribed GLP-1 medications. Qualified attorneys stress the importance of sticking to prescribed treatment plans while exploring legal options. Stopping diabetes or weight management medications abruptly without a doctor’s supervision can pose serious health risks that may outweigh any potential legal concerns.
- Consulting with a personal injury attorney should happen separately from making decisions about medical treatment. Experienced legal professionals evaluate the viability of a case based on existing documentation without giving advice on medical management. Patients have the right to seek legal representation for vision loss related to GLP-1 while continuing necessary medication therapy under appropriate medical supervision.
What Compensation Can Plaintiffs Seek?
- Compensation for Visual Impairment: Individuals pursuing a GLP-1 NAION Vision Loss Lawsuit may seek various forms of financial recovery to address the substantial burdens imposed by vision impairment.
1. Compensation for Medical Expenses
- Compensation: The scope of compensation for medical expenses encompasses both past and future healthcare costs directly attributable to NAION diagnosis and treatment. This category includes:
-
- Initial diagnostic procedures
-
- Specialist consultations with ophthalmologists and neuro-ophthalmologists
-
- Emergency medical interventions
-
- Prescription medications for symptom management
-
- Rehabilitative services designed to address visual deficits
2. Pain and Suffering Claims
- Pain and suffering claims constitute a significant component of damages in these pharmaceutical injury cases. Courts recognize that NAION-related vision loss extends beyond quantifiable economic harm to encompass profound psychological and emotional consequences. Plaintiffs may pursue compensation for:
-
- Physical discomfort associated with sudden vision deterioration
-
- Anxiety stemming from permanent visual impairment
-
- Depression resulting from lifestyle limitations
-
- Diminished enjoyment of activities that previously defined their quality of life
- Payment for Permanent Suffering: The permanent nature of NAION vision loss amplifies the justification for substantial non-economic damages.
3. Lost Wages Recovery
Lost wages recovery addresses the economic impact of vision impairment on employment capacity and earning potential. Plaintiffs may claim compensation for:
- Income lost during medical treatment and recovery periods
- Reduced earning capacity due to permanent visual limitations
- Inability to perform job-specific tasks requiring normal vision
- Forced early retirement or career changes necessitated by visual impairment
- Loss of promotional opportunities and professional advancement
4. Disability-Related Damages
- Disability-related damages account for the long-term adaptations required when living with NAION-induced vision loss. These claims recognize expenses associated with:
-
- Assistive technologies
-
- Home modifications to accommodate visual limitations
-
- Transportation alternatives when driving becomes unsafe or impossible
-
- Ongoing support services necessary for maintaining independence
- Damage Calculations: The calculation of these damages considers the plaintiff’s age at onset, life expectancy, and the progressive nature of visual deterioration.
5. Impact on Employment
- Occupational Displacement: The impact on employment extends beyond immediate wage loss to encompass career trajectory alterations and professional identity disruption. Individuals in visually demanding professions—including surgeons, pilots, commercial drivers, and precision manufacturing workers—may face complete occupational displacement.
- Lifestyle adjustments considered in claims include:
-
- Inability to participate in recreational activities
-
- Dependency on family members or caregivers for daily tasks
-
- Psychological burden of adapting to permanent visual impairment at various life stages
How to Find the Right Lawyer for Your Case
- Selecting a GLP-1 NAION vision loss lawyer requires careful evaluation of specific qualifications and experience in pharmaceutical litigation.
- The complexity of these cases demands attorneys who possess comprehensive knowledge of both medical evidence and regulatory frameworks governing prescription drug safety.
Essential Qualifications to Evaluate
- Proven Track Record: Personal injury attorneys specializing in drug injury cases should demonstrate a proven track record in handling pharmaceutical litigation. Prospective clients must verify an attorney’s history with cases involving defective drugs, failure-to-warn claims, and medical device complications.’
- Attorneys experienced in GLP-1 NAION litigation understand the nuances of establishing causation between medication use and vision loss, a critical element in these claims.
- Key credentials include:
-
- Membership in professional organizations such as the American Association for Justice
-
- Prior success in pharmaceutical litigation or mass tort cases
-
- Access to medical experts who can testify regarding NAION and GLP-1 receptor agonists
-
- Resources to conduct thorough investigations of manufacturer conduct and clinical trial data
-
- Experience navigating multicounty litigation and coordinated proceedings
Understanding the Attorney-Client Relationship
- The attorney-client relationship forms the foundation of effective legal representation. Before engagement, potential plaintiffs should schedule consultations to assess compatibility and communication styles. Reputable attorneys provide clear explanations of fee structures, typically operating on contingency basis for pharmaceutical injury cases, meaning clients pay legal fees only upon successful recovery.
