Introduction to the Optimum Patient Guide to the Dupixent Lawsuit
- Dupixent Lawsuit: If you who were prescribed Dupixent and were subsequently diagnosed with T-Cell Lymphoma or experienced other severe Dupixent side effects, call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation as you may be eligible for a Dupixent Cancer Lawsuit and possibly entitled to substantial compensation. (855) 846-6529
- Dupixent (dupilumab): Is an injectable biologic medication that is used to treat particular types of inflammatory diseases. The medicine is currently the subject of a growing number of lawsuits alleging it causes or accelerates the development of cutaneous T-cell lymphoma (CTCL), a rare form of cancer. As of November 2025, the litigation is in early stages, with no settlements or jury verdicts yet.
- Recent studies on Dupixent and Cancer: Have shown that Dupixent use may increase the risk of certain types of cancers known as Cutaneous T-cell Lymphomas (CTCL), including its subtypes Mycosis fungoides and Sezary syndrome, by as much as 300% or more in people who used the medication.
- Potential Compensation: People who used Dupixent and were later diagnosed with CTCL may be eligible to file a Dupixent Cancer Lawsuit and potential entitled to substantial compensation. If you suffered Dupixent and Cancer call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation. (855)-846-6529 or [email protected].
- Who Is Eligible for a Dupixent Lawsuit. If you who were prescribed Dupixent and were subsequently diagnosed with T-Cell Lymphoma or experienced other severe Dupixent side effects, call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation as you may be eligible for a Dupixent Lawsuit and possibly entitled to substantial compensation. (855) 846-6529 or [email protected]
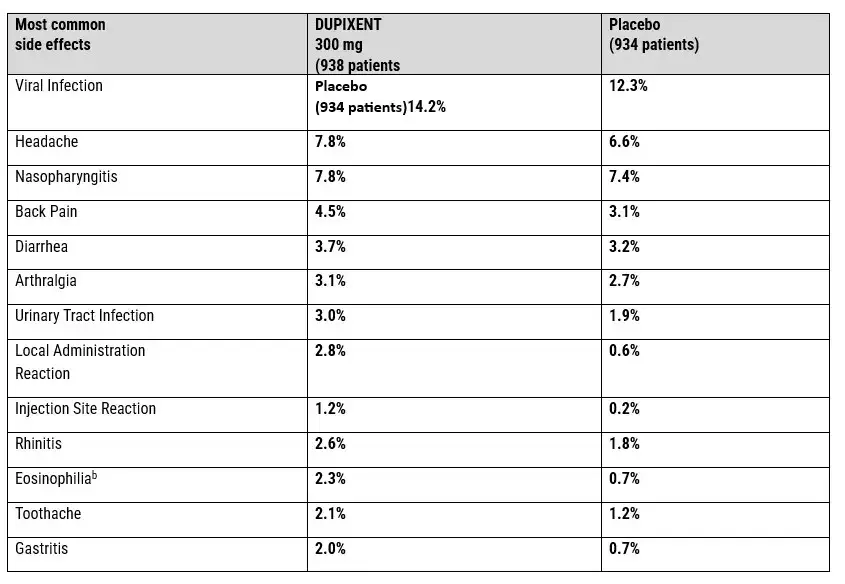
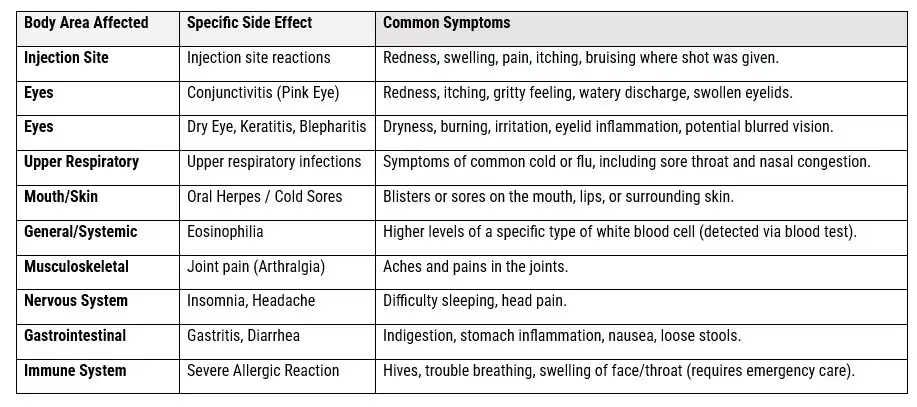
THE DUPIXENT COMMON SIDE EFFECTS PROFILE
Dupixent Lawsuit Status (as of November 2025)
- Dupixent Lawsuit: Individual lawsuits have been filed across the U.S., but they have not yet been consolidated into a formal multidistrict litigation (MDL).
- First Lawsuit: The first known Dupixent Lawsuit for wrongful death lawsuit was filed in a federal court in Tennessee in October 2025. The lawsuit alleges the drug caused a woman’s fatal T-cell lymphoma and that the manufacturers failed to provide adequate warnings.
- No MDL or Class Action: There is currently no formal multidistrict litigation (MDL) or class-action lawsuit. However, legal experts anticipate that cases may be consolidated into an MDL in late 2025 or early 2026 to streamline the legal process if more cases emerge.
- Allegations: Lawsuits primarily allege that the manufacturers, Sanofi and Regeneron, failed to adequately warn patients and healthcare providers about Dupixent and Cancer risks associated with the drug.
- FDA Investigation: The U.S. Federal and Drug Administration (FDA) is formally investigating the potential link and evaluating whether new cancer warnings should be added to the Dupixent label. The current label does not mention a CTCL risk.
- Settlements: No settlements have been reached in any Dupixent Lawsuit , and there are no jury verdicts, making it difficult to estimate potential settlement amounts.