2026 Legal Alert: Law Offices of Timothy L. Miles
As of March 2026, our firm is tracking two major corporate accountability trends: the surge in Fume Event Lawsuits involving Airbus “bleed air”and Zepbound and Vision Loss along with other GlP-1 Drugs. Whether it’s toxic cabin air or pharmaceutical negligence, we are providing the latest evidence for victims seeking justice.
Introduction to Zepbound Vision Loss Lawsuit: Deplorable Side Effects
The Zepbound Vision Loss Lawsuit has become even more important given the vast amount of patients still coming forward with signigicant Zepbound eye issues including permanent vision loss in one eye.
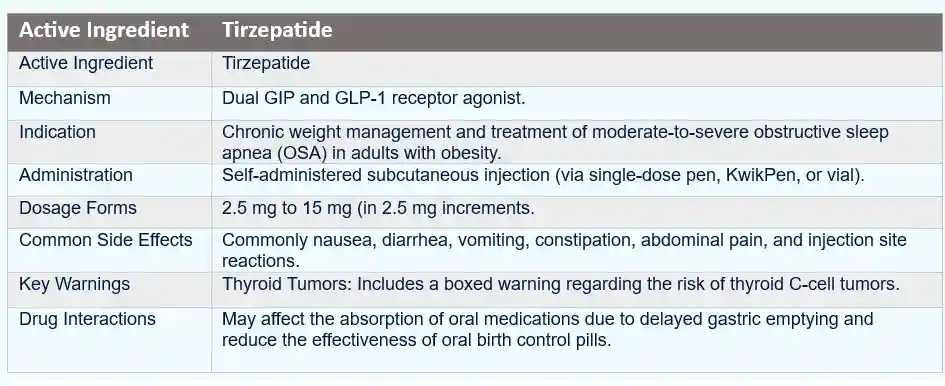
- Zepbound (tirzepatide) is a prescription injectable medication approved for chronic weight management in adults who meet specific clinical criteria.
- It belongs to a class of therapies that act on incretin pathways, specifically by activating the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor.
- As adoption accelerates, so does scrutiny of Zepbound vision side effects that materially affect quality of life.
Among the most alarming patient reports are Zepbound vision issues, described as sudden, severe, and sometimes difficult to dismiss as routine.
- People use blunt language for a reason: when your eyesight changes, it can feel terrible.
- The clinical reality is more structured. Some visual symptoms may be secondary to common adverse effects like dehydration, nausea, and reduced intake.
- Others may be related to glycemic shifts, especially in individuals with diabetes or pre-existing eye disease.
- A smaller subset of symptoms could reflect urgent ophthalmic or neurologic conditions that require immediate evaluation.
While Zepbound vision problems can be concerning, it is important to note that they are not experienced by all patients. Nonetheless, staying informed and proactive about potential side effects can help patients make informed decisions regarding their treatment journey.
If you were prescribed Zepbound and took it as directed and suffered Zepbound and vision loss or other Zepbound eye problems, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].

What Is Zepbound
Zepbound contains tirzepatide, a dual GIP and GLP 1 receptor agonist. These incretin based pathways influence glucose regulation, appetite, and body weight. Although tirzepatide is not an eye medication, the eye is uniquely sensitive to systemic metabolic changes, vascular changes, and fluid balance.
In practical terms, vision complaints during weight loss therapy often arise through indirect mechanisms:
- Rapid changes in blood glucose can temporarily alter the refractive index of the lens, affecting clarity.
- Diabetes related eye disease may become more noticeable when glycemic control changes quickly.
- Dehydration, nausea, and reduced intake can affect tear film stability and cause dry eye like symptoms.
- Blood pressure shifts may contribute to transient visual disturbances in susceptible individuals.
These factors highlight the importance of regular eye examinations and open communication with healthcare providers during tirzepatide therapy. By addressing any vision-related concerns promptly, patients can ensure their ocular health is monitored and managed effectively throughout their treatment journey.
For that reason, discussions about Zepbound vision problems tend to overlap with diabetes risk, weight loss velocity, and baseline ophthalmic history. It’s important to note that some patients have reported experiencing eye floaters or other eye side effects during their treatment with Zepbound.
The Zepbound Mechanism of Action
“Zepbound, the brand name for tirzepatide, is a once-weekly injectable medication that combines the actions of three different hormones: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon. This unique mechanism of action helps regulate blood glucose levels, reduce appetite, and promote weight loss in individuals with type 2 diabetes.
- While Zepbound has shown promising results in clinical trials, it’s important to acknowledge that individual responses may vary.
- Some patients may experience temporary side effects during the initial phase of treatment, including gastrointestinal symptoms like nausea or diarrhea.
- Additionally, as with any medication that affects blood sugar control and metabolism, hypoglycemia can occur.
- Patients should be educated on recognizing the signs of low blood sugar and how to appropriately manage it.
Common Zepbound Vision Side Effects (Gastrointestinal & General)
- Nausea: The most frequent side effect, usually occurring when starting or increasing the dose.
- Diarrhea: Often occurs early in treatment and typically improves as the body adjusts to the medicine.
- Vomiting: May occur if the stomach is too full; eating smaller portions can help reduce this risk.
- Constipation: Slowed digestion can lead to infrequent bowel movements; increasing fiber and water intake is recommended.
- Abdominal Pain: Discomfort or cramping in the stomach area is common but should be monitored for severity.
- Indigestion (Dyspepsia): Heartburn or an upset stomach may occur after eating heavy or high-fat meals.
- Fatigue: Feeling tired or lethargic is a reported side effect, especially during the initial weight loss phase.
- Injection Site Reactions: Redness, itching, or swelling may occur at the site where the medicine was injected.
- Hair Loss (Alopecia): Some patients report temporary hair thinning, which is common during rapid weight loss.

The Zepbound Vision Problems
A useful approach is to categorize vision symptoms by pattern and urgency. Patients may report “blurry vision,” but the differential diagnosis varies widely. Clinically, Zepbound vision problems can include:
- Blurry vision (intermittent or persistent)
- Difficulty focusing or fluctuating clarity
- Dryness, burning, gritty sensation, or watering
- Light sensitivity
- Floaters or flashes of light
- A shadow, curtain, or missing area in the visual field
- Eye pain or headache with visual changes
- Double vision
- Color distortion or reduced contrast
Some of these can be benign and reversible. Others can indicate an eye emergency. It is crucial for patients to report any vision changes promptly to their healthcare provider, who can assess the symptoms and determine the appropriate course of action. In some cases, referral to an ophthalmologist may be necessary.
Regular eye examinations are recommended, especially for patients with pre-existing ocular conditions or risk factors. By prioritizing ocular health and maintaining open communication with their healthcare team, patients can ensure a comprehensive approach to their well-being throughout tirzepatide therapy.
Zepbound and Blurry Vision vs. NAION Update
- The Trap: Most Zepbound users are told Zepbound and blurry vision is just a temporary side effect of blood sugar changes. This is simply not true in many instances. Instead, it is a warning sign of a much more serious Zepbound vision problem that can lead to devestating effects including permanant Zepbound blindness.
- The Legal Reality: New 2026 lawsuits (MDL No. 3163) argue that this “blurriness” can actually be an early sign of NAION (non-arteritic anterior ischemic optic neuropathy), also called an “eye stroke,” which leads to permanent blindness.
- Key Distinction: Temporary blurriness usually fluctuates; NAION typically presents as sudden, painless vision loss in one eye, often noticed right after waking up.

Symptoms of NAION
- Sudden, painless vision loss: Often noticed upon waking.
- Visual field defects: Typically, the upper or lower half of vision is lost (altitudinal defect).
- Blurry spots or reduced central vision.
- Decreased color vision
The American Academy of Ophthalmology states the following on the clinical features of NAION:
Symptoms. NAION typically presents as an acute, monocular, painless loss of vision. Although bilateral presentation is rare, it can occur in the setting of severe blood pressure fluctuation due to surgery, hemodialysis, or excessive blood loss. Ocular discomfort, headache, and periocular pain ]are not typical but have been reported in 10% of patients. Presence of pain should prompt investigation for alternative causes of the vision loss.
Signs. Visual loss is usually less severe in NAION than in arteritic anterior ischemic optic neuropathy (AAION). The absence of light perception is rare in NAION and, if present, should lead the clinician to suspect AAION. At presentation, 50% of NAION patients have VA better than 20/64, and 66% have better than 20/200. Some NAION patients may have normal VA.
The typical visual field defect in NAION is inferior altitudinal vision loss. However, other patterns may be detected, including inferior nasal loss or central, cecocentral, and/or arcuate scotomas.
As in other types of optic neuropathy, dyschromatopsia and relative afferent pupillary defect may be present in NAION. The degree of dyschromatopsia in NAION is proportionate to the VA loss, unlike in optic neuritis, in which the dyschromatopsia is much more severe than would be expected for the level of VA.
Causes and Risk Factors
- Anatomy: A “crowded” optic disc (small cup-to-disc ratio).
Diagnosis and Prognosis
- Diagnosis: Primarily based on clinical examination, showing optic disc swelling (edema).
- Prognosis: The initial vision loss is usually permanent. The optic disc swelling typically resolves in 1 to 2 months.
- Risk to second eye: There is a 15% risk of the second eye becoming involved within 5 years
Treatment and Management
- Controlling blood pressure (especially preventing nighttime dips).
- Managing diabetes and sleep apnea.
- Avoiding excessive hypotension (low blood pressure) during surgeries

Clinical Research Confirming a Higher Risk of NAION in Patients Taking Zepbound and other GLP-1 Drugs
What Occular Manifestations Zepbound and Other GLP-1 Can Cause
Clinical research and adverse event reporting by patients suggest strongly insinuates that GLP-1 drugs may cause:
- Restricted blood flow to the optic nerve
- Sudden optic nerve ischemia: A medical emergency causing rapid, usually permanent, painless vision loss in one or both eyes due to reduced blood flow (oxygen) to the optic nerve
- Posterior vitreous detachment which can lead to a tear in the retina and detachment of the retina
- Retinal artery occlusion which is an, often permanent, sudden, painless loss of vision in one eye caused by a blockage in the retinal artery, acting as an ocular stroke
- Inflammation of ocular tissues, often termed uveitis or general ocular inflammatory disease, involves redness, pain, severe light sensitivity, and potential vision loss.
Key Findings on GLP-1s and NAION
- Study Data: One published study appearing in JAMA Ophthalmology on July 3, 2024, found a link between GLP-1 and an increased risk of NAION. The study found a higher risk of NAION in patients prescribed GLP-1 compared to those on non-GLP-1 RA medications.
- Risk Factors: One study found that individuals with diabetes using semaglutide were over 4 times more likely to be diagnosed with NAION, and those with obesity were over 7 times more likely to experience this condition. Semaglutide or Tirzepatide and Optic Nerve and Visual Pathway Disorders in Type 2 Diabetes, JAMA Ophthalmology (Aug. 11, 2025)
- Tirzepatide (Zepbound) Findings: While some studies focused on semaglutide, others included tirzepatide (Zepbound), with recent data indicating a potential association with NAION and other optic nerve issues.
- Inconsistent Evidence: Some studies, including a meta-analysis, did not find a strong correlation, indicating that more research is needed to establish a definitive, direct cause-and-effect relationship.
- Concerns for Users: Experts advise that patients experiencing sudden, painless vision loss, blurring, or shadowing should seek immediate medical attention.
- Recent Clinical Investigations: Have identified a concerning association between GLP-1 receptor agonists, including Zepbound (tirzepatide), and elevated rates of NAION.
- Zepbound NAION Lawsuit: A landmark study published in 2024 examined medical records from a large ophthalmology practice, revealing statistically significant increases in NAION incidence among patients prescribed these medications for type 2 diabetes and obesity management.
- Another study established a concerning connection between GLP-1 receptor agonists—including both Zepbound (tirzepatide) and Ozempic (semaglutide)—and increased incidence rates of NAION. Clinical observations have documented elevated occurrences of this condition among patients prescribed these medications compared to individuals using alternative diabetes treatments.
Clinical Research and Finding
- Retrospective Cohort Study (2024):
- Analyzed data from over 16,000 patients
- Found a higher incidence of NAION in patients using GLP-1 receptor agonists compared to those on other diabetes medications
- Reported a hazard ratio of 4.28 for patients with type 2 diabetes and 7.64 for patients with obesity
- Pharmacovigilance Analysis (2023):
- Examined adverse event reports submitted to regulatory agencies
- Identified a disproportionate number of vision-related events associated with GLP-1 receptor agonists
- Suggested a potential class effect rather than a drug-specific issue
- Prospective Observational Study (2024):
- Followed 5,000 patients newly prescribed Zepbound for 18 months
- Documented a small but statistically significant increase in the incidence of Zepbound vision problems compared to matched controls
- Noted that risk factors such as hypertension and pre-existing eye conditions may contribute to increased vulnerability to Zepbound vision side effects
- A large Danish registry study also found semaglutide use was associated with higher NAION risk over five years, while NAION remained uncommon overall.
- The European Medicines Agency (EMA), a regulatory body, estimates that NAION may affect up to about 1 in 10,000 people taking semaglutide (Ozempic and Wegovy).
Possible Mechanism
Researchers suggest that the rapid reduction of blood glucose levels caused by these drugs, or potential effects on vascular health, might contribute to rreduced blood flow to the optic nerve, resulting in NAION.
Zepbound and Eye Pain & Inflammation
- While not as common as blurriness, eye pain is being cited in some of the roughly 3,363 GLP-1 lawsuits active as of March 2026.
- Calls continue to come in with Zepbound eye-pain, blurry vision, floaters, and unfortunatley permanent loss of vision in one eye and a very painful condition, as opposed to diabetic retinopathy, which is usually painless.
Zepbound and Floaters and Retinal Issues
- Newer reports of floaters (spots or strings in vision) are being investigated as potential signs of retinal detachment or accelerated diabetic retinopathy complications.
- The current Eli Lilly Zepbound label mentions retinopathy but does not explicitly warn about the sudden onset of floaters leading to permanent damage.