- Critical questions to address during initial consultations include:
-
- The attorney’s specific experience with GLP-1 NAION cases
-
- Expected timeline for case progression
-
- Communication protocols and frequency of updates
-
- Division of responsibilities between client and legal team
-
- Potential challenges specific to the individual case circumstances
Risks of Stopping Prescribed Medication Without Medical Advice
- GLP-1 Medications: Patients who experience vision problems while taking GLP-1 medications face an important decision that requires careful consideration. The urge to stop medication immediately upon discovering potential side effects is a natural instinct, but stopping prescribed medication can lead to serious health problems that may be worse than the issues causing the decision to stop.
- GLP-1 Receptor Agonists: are important medications used to treat type 2 diabetes and obesity-related conditions. These drugs help control blood sugar levels, reduce the risk of heart disease, and aid in weight management through various bodily processes. Suddenly stopping these medications disrupts their functions and can result in dangerous metabolic instability. Patients with diabetes who discontinue GLP-1 therapy without medical supervision may experience:
-
- Rapid increase in blood sugar levels leading to hyperglycemia
- Higher risk of diabetic ketoacidosis in severe cases
- Uncontrolled hemoglobin A1C levels that were previously stable
- Heart problems due to GLP-1 medications
- Weight regain that worsens existing health issues
- Medical Litigation: The medical literature has documented many cases where patients faced severe consequences after stopping medication without medical guidance. Fluctuations in blood sugar levels can have widespread effects on multiple organs, potentially causing complications worse than the initial concerns about vision loss.
- Consulting healthcare providers before stopping medication creates a framework for safe decision-making that protects patient health while addressing valid worries about side effects. Doctors have the clinical knowledge to assess individual risks and benefits, taking into account factors such as disease severity, alternative treatment options, and proper monitoring during any transition off medication.
- Healthcare providers can use several strategies to address patient concerns while ensuring stability in treatment:
-
- Gradual reduction of medication dosage to minimize withdrawal effects
-
- More frequent monitoring appointments to catch and address complications early on
-
- Exploring different medication options that offer similar benefits but with lower risks
-
- Collaborating with eye specialists for thorough vision assessments
-
- Keeping detailed records of symptoms and their timing to support both medical care and potential legal claims
-
- Patients considering legal action against pharmaceutical companies have stronger cases when they can show that they followed medical advice throughout their treatment. Documentation proving that patients continued receiving appropriate medical care, followed doctor recommendations, and made informed choices strengthens both their medical records and potential legal claims.
-
- The overlap between legal matters and medical needs requires patients to maintain open communication with their healthcare team. Doctors can provide crucial documentation of side effects, treatment changes, and ongoing health impacts that serve as evidence for legal claims. This collaborative approach ensures that patients safeguard both their immediate health requirements and long-term legal interests without compromising either goal.
Conclusion
GLP-1 and NAION Vision Loss: Patients experiencing vision problems after using GLP-1 medications have important choices to make that require both medical knowledge and legal advice. The world of GLP-1 NAION Vision Loss Lawsuits is constantly changing as regulatory bodies review new evidence and affected individuals seek justice through legal action.
Guidance for patients considering a GLP-1 NAION lawsuit requires immediate attention to protect both health outcomes and legal rights. If you have developed NAION or other serious eye conditions while taking GLP-1 receptor agonists, it is important to:
-
- Schedule thorough eye examinations to document the extent of your vision loss
- Keep detailed records of your medication history, including dosage information and how your symptoms have progressed
- Seek advice from qualified legal professionals who specialize in pharmaceutical litigation without delay
- Continue taking prescribed treatments unless your healthcare providers instruct you otherwise
Legal Options: To understand the legal options available for victims of vision loss, it is crucial to carefully assess your specific situation. This includes considering the timing between when you started taking the medication and when your symptoms began, the severity of your visual impairment, and how it affects your daily life and overall well-being.
FREQUENTLY ASKED QUESTIONS ABOUT A GLP-A NAION VISON LAWSUIT
What Are the GLP-1 NAION vision side effects?
What evidence do I need to support my GLP-1 NAION Vision Loss Lawsuit
To support your claim, you will need medical records documenting your use of GLP-1 drugs, details about when you started experiencing GLP-1 NAION vision side effects, and any diagnoses or treatments related to eye issues. Prescription records, notes from your doctor, and documentation of how your life has been affected by vision loss can also strengthen your case.
If You Suffered from GLP-1 NAION Vision Side Effects, Contact GLP-1 NAION Vision Loss Lawyer Timothy L. Miles Today