Introduction to the 7 Critical Points Consumers Must Know
- Mounjaro Blindness Lawsuit: Refers to an onslaught of lawsuits and represents a critical
progression for consumers who have experienced detrimental Mounjaro vision problems while using this medication. Medical professionals and researchers have evidenced divergent ocular complications ranging from temporary visual disturbances to permanent vision loss, prompting a panorama of litigation against the pharmaceutical manufacturer. These Mounjaro vision side effects have led to allegations that Eli Lilly and Company failed to provide adequate warnings about the potential risks to patient eyesight.
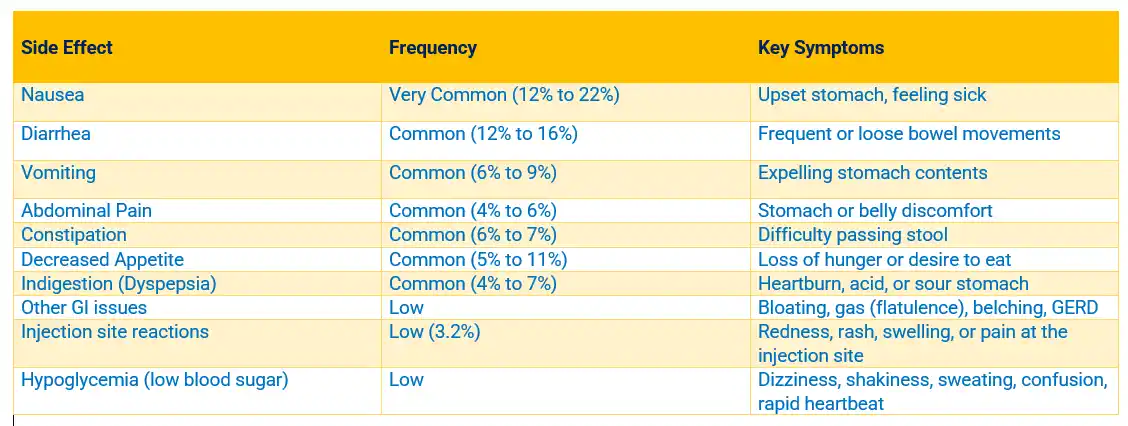
- Mounjaro (tirzepatide): Is manufactured by Eli Lilly, represents a significant betterment in treating type 2 diabetes and facilitating weight loss. This GLP-1 receptor agonist works by mimicking naturally occurring hormones that regulate blood sugar levels and appetite control. The medication has gained substantial attention for its efficacy in managing glycemic control and promoting substantial weight reduction in clinical trials and real-world applications.
- Mounjaro Vision Problems: New evidence has raised serious concerns about vision-related complications linked to Mounjaro use. Clinical research and reports of sudden vision loss, blindness, and a condition called nonarteritic anterior ischemic optic neuropathy (NAION) have led thousands of patients filing an against the manufacturer. These complications are potentially life-changing injuries that go beyond the usual risks associated with diabetes medications.
- Mounjaro Vision Damage: If you suffered the debilitating Mounjaro vision side effects, contact Mounjaro vision loss lawyer Timothy L. Miles for a free case evaluation. (855) 846–6529 or [email protected].
- The Purpose of this Patient Guide: Patients need reliable information to make informed choices about their health and legal options when dealing with potential medication-related injuries. However, that can be challenging and seem overwhelming because of the vast quality of information spread out throughout various medical and clinical research reports. Therefore, we have attempted to the best of our ability to provide you with the most important information concerning Mounjaro vision problems with links to the most authoritative and reliable sources, such as the National Library of Medicine, and other trustworthy references you can depend on. We hope you find this information helpful and useful.
MOUNJARO VS. OPZEMPIC COMPARISON CHART
1. Nonarteritic Anterior Ischemic Optic Neuropathy
- Breaking down the meaning of the name component-by-component:
-
- Non-Arteritic: The reason it is called “non-arteritic” is because there is reduced blood flow without true inflammation of the blood vessels (as there is in arteritis).
-
- Anterior: It is called “anterior” because the reduced blood flow and injury to the optic nerve happen at the front-most part of the nerve, where the nerve meets the eye.
-
- Ischemic: The reason it is called “ischemic” is because that is the word that describes an injury due to reduced blood flow.
-
- Optic Neuropathy: The reason it is called an “optic neuropathy” because it is an injury to the optic nerve, which disrupts the ability of eye to send information to the brain.
Defining the Characteristics of NAION
- NAION is characterized by sudden, painless, unilateral vision loss upon awakening.
- Is the most common cause of optic nerve swelling and optic neuropathy in adults older than 50.
- NAION and is caused by decreased blood flow to the front part of the optic nerve (optic disc). However, the exact mechanism causing reduced blood flow to the optic nerve is not proven.
- What is known is that NAION occurs more often when a patient has conditions such as diabetes, high blood pressure, and sleep apnea. Additionally, smoking may also elevate the risk of developing NAION.
- Additionally, it happens more often in patients born with small optic discs (the front part of the optic nerve that can be seen within the eye).
- It typically affects one eye, although the other eye sometimes suffers similar loss months or years later (there is about a 15% risk of second eye involvement within 5 years). Rarely, both eyes may be affected at the same time.
- Approximately 15% of people with NAION in one eye will develop NAION in the other eye within 5 years
2. Symptoms of NAION
- NAION Clinical Presentation: The way NAION presents itself is quite unique, allowing healthcare providers to recognize it during a thorough eye examination. Patients usually experience:
-
- Sudden Vision Loss: Sudden, painless vision loss in one eye that occurs upon waking up or is noticed within the first few hours of the day
- One Side Involvement: With one eye being affected initially, although there’s an increased risk for the other eye
- Visual Filed Defects: Specific visual field defects, most commonly affecting the lower part of the visual field
- Optic Disc Swelling: Swelling of the optic disc and visible bleeding during an eye examination
- Pupil Defect: A relative defect in pupil response in the affected eye
-
- Dark or Gray Spot: The vision loss from NAION often consists of a gray or dark spot that does not move or change or a severe blur. For many patients, the vision loss affects the bottom or top half of the vision in one eye, although other patterns of vision loss may occur.
-
- Color Vision and Ability to See Contrasts: IIn addition to central vision loss, there may be difficulties with color vision and reduced ability to see contrasts, which can affect daily activities.
3. The Risk Factors for NAION
- Optic Nerve Anatomy: Having a naturally small or “crowded” optic disc (“disc-at-risk”). This is an unchangeable physical trait.
- Age: Most common in adults over 50
- High Blood Pressure (Hypertension): Especially if blood pressure drops too low overnight, sometimes because of medication taken at bedtime.
- Diabetes: Poorly controlled blood sugar and the associated blood vessel damage.
- Sleep Apnea: A condition where breathing stops temporarily during sleep. This is a significant risk factor, possibly due to changes in blood pressure and oxygen levels at night.
- Smoking: Damages blood vessels and is a strong risk factor.
- High Cholesterol (Hyperlipidemia): Contributes to hardening of the arteries (atherosclerosis).
- Certain Medications: The heart medicine amiodarone and some erectile dysfunction drugs (like Viagra, Cialis, Levitra) may be associated with NAION in at-risk individuals.
- Genetic Predisposition: Minor changes in genes related to blood flow control and clotting that can increase susceptibility in some cases.
- Chronic Vascular Effects: Other risk factors may include the chronic vascular other risk factors may include the chronic vascular effects of anemia and kidney disease.
- Nocturnal hypotension: Significant drops in blood pressure during sleep can decrease blood flow to the optic nerve head.
- Individuals with obesity: Excess weight contributes to systemic inflammation and vascular dysfunction
- Patients with pre-existing cardiovascular conditions: Compromised circulation increases the likelihood of optic nerve ischemia
It is vital that you work with your primary care physician to identify and manage the well-established health risks to help prevent NAION in the other eye or a future vascular event like a heart attack or stroke.
4. The Link Between Mounjaro and NAION: Evidence and Research Findings
- Recent Clinical Investigations: Have identified a concerning association between GLP-1 receptor agonists, including Mounjaro (tirzepatide), and elevated rates of nonarteritic anterior ischemic optic neuropathy.
- Mounjaro Vision Loss Lawsuit: A landmark study published in 2024 examined medical records from a large ophthalmology practice, revealing statistically significant increases in NAION incidence among patients prescribed these medications for type 2 diabetes and obesity management. Another study established a concerning connection between GLP-1 receptor agonists—including both Mounjaro (tirzepatide) and Ozempic (semaglutide)—and increased incidence rates of NAION. Clinical observations have documented elevated occurrences of this condition among patients prescribed these medications compared to individuals using alternative diabetes treatments.
Epidemiological Evidence of Increased NAION Risk
- NAION Occurrence Rates: Clinical Results: The research documented NAION occurrence rates across distinct patient populations:
-
- Type 2 diabetes patients: Those prescribed semaglutide or tirzepatide demonstrated a 4.28-fold increased risk of developing NAION compared to patients using alternative diabetes medications
-
- Obesity treatment cohorts: Individuals receiving GLP-1 drugs for weight management exhibited a 7.64-fold elevated risk relative to those prescribed other weight-loss interventions
-
- Absolute incidence rates: The cumulative three-year probability of NAION development reached 8.9% among GLP-1 drug users versus 1.8% in comparison groups
- Linking Mounrjaro to Optic Nerve Problems: These findings represent the first substantial epidemiological evidence linking Mounjaro vision problems to this severe optic nerve disorder. The magnitude of risk elevation has prompted calls for enhanced surveillance protocols and patient counseling regarding potential ocular complications.
Drug Safety Findings
- The Link Between Mounjaro and Vision Loss: Reviews of post-marketing surveillance data have found scientific evidence linking Mounjaro to vision loss through multiple reporting channels. Regulatory agencies have received notifications of eye-related side effects at rates higher than expected for the general population. These analyses have documented:
-
- Disproportionate reporting ratios for NAION among users of GLP-1 receptor agonists
- Increased progression of diabetic retinopathy during treatment intensification
- Reports of sudden visual disturbances requiring immediate eye care intervention
5. Types of Vision Problems Reported with Mounjaro Use
Breaking Down the Mounjaro Vision Problems:: Mounjaro use has been linked to various eye issues beyond just NAION. These problems involve different parts of the eye and include several specific conditions that require thorough assessment and supervision.
1. Diabetic Retinopathy
-
- Diabetic retinopathy is a major concern for patients using GLP-1 receptor agonists like Mounjaro. It involves gradual harm to the blood vessels in the retina, which can be seen through signs such as microaneurysms, bleeding, and new blood vessel growth. Interestingly, while Mounjaro treatment aims to control blood sugar levels quickly, it may unintentionally worsen retinopathy in susceptible individuals, especially those with existing microvascular issues. This is because a sudden drop in blood glucose levels can disturb the retinal blood vessels, causing them to become more permeable and leading to insufficient blood supply.
2. Macular Edema
-
- Another complication associated with Mounjaro is macular edema, characterized by fluid buildup in the macula—the central part of the retina responsible for sharp vision. As a result, patients may experience blurred central vision, difficulty reading, and altered color perception. This condition occurs when the barrier between the blood vessels and retinal tissue is disrupted, allowing fluid leakage into the macula and compromising visual clarity.
3. Dry Eye Syndrome
-
- Mounjaro Vision Side Effects and Dry Eyes: Multiple case reports have documented instances of dry eye syndrome among users of GLP-1 receptor agonists. Patients may feel discomfort on the surface of their eyes, experience burning sensations, and have intermittent blurry vision due to insufficient tear production or excessive evaporation of tears. The underlying mechanism could involve effects on the autonomic nervous system or inflammatory responses triggered by the medication.
4. Myodesopsias (Floaters)
-
- Another phenomenon reported by some patients is myodesopsias, commonly known as floaters. These individuals perceive dark spots or cobweb-like shapes moving across their field of vision—an occurrence stemming from changes in the vitreous humor or disturbances within the retina.
5. Blurred Vision from Hypoglycemia
-
- An acute complication that can arise when Mounjaro’s glucose-lowering effects become too strong is blurred vision due to hypoglycemia. The visual cortex and retinal neurons rely on a steady supply of glucose for proper functioning; when this supply is compromised, it leads to impaired visual processing—resulting in temporary but potentially hazardous vision impairment.
6. Scientific Studies Investigating the Correlation Between Mounjaro and Vision Loss Incidents
- Investigating the Link to GLP-1 Receptors: The medical community has initiated rigorous investigations into the potential association between GLP-1 receptor agonists and adverse ocular outcomes. Research published in JAMA Ophthalmology has provided critical insights into the relationship between these medications and vision-threatening conditions, particularly nonarter
Insights on Mechanisms Behind Adverse Effects
- Mounjaro Eye Problems Due to Reduced Blood Flow to the Optic Nerve: Research emphasizes the role of rapid glycemic fluctuations in compromising optic nerve perfusion. The sudden normalization of blood glucose levels, while therapeutically beneficial for metabolic control, may paradoxically create conditions that reduce blood flow to the optic nerve head.
- Normalization of Ichemia (Oxygen Supply to Blood): This phenomenon, termed “normalization ischemia,” represents a critical pathway through which GLP-1 medications may precipitate vision loss.
Clinical Monitoring Protocols
- Establishing a Bassline Assessment: Physicians recommend establishing baseline ophthalmologic assessments before initiating Mounjaro therapy. This initial evaluation creates a reference point for detecting subsequent changes in ocular health. Patients should receive clear instructions to report any visual disturbances immediately, including:
-
- Sudden onset of blurred vision or visual field defects
- Unexplained eye pain or pressure
- Changes in color perception
- Appearance of floaters or flashing lights
- Difficulty with night vision or contrast sensitivity
- FDA Warnings: The FDA warnings on GLP-1 drugs underscore the necessity for heightened vigilance during the first several months of treatment, when metabolic changes occur most rapidly. Healthcare teams must educate patients about the distinction between temporary visual fluctuations related to blood sugar stabilization and potentially serious complications requiring urgent evaluation.
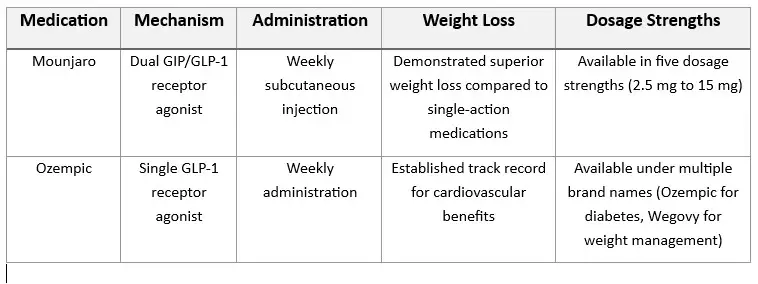
MOUNJARO COMMON SIDE EFFECTS PROFILE
FREQUENTLY ASKED QUESTIONS ABOUT MOUNJARO VISION PROBLEMS
1. What Are the Mounjaro vision side effects?
- A study published in 2024 revealed alarming statistics about NAION risk among patients using GLP-1 receptor agonists like Mounjaro. The research demonstrated that individuals with type 2 diabetes taking these medications faced up to seven times higher risk of developing NAION compared to those using alternative diabetes treatments.
- While not an exhaustive list, some other Mounjaro vision problems include:
-
- The relationship between diabetes complications and retinopathy risk when on Mounjaro requires careful examination, as the medication’s glycemic effects create a complex interaction with existing retinal pathology. Research published in Diabetes Care journal has documented that rapid improvements in glycemic control—while therapeutically beneficial—may paradoxically accelerate diabetic retinopathy progression in susceptible individuals.
-
- Eye floaters
-
- Eye twitching
-
- Inflammation of the eye
2. Is there a link between Mounjaro and Vision Loss?
- Yes, Nonarteritic Anterior Ischemic Optic Neuropathy (NAION) represents one of the most severe vision-related risks documented in association with tirzepatide therapy.
- This condition manifests through sudden vision loss resulting from compromised blood flow to the optic nerve head, leading to optic nerve damage that may be permanent. NAION symptoms and risks include:
-
- Acute, painless vision loss typically affecting one eye
- Visual field defects, particularly altitudinal field loss
- Optic disc swelling observable during fundoscopic examination
- Irreversible vision impairment in the majority of cases
3. What should I do if I experience Mounjaro vision problems?
- Stop taking the medication and contact your healthcare provider immediately.
- Prompt assessment can help prevent further Mounjaro vision side effects.
Am I eligible to file a Mounjaro blindness lawsuit if I suffered Mounjaro vision problems?
- You may be eligible for a Mounjaro blindness lawsuit if you experienced significant eye-related side effects after using Mounjaro and can establish a connection between your injury and the medication.
- To see if you are eligible, contact Mounjaro blindness lawyer Timothy L. Miles for a free case evaluation. (855) 846–6529 or [email protected].
Conclusion
Mounjaro Vision Side Effects: The emerging evidence surrounding Mounjaro Vision Problems necessitates heightened awareness among patients and healthcare professionals. A comprehensive summary of Mounjaro vision problems reveals potential associations with nonarteritic anterior ischemic optic neuropathy, diabetic retinopathy exacerbation, and other serious ocular complications that demand immediate attention.
Patient Vigilance Remains Paramount: In mitigating these risks. Individuals prescribed Zepbound must maintain proactive communication with healthcare providers, reporting any visual disturbances—no matter how subtle—without delay. Regular ophthalmologic examinations should constitute a non-negotiable component of treatment protocols, particularly for patients presenting with diabetes or pre-existing vascular conditions.