Introduction to the Link Between Wegovy and Nonarteritic Anterior Ischemic Optic Neuropathy
- Wegovy Blindness Lawsuit: Refers to recent developments which have brought significant attention to potential ocular side effects associated with Wegovy use. Reports of serious Wegovy vision side effects, including vision loss and blindness, have resulted in legal action against the manufacturer. These concerns have coalesced into the Wegovy Blindness Lawsuit, with patients seeking representation from legal professionals, including Wegovy Blindness Lawyers, to address alleged inadequate warnings regarding vision-related risks.
- Wegovy (semaglutide): A popular prescription medication for long-term weight management. It has been approved by the U.S. Food and Drug Administration for adults with obesity or overweight conditions, especially those with weight-related health issues. Wegovy works by activating the glucagon-like peptide-1 (GLP-1) receptor, which helps people lose weight by reducing their appetite and regulating their metabolism
- Wegovy and Vision Loss: However, recent clinical observations and scientific studies have raised serious concerns about a possible connection between Wegovy and vision loss, specifically Nonarteritic Anterior Ischemic Optic Neuropathy (NAION). NAION is a condition that causes significant damage to the optic nerve and can lead to sudden, permanent vision loss. As a result of this emerging evidence, affected patients are now filing lawsuits against the manufacturers of Wegovy, claiming that they were not adequately warned about these potential vision-related risks.
- Wegovy Vision Side Effects: It is crucial for patients, healthcare providers, and legal professionals to understand the potential vision side effects of semaglutide as they navigate this evolving optical situation. The relationship between GLP-1 receptor agonists and eye health needs careful investigation, especially since millions of people are using these medications to manage their weight.
- Wegovy Blindness Lawsuit: This article offers an in-depth look at the Wegovy eye damage lawsuits. It explores the scientific evidence linking semaglutide to NAION, the legal frameworks governing lawsuits related to vision loss caused by weight loss drugs, and the implications for patient safety and accountability in the pharmaceutical industry.
Understanding Wegovy and Related Medications
- What is Wegovy?: Wegovy (semaglutide 2.4 mg) is part of a group of medications called GLP-1 receptor agonists. The FDA has specifically approved it for long-term weight management in adults with obesity or overweight conditions, especially those with weight-related health issues. Made by Novo Nordisk, this injectable drug is a higher dose version of the active ingredient found in Ozempic, which is mainly used for managing type 2 diabetes.
- GLP-1 Receptor Agonists: There are other GLP-1-based therapies available besides semaglutide products. Mounjaro and Zepbound, both containing the active ingredient tirzepatide, work as dual GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptor agonists. Mounjaro received FDA approval for treating type 2 diabetes, while Zepbound got authorization specifically for weight management. Although these medications have different regulatory classifications and dosing protocols, they share similar goals in terms of treatment.
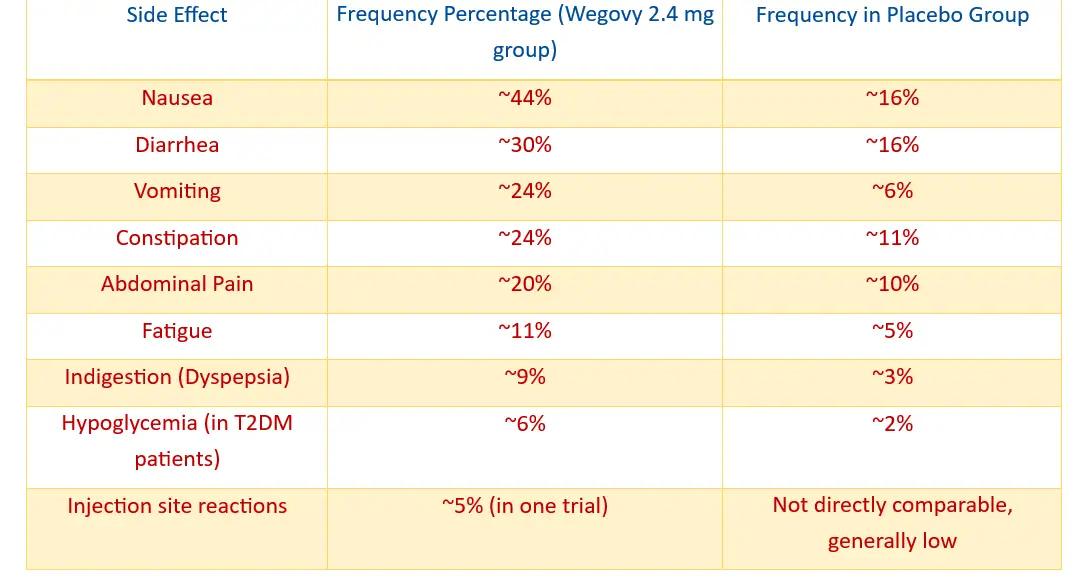
WEGOVY COMMON SIDE EFFECTS
How GLP-1 Receptor Agonists Work
- GLP-1 Receptor Agonists: work by imitating the natural hormone called glucagon-like peptide-1 (GLP-1). This mechanism involves several processes in the body:
-
- Appetite control: Activation of GLP-1 receptors in the brain reduces feelings of hunger and increases the sensation of fullness
-
- Stomach emptying: Slowing down the emptying of the stomach prolongs the feeling of fullness aftermeals
-
- Insulin release: Enhancing the release of insulin from pancreatic beta cells in response to glucose
-
- Glucagon reduction: Decreasing the secretion of glucagon, which leads to reduced production of glucose by the liver
-
- Continuous Receptor Activation: By continuously activating these receptors through synthetic versions like semaglutide and tirzepatide, significant metabolic effects occur. Clinical trials have shown that patients using Wegovy achieved average weight losses of 15-20% of their initial body weight, which is much higher than what traditional weight loss methods can achieve.
FDA Approvals and Growing Market Demand
- FDA Approval: In June 2021, the FDA approved Wegovy for long-term weight management in adults with a body mass index (BMI) of 30 kg/m² or higher, or 27 kg/m² or higher with at least one weight-related health condition such as high blood pressure, type 2 diabetes, or abnormal lipid levels. In December 2022, this approval was extended to include adolescents aged 12 years and older with obesity.
- Social Media Testimonials: Since their launch, these medications have become extremely popular. Prescriptions for semaglutide-based products have skyrocketed due to impressive weight loss results reported in clinical studies and amplified through social media testimonials. This surge in demand has led to supply shortages throughout 2022 and 2023, prompting manufacturers to increase production capacity.
- Prescribed Off-Label for Weight Loss: Healthcare providers have also started prescribing these medications off-label for various weight management situations, further contributing to their widespread use among different patient groups seeking effective pharmaceutical solutions for obesity.
Conditions Associated with Painless Acute Vision Loss
- Acute Painless Vison Loss: A detailed medical history is crucial for differential diagnosis. Common causes of acute, painless vision loss include:
-
- Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION): The hallmark presentation is sudden, painless vision loss (often noticed upon waking) in one eye, typically with an altitudinal visual field defect. It is a common nonglaucomatous optic nerve disorder in patients over 50.
-
- Central Retinal Artery Occlusion (CRAO): This condition, a “painless” cause of vision loss, is due to a blockage of the main artery supplying the retina. It is often a medical emergency because it can indicate a broader issue with blood clots that might lead to a stroke or heart attack.
-
- Central Retinal Vein Occlusion (CRVO): Similar to CRAO, this involves a blockage in a retinal vein and typically presents as painless vision loss.
-
- Retinal Detachment: While some people might experience flashing lights or new floaters beforehand, the actual loss of vision (often described as a “curtain” or “shade” over the vision) is typically painless.
