Introduction to Wegovy and NAION Update: The Eye-Opening Side Effect
Welcome to this authoritative Wegovy and NAION update. Wegovy (semaglutide) has become a cornerstone medication in modern weight management. It is clinically validated, widely prescribed, and increasingly normalized as a long-term treatment for chronic obesity. That broader adoption is also changing the risk conversation.
As use expands, so does real-world surveillance. And in that surveillance, one potential safety issue continues to attract attention from clinicians and patients alike: NAION.
NAION, short for non-arteritic anterior ischemic optic neuropathy, is an uncommon but vision-threatening optic nerve event that can cause sudden, painless vision loss. In plain terms, it is an “eye-opening” side effect not because it is common, but because the consequences can be irreversible. The question patients are asking is also straightforward: is there a credible connection between Wegovy and NAION, and what should I do with that information?
This article provides an evidence-based update, explains what NAION is, clarifies what is known and unknown about semaglutide-associated ocular risk, and outlines practical steps for safer decision-making.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Timothy L. Miles, a Wegovy Vision Loss Lawyer today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.

What Wegovy Is, and Why Safety Monitoring Matters
Wegovy is the brand name for semaglutide at a dose and indication approved for chronic weight management. Semaglutide belongs to the GLP-1 receptor agonist class, a group of medications that mimic the incretin hormone GLP-1 to support:
- Appetite suppression and increased satiety
- Delayed gastric emptying
- Improved glycemic regulation
- Meaningful, sustained weight loss for eligible patients
This is not a short-term cosmetic drug. It is a metabolic therapy used for a chronic condition, often alongside hypertension management, lipid control, and diabetes risk reduction.
That context matters because the safety standard is not simply “does it work?” but “does the benefit remain favorable over years of use in diverse patient populations?” Rare adverse events may not appear clearly in pre-approval trials, especially if the event rate is low or the study population is not representative of broad real-world use.
That is why post-marketing surveillance, registry studies, and large observational datasets become crucial. NAION is one of those adverse events that is rare enough to be hard to capture early but serious enough to warrant careful attention when signals emerge.
Patients have raised concerns about Wegovy’s potential link to vision loss, including serious conditions like NAION which could lead to sudden blindness. There are ongoing lawsuits related to Wegovy’s association with vision loss which highlight these risks further.
NAION Explained: The Vision Event at the Center of the Concern
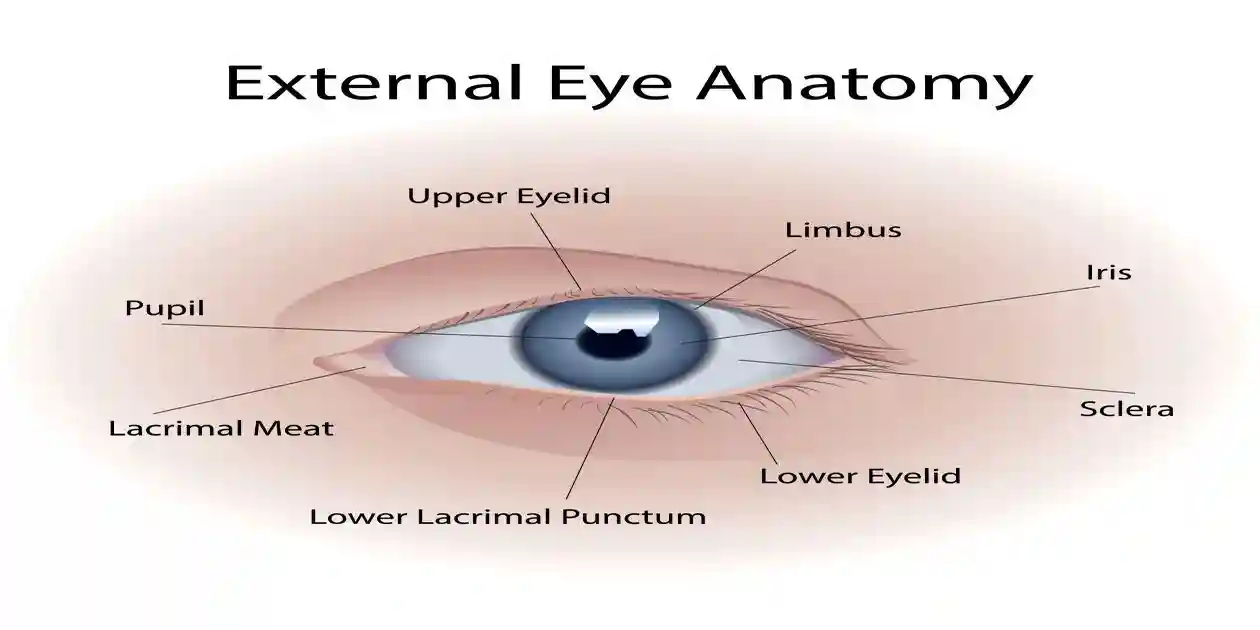
Non-arteritic anterior ischemic optic neuropathy (NAION) is an acute ischemic injury to the optic nerve head. It is not an infection, a retinal detachment, or a typical stroke. Instead, it is a circulation-related injury affecting the anterior portion of the optic nerve.
Core clinical features
NAION typically presents with:
- Sudden, painless vision loss in one eye
- Vision changes noticed upon waking in many cases
- A visual field defect (often inferior altitudinal loss)
- Reduced color vision in some patients
- Optic disc swelling seen on eye examination early in the course
The condition is termed “non-arteritic” to distinguish it from arteritic anterior ischemic optic neuropathy, which is commonly linked to giant cell arteritis and requires immediate steroid treatment. NAION, however, is usually associated with vascular risk factors and structural susceptibility.
Common risk factors for NAION
NAION is more likely in people with:
- Hypertension
- Diabetes or insulin resistance
- Hyperlipidemia
- Obstructive sleep apnea
- Smoking history
- Nocturnal hypotension (low blood pressure during sleep)
- A “crowded” optic disc anatomy (often described as a small cup-to-disc ratio)
- Age typically over 50, though it can occur earlier
These risk factors overlap substantially with the population most likely to be prescribed weight loss medications like Wegovy, Saxenda, Mounjaro, and Trulicity. Such medications have been associated with serious side effects, including vision loss. For instance, Mounjaro has been linked to impairing vision side effects. Similarly, Saxenda has also shown to have detrimental effects on vision.
Moreover, there are reports of alarming vision-related side effects associated with other weight loss drugs such as Zepbound and Wegovy. These complications complicate causal interpretation and make rigorous analysis essential when evaluating the relationship between these medications and NAION’s onset.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Timothy L. Miles, a Wegovy Vision Loss Lawyer today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
Why NAION is taken seriously
NAION can improve somewhat, but many patients are left with persistent visual field loss. There is also risk to the fellow eye over time. Because the event can be sudden and functionally significant, even a small change in risk profile is clinically important.
The Wegovy and NAION Signal: What “Update” Means in Practice
When people search for a “Wegovy and NAION update,” they are usually looking for one of three things:
- Has a causal link been proven?
- Has the FDA (or another regulator) issued a warning?
- What should patients do right now?
The most responsible answer requires careful language.
1) A signal is not the same as proof
A safety signal can emerge through case reports, adverse event reporting systems, or observational database studies. These sources can suggest an association. They rarely prove causality by themselves.
NAION is uncommon. It also occurs disproportionately in people with cardiometabolic disease. Since Wegovy is used in the same population, cases of NAION will happen in Wegovy users even if Wegovy has no causal role. However, recent reports have raised concerns about potential links between Wegovy and NAION, prompting some patients to seek legal advice from Wegovy blindness lawyers.
The scientific question is whether the rate of NAION is higher than expected after accounting for baseline risk.
2) Observational evidence is inherently limited
Large-scale observational studies can be informative, but they are subject to:
- Confounding by indication (patients prescribed semaglutide may be systematically different from those who are not)
- Differences in monitoring intensity (patients on newer therapies may see clinicians more often, increasing detection)
- Misclassification (diagnostic coding may be imperfect)
- Time-window assumptions (when, exactly, the risk is hypothesized to increase)
That said, observational work is still valuable because it captures real-world diversity that trials cannot.
Additionally, other medications like Trulicity, Saxenda, Zepbound, and Mounjaro have also been associated with NAION, leading to various lawsuits regarding these medications as well.
3) Regulatory labeling evolves slowly and conservatively
Even when signals arise, regulators typically require consistent evidence before mandating label changes. They may also examine mechanistic plausibility and whether the event appears across the medication class.
Patients should not interpret the absence of a prominent warning as proof of zero risk. Similarly, they should not interpret early signals as definitive proof of causation. The correct posture is risk-aware, evidence-guided, and individualized.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Timothy L. Miles, a Wegovy Vision Loss Lawyer today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
A Practical Risk Framing: Who Might Be More Vulnerable?
Since NAION risk is strongly shaped by baseline factors, the practical question becomes: which Wegovy candidates might need extra caution and extra monitoring?
Higher baseline NAION risk profiles
You may warrant a more detailed discussion with both your prescriber and an eye-care professional if you have:
- A prior history of NAION in either eye
- Known optic nerve “crowding” or a previously noted small cup-to-disc ratio
- Significant obstructive sleep apnea, especially if untreated
- Poorly controlled hypertension or diabetes
- Multiple vascular risk factors plus a history of nocturnal hypotension
- A strong smoking history
- Prior episodes of sudden visual field defects or unexplained vision changes
This does not automatically mean “do not use Wegovy.” It means the benefit-risk calculation should be more explicit, and the monitoring plan should be more structured.
The special case: prior NAION
If you have had NAION in one eye, clinicians often view the fellow eye as at increased risk over time. In that context, any therapy that might plausibly influence optic nerve perfusion, blood pressure patterns, or vascular reactivity deserves careful scrutiny. For instance, patients using medications like Wegovy, Trulicity, Saxenda, or Mounjaro should be particularly vigilant.
Patients with prior NAION should not discontinue or start medications based on internet summaries. They should coordinate care across primary care, endocrinology or obesity medicine, and ophthalmology or neuro-ophthalmology.
It’s also important to note that certain medications can lead to specific eye problems like eye pain or other eye-related issues. Therefore, maintaining open communication with healthcare providers about any unusual visual symptoms is crucial for patient safety.
Mechanisms: How Could a GLP-1 Drug Be Involved?
At present, the mechanism, if any, remains uncertain. Still, understanding plausible pathways helps patients ask better questions and helps clinicians design safer follow-up.
Potential theories discussed in clinical circles include the following.
Hemodynamic and perfusion-related effects
NAION is often conceptualized as a “perfusion problem” at the optic nerve head. That makes clinicians attentive to factors that can influence perfusion pressure, particularly at night.
Semaglutide can contribute indirectly to changes in:
- Blood pressure (weight loss often lowers blood pressure over time)
- Volume status (nausea, reduced intake, dehydration in some patients)
- Medication regimens (patients may need antihypertensive dose adjustments)
If blood pressure drops too low overnight, optic nerve perfusion can be compromised in susceptible individuals. This does not mean semaglutide “causes” NAION, but it underscores why blood pressure management and hydration patterns matter during rapid metabolic change.
Metabolic shifts and rapid risk-factor change
Rapid improvement in glucose control, especially in diabetes, has been linked historically to transient worsening of some microvascular complications in certain contexts. The best-known example is diabetic retinopathy changes observed with rapid A1c reduction.
NAION is not diabetic retinopathy. Still, it exists within a vascular vulnerability landscape. Rapid metabolic shifts, changes in lipid profiles, and changes in vascular tone have been raised as theoretical contributors.
Class effect versus drug-specific effect
If NAION risk is truly increased, the next question is whether it reflects:
- A semaglutide-specific issue
- A GLP-1 receptor agonist class issue
- A population-risk issue related to the treated cohort
Only broader data across multiple GLP-1 agents, indications, and dosing patterns can clarify that.
Symptoms Patients Should Treat as Urgent
NAION is an eye emergency in the sense that sudden vision loss requires immediate evaluation. Treatment options are limited, but time-sensitive assessment is still essential. A clinician must also rule out arteritic causes, retinal detachment, retinal vascular occlusion, and other urgent conditions.
Seek urgent medical care, ideally emergency evaluation or same-day ophthalmology, if you experience:
- Sudden vision loss in one eye
- New blind spots or a “curtain” or “shadow” in the visual field
- Sudden loss of color intensity or contrast in one eye
- Noticeable asymmetry between eyes that appears over hours, not weeks
- Severe headache, scalp tenderness, jaw claudication, or systemic symptoms alongside vision changes (important for ruling out giant cell arteritis in appropriate age groups)
Do not wait to “see if it goes away,” and do not take your next dose until you have medical advice. The priority is diagnosis and appropriate triage.
What Patients Should Do Before Starting Wegovy (If Concerned About NAION)
Most patients will never experience NAION. However, proactive governance of risk is what separates safe long-term therapy from preventable harm.
1) Establish baseline ocular context
Consider a baseline eye exam if you have significant vascular risk factors, known optic nerve anatomy concerns, or a history of eye disease. You can ask your eye-care provider specifically to document:
- Optic nerve appearance, including cup-to-disc ratio
- Any asymmetry between eyes
- Retinal and optic nerve health baseline
This does not “prevent” NAION, but it improves diagnostic clarity if symptoms arise later.
2) Be Aware of Potential Side Effects from Medications like Wegovy
Before starting medications such as Wegovy, it’s important to be aware of the potential eye side effects that could arise. While most patients will not experience severe issues like NAION, understanding the risks can help in making informed decisions about your health.
3) Monitor for Signs of Eye Damage
If you have started taking Wegovy and notice any unusual eye damage or side effects, seek immediate medical attention. This is crucial as some side effects may require urgent intervention.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Timothy L. Miles, a Wegovy Vision Loss Lawyer today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
4) Consider Other Medications with Similar Risks
If you are also considering other medications such as Zepbound or Mounjaro, be aware that they too may carry similar eye side effect risks or require lawsuits due to severe side effects. Always consult with your healthcare provider about the potential risks associated with any new medication.
2) Review sleep apnea status
Obstructive sleep apnea is a recurring risk factor in NAION literature. If you have loud snoring, witnessed apneas, or excessive daytime sleepiness, treat sleep apnea as part of the Wegovy safety plan, not an unrelated issue.
3) Coordinate blood pressure management, especially at night
Patients on antihypertensive medications should discuss whether they are experiencing dizziness, lightheadedness, or low morning blood pressures during weight loss. Clinicians sometimes adjust timing and dose to avoid excessive nocturnal hypotension in vulnerable individuals.
Do not change antihypertensive regimens on your own. The goal is controlled blood pressure, not minimized blood pressure.
4) Prevent dehydration during dose escalation
Many Wegovy users experience nausea, reduced appetite, and lower fluid intake during titration. Dehydration can worsen hypotension and can contribute to systemic stress.
A practical approach is to:
- Track daily fluid intake intentionally during titration weeks
- Address persistent vomiting or diarrhea early
- Discuss anti-nausea strategies with your prescriber if needed
What Patients Should Do If They Are Already Using Wegovy
If you are stable on Wegovy with no visual symptoms, the most rational approach is structured vigilance rather than panic.
Do not discontinue abruptly without medical advice
Stopping semaglutide can lead to rebound appetite, weight regain, and loss of metabolic improvements. Those changes can raise cardiovascular risk over time. A medication decision should reflect total risk, not one feared outcome.
Do a targeted risk review at your next visit
At your next follow-up, consider asking:
- Based on my risk factors, do you view NAION as a material risk for me?
- Should I have a baseline eye exam or neuro-ophthalmology consult?
- Are my blood pressure readings trending too low with weight loss?
- Should we adjust other medications as my weight and A1c improve?
- If I have visual symptoms, what is the exact plan for urgent evaluation?
This is not defensive medicine. It is appropriate governance of a chronic therapy.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Timothy L. Miles, a Wegovy Vision Loss Lawyer today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
Clinician Perspective: How Responsible Prescribing Usually Looks
In high-quality obesity medicine practice, clinicians generally do three things well:
- They quantify baseline cardiometabolic risk.
- They monitor change over time, particularly during titration and rapid weight loss.
- They educate patients on specific red-flag symptoms and what to do.
When a potential NAION association is raised, responsible prescribers do not default to denial or alarm. They shift toward more explicit documentation, clearer informed consent, and more deliberate coordination with eye care for higher-risk patients.
This approach is also forward-thinking. It reflects a healthcare system that values prevention, early detection, and integrity in safety communication.
Benefit-Risk Balance: Why the Conversation Must Stay Nuanced
It is easy for NAION discussions to become polarized. Either Wegovy is framed as completely safe, or it is framed as unacceptably dangerous. Neither framing is accurate.
Wegovy’s benefits are real and clinically meaningful
For appropriate patients, Wegovy can:
- Reduce body weight significantly
- Improve blood pressure and lipid profiles
- Improve glycemic control and insulin resistance
- Reduce progression to type 2 diabetes in high-risk individuals
- Support functional improvements in mobility and quality of life
Obesity is not a benign condition. It is a chronic disease associated with cardiovascular disease, sleep apnea, fatty liver disease, osteoarthritis, and reduced life expectancy. Effective therapy has substantial upside.
NAION’s severity requires respect even if the absolute risk is low
A rare adverse event can still be decisive for a particular patient, especially one with prior NAION or high vulnerability.
The correct conclusion is not “ignore the signal,” and it is not “stop the drug.” The correct conclusion is: treat the signal as clinically relevant, keep evaluating the data, and individualize care.
The Most Important Takeaway
If you are using Wegovy or considering it, the practical “update” is this:
- NAION is a rare but serious optic nerve event that can cause sudden vision loss.
- People with obesity, diabetes, hypertension, and sleep apnea already have higher baseline risk for NAION, independent of medication choice.
- Emerging safety discussions have increased awareness of a potential association between semaglutide therapy and NAION, but causality is not definitively established in a way that applies uniformly to all patients.
- The correct response is proactive monitoring, symptom education, and individualized risk assessment, particularly in patients with prior NAION or multiple vascular risk factors.
If you want a simple rule that remains medically conservative and patient-protective: any sudden change in vision while on Wegovy should be treated as urgent until proven otherwise.
If you were prescribed Wegovy and took it as directed and suffered Wegovy and vision loss or other Wegovy eye damage, contact Timothy L. Miles, a Wegovy Vision Loss Lawyer today. You could be eligible for a Wegovy Vision Loss Lawsuit and potentially entitled to substantial compensation.
Frequently Asked Questions about Wegovy and NAION
What is Wegovy (semaglutide) and how does it work for weight management?
Wegovy is the brand name for semaglutide, a GLP-1 receptor agonist medication approved for chronic weight management. It supports appetite suppression, increased satiety, delayed gastric emptying, improved glycemic regulation, and meaningful sustained weight loss in eligible patients. It is used as a metabolic therapy for chronic obesity alongside management of related conditions like hypertension and diabetes risk.
What is non-arteritic anterior ischemic optic neuropathy (NAION) and why is it a concern with Wegovy?
NAION is an uncommon but serious optic nerve event causing sudden, painless vision loss due to acute ischemic injury to the optic nerve head. It is vision-threatening and can lead to irreversible damage. NAION has attracted attention in real-world surveillance of Wegovy because some patients have raised concerns about a potential link between semaglutide use and this rare eye condition.
Who is at higher risk of developing NAION while using weight loss medications like Wegovy?
Individuals with vascular risk factors such as hypertension, diabetes or insulin resistance, hyperlipidemia, obstructive sleep apnea, smoking history, nocturnal hypotension, crowded optic disc anatomy (small cup-to-disc ratio), and age over 50 are at higher risk. These factors overlap with the population commonly prescribed weight loss drugs including Wegovy.
Are there confirmed safety risks of vision loss associated with Wegovy and similar medications?
While NAION and other serious vision-related side effects are rare, ongoing post-marketing surveillance and lawsuits highlight concerns about potential associations between GLP-1 receptor agonists like Wegovy, Saxenda, Mounjaro, Trulicity, Zepbound and vision loss. Due to overlapping risk factors in the patient population, rigorous analysis is essential to clarify causality.
What symptoms should patients watch for that may indicate NAION during treatment with Wegovy?
Patients should be alert for sudden, painless vision loss in one eye, especially if noticed upon waking. Other signs include visual field defects (often inferior altitudinal loss), reduced color vision, or swelling of the optic disc detected during an eye examination. Prompt medical evaluation is critical if these symptoms occur.