-
- Vitreous Hemorrhage: Bleeding into the vitreous gel that fills the eyeball can cause sudden vision loss that is painless.
-
- Amaurosis Fugax: This is temporary sudden vision loss that lasts for seconds to minutes and resolves spontaneously, often indicating a temporary disruption in blood flow to the retina. It requires immediate medical evaluation to identify the source of the blockage.

What is Nonarteritic Anterior Ischemic Optic Neuropathy (NAION)?
- Nonarteritic Anterior Ischemic Optic Neuropathy (NAION): Is the most common type of sudden optic nerve damage in people over 50. It happens when blood flow to the front part of the optic nerve is reduced, causing damage to the nerve fibers that carry visual signals from the eye to the brain.
- Wegovy and Vision Loss: This lack of oxygen supply leads to death of the nerve tissue and loss of vision.
NAION Symptoms and Clinical Presentation
- Painless Vision Loss: The main sign of NAION is sudden, painless vision loss that usually occurs upon waking up. Patients often notice this vision problem when they first open their eyes in the morning. The extent of vision loss can vary widely among individuals:
-
- Sudden blurring or dimming of vision in one eye
- Loss of upper or lower part of the visual field (altitudinal visual field defects)
- Difficulty seeing clearly or recognizing faces (central vision impairment)
- Complete blindness in severe cases
- No eye pain or discomfort during the acute phase
Risk Factors and Vulnerable Populations
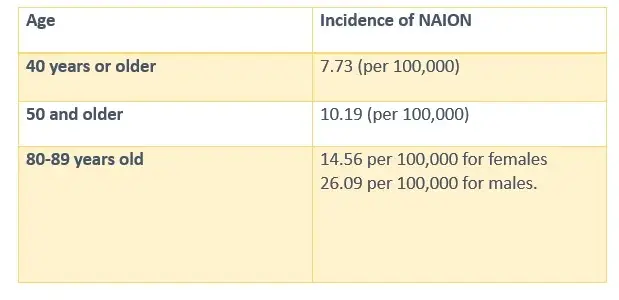
- Certain demographic and medical factors make some people more likely to develop NAION. Age is a major risk factor, with rates increasing significantly after age 50. People with diabetes are at higher risk because the disease affects blood vessel health and circulation. Other conditions that may increase susceptibility include:
-
- High cholesterol levels and hardening of arteries (atherosclerosis)
NAION RATE OF INCIDENCE BY AGE
Permanent Nature of Vision Impairment
- NAION causes permanent damage to the optic nerve tissue. The lack of blood supply leads to death of the nerve fibers, which cannot regenerate or heal themselves. This means that any visual deficits caused by NAION will not improve over time.
- While some patients may experience slight improvement in their vision within a few weeks after onset, significant recovery is rare. In about 15% of patients, NAION may also affect the other eye within five years after the first event.
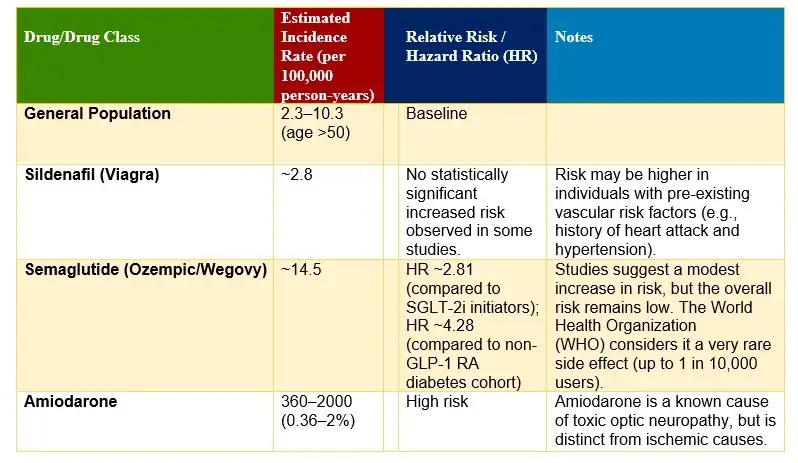
NAION Incidence Rates and Risk by Drug
Scientific Evidence Linking Wegovy and Vision Loss
- Observational Research: The connection between semaglutide-based medications and eye problems came from observational research done at Mass Eye and Ear, which is associated with Harvard Medical School.
- Link Between GLP-1 Drugs and NAION: This retrospective cohort study, published in JAMA Ophthalmology in July 2024, looked at medical records of over 16,800 patients treated at the facility from 2017 to 2023. The study found a significant link between the use of GLP-1 receptor agonists and an increased occurrence of NAION.
Key Findings from Clinical Research
- Heightened Risk for Type 2 Diabetes: The Harvard study showed that patients prescribed semaglutide for type 2 diabetes had more than four times the risk of developing NAION compared to those taking other diabetes medications. The difference was even greater among individuals using semaglutide specifically for weight loss, where the risk increased by more than seven times compared to patients on non-GLP-1 weight loss treatments. These findings were the first substantial evidence connecting this class of medications to serious eye complications.
- Link to Diabetes Management: Researchers found 17 cases of NAION among 710 patients taking semaglutide for diabetes management, while only six cases were found among 2,407 patients using other diabetes medications. For weight management, the study reported 20 NAION cases among 979 patients prescribed semaglutide, compared to three cases among 2,331 patients receiving different weight loss interventions.
FDA Safety Data on Weight Loss Drugs Eye Damage
- Documented Wegovy Vision Side Effects: The FDA Adverse Event Reporting System (FAERS) database contains many reports documenting vision-related issues linked to Wegovy and Ozempic. These reports include instances of sudden vision loss, blurred vision, and diagnosed NAION occurring shortly after starting semaglutide treatment.
- FDA Heightened Alert to Wegovy and Vision Loss: The growing number of adverse event reports has led regulatory authorities and healthcare providers to pay closer attention to the eye safety profile of GLP-1 receptor agonists.
Studies on Semaglutide and NAION
- Ischemic Events Affecting the Optic Nerve: Other studies have looked into the possible biological reasons behind the connection between semaglutide use and damage to the optic nerve. Researchers have suggested that changes in blood flow to the optic nerve head, combined with metabolic alterations caused by GLP-1 receptor agonists, may lead to ischemic events affecting the optic nerve. The rapid weight loss associated with these medications could also impact blood vessel dynamics, potentially reducing blood supply to vulnerable eye tissues in susceptible individuals.
- Wegovy Blindness Lawsuit: Heavily relies on this emerging scientific evidence. Plaintiffs are using the Harvard study and FDA safety data as proof that Novo Nordisk did not adequately inform patients and healthcare providers about the risk of vision loss linked to semaglutide-based treatments.
Legal Landscape: The Wegovy Blindness Lawsuit
- Wegovy Blindness Lawsuit Legal Landscape: The emergence of scientific evidence connecting semaglutide-based medications to vision complications has led to a surge of legal action against weight loss drug manufacturers.
- Failure to Warn: Multiple lawsuits have been filed against Novo Nordisk, the pharmaceutical company responsible for manufacturing Wegovy and Ozempic, with plaintiffs claiming the company failed to adequately warn patients and healthcare providers about the risk of developing NAION.
Primary Legal Claims
Plaintiffs pursuing legal action against Novo Nordisk have centered their claims on several key allegations:
- Failure to warn: The manufacturer did not provide sufficient information about the potential risk of NAION and other serious vision complications on product labeling or through direct communication with medical professionals
- Inadequate testing: Claims that pre-market clinical trials failed to identify or properly investigate the connection between semaglutide use and optic nerve damage
- Negligent misrepresentation: Allegations that the company promoted Wegovy’s safety profile without disclosing known or discoverable risks to ocular health
- Design defect: Arguments that the medication’s formulation presents unreasonable dangers that outweigh its therapeutic benefits for certain patient populations
Legal Representation and Case Development
- Wegovy Blindness Lawsuit: Law firms have begun accepting cases from individuals who experienced Wegovy and Vision Loss or similar GLP-1 receptor agonists. The Law Offices of Timothy L. Miles and other firms focusing on pharmaceutical litigation have established dedicated practices to represent affected patients. These Wegovy blindness lawyers are building cases by collecting medical records, ophthalmologic evaluations, and documentation of the temporal relationship between medication use and vision deterioration.
- Law Offices of Timothy L. Miles: If you suffered Wegovy vision problems, contact the Law Offices of Timothy L. Miles for a free case evaluation as you may be eligible for a Wegovy Blindness Lawsuit and possibly entitled to significant compensation. (855) 846–6529 or [email protected].
- The Legal Proceedings: Involve patients who suffered varying degrees of Wegovy vision problems, from partial visual field defects to complete blindness in one or both eyes. Many plaintiffs report that their vision loss occurred suddenly and without prior warning, fundamentally altering their ability to work, drive, and maintain independence in daily activities.
Patient Experiences and Testimonials with Wegovy Vision Problems
- Patient Testimonials Vision Loss Weight Loss Drugs: Reveal the profound personal toll of unexpected Wegovy vision side effects. Individuals who started treatment with semaglutide-based medications for weight management report experiencing sudden and alarming changes to their vision, often without any warning or proper information about potential risks.
- One patient described waking one morning to discover complete vision loss in their left eye after several months of Wegovy treatment. The sudden onset occurred without pain, leaving the individual disoriented and frightened. Despite immediate medical intervention, eye specialists confirmed permanent damage to the optic nerve consistent with NAION, effectively ending the patient’s career as a graphic designer and fundamentally altering their independence.
- Another account details a 52-year-old individual who noticed progressive blurring in their peripheral vision while using the medication for diabetes management and weight reduction. Within weeks, the condition worsened to significant visual field defects that made driving impossible. The patient reports experiencing:
-
- Inability to perform routine tasks requiring depth perception
- Loss of employment due to vision-dependent job requirements
- Psychological distress including anxiety and depression
- Financial strain from medical expenses and lost income
- Dependence on family members for daily activities
- Wegovy Vision Problems: Extend beyond complete blindness to include various degrees of visual impairment that significantly reduce quality of life. Patients describe struggling with reading, recognizing faces, and navigating unfamiliar places. The permanence of NAION-related damage adds to the emotional burden, as affected individuals face irreversible changes to their eyesight.
- Failure to Warn: Medical records submitted in legal proceedings document cases where patients specifically asked about potential side effects before starting treatment but received no information regarding eye risks. These documented exchanges highlight allegations that healthcare providers and pharmaceutical manufacturers failed to properly communicate the possibility of vision-threatening complications associated with GLP-1 receptor agonist therapy.
Regulatory Responses to Emerging Evidence Linking GLP-1 Receptor Agonists with Eye Disorders
- Regulatory Monitoring: The FDA has been actively monitoring reports of adverse events related to semaglutide-based medications since they were approved.
- Safety Data Review: After research was published suggesting a possible link between GLP-1 receptor agonists and NAION, the FDA began conducting thorough reviews of safety data collected after the drugs were on the market. The goal is to understand the extent and seriousness of reported eye problems.
FDA Drug Safety Communications and Monitoring
- FDA’s Adverse Event Reporting System (FAERS) Database: Has recorded many cases of Wegovy vision side effects as well as Ozempic, and similar medications. These reports have led the agency to take several actions:
-
- Conduct detailed analyses of temporal relationships between medication initiation and vision loss onset
- Evaluate the biological plausibility of proposed mechanisms linking GLP-1 receptor agonists to optic nerve damage
- Coordinate with pharmaceutical manufacturers to assess clinical trial data for previously unidentified safety signals
- Consider potential label modifications to reflect emerging safety information
Pharmaceutical Manufacturer Obligations
- Novo Nordisk, the manufacturer of Wegovy and Ozempic, is facing increased regulatory scrutiny regarding whether it adequately warns healthcare professionals and patients about potential risks. The company has submitted additional safety data to regulatory authorities while also defending its position in the Wegovy Blindness Lawsuit. Regulatory expectations require manufacturers to:
-
- Report serious adverse events within specified timeframes
- Update prescribing information when new safety data emerges
- Conduct post-marketing surveillance studies to identify rare complications
- Implement Risk Evaluation and Mitigation Strategies (REMS) when necessary
International Regulatory Coordination
- International Regulatory Bodies: The European Medicines Agency (EMA) and Health Canada are also reviewing the safety profiles of GLP-1 receptor agonists. These agencies work together through international networks focused on monitoring drug safety in order to share information about risks associated with these medications.
- FDA Drug Safety Alerts: When significant dangers are discovered, the FDA drug safety alerts system allows for quick communication of important details directly to healthcare providers.
How to Identify Symptoms and Seek Medical Help for NAION
- Recognizing The Symptoms of NAION: Is crucial in minimizing permanent vision damage for patients undergoing treatment with semaglutide-based medications. The condition usually presents itself through specific eye warning signs that require immediate medical attention.
Primary Symptoms Requiring Urgent Evaluation
- Patients using Wegovy or related GLP-1 receptor agonists should be vigilant about certain visual disturbances:
-
- Sudden, painless vision loss in one eye, occurring upon waking or developing rapidly throughout the day
-
- Blurred vision or clouding that does not improve with blinking or rubbing the eyes
-
- Visual field defects, especially loss of peripheral or central vision creating blind spots
-
- Change in color perception, where colors appear washed out or less vibrant
-
- Difficulty with depth perception affecting daily activities such as driving or reading
- The Painless Nature of NAION: Sets it apart from other eye emergencies. Patients often describe the experience as “a curtain coming down” over their vision or noticing significant changes immediately upon opening their eyes in the morning.
Critical Timeline for Medical Intervention
- Seek Immediate Medical Help: The opportunity for potential intervention becomes much smaller within hours of symptom onset. Patients experiencing any sudden Wegovy vision problems must contact an ophthalmologist or go to an emergency department without delay.
- Consult Retinal Specialist: Regular eye care providers may not have the specialized equipment needed for diagnosing NAION, making it necessary to consult with a neuro-ophthalmologist or retinal specialist for accurate assessment.
Diagnostic Procedures and Documentation
- Get a Comprehensive Medical Evaluation for NAION: Which includes comprehensive eye examination, optical coherence tomography (OCT), visual field testing, and fluorescein angiography. Patients should inform healthcare providers about their current use of semaglutide medications, dosage information, and treatment duration.
- Document Everything Related to Wegovy and Vision Loss: Documenting the timing relationship between medication use and symptom onset strengthens both medical records and potential legal claims. Keeping detailed records of all medical appointments, diagnostic test results, and treatment recommendations provides essential evidence for understanding the progression of vision impairment.
Legal Options for Affected Patients
- Medical Timeline Documentation: Patients who have experienced vision complications following Wegovy use have specific legal options to seek compensation for their injuries.
- Wegovy Blindness Lawsuit: The process of filing a Wegovy Blindness Lawsuit begins with documenting the medical timeline connecting semaglutide administration to the onset of visual symptoms.
Initial Steps for Potential Claimants
Affected individuals should gather comprehensive medical records demonstrating:
- Prescription history for Wegovy or related GLP-1 receptor agonists
- Ophthalmologic evaluations confirming NAION diagnosis or other vision impairments
- Documentation of symptom onset dates relative to medication use
- Records of all treatments attempted to address Wegovy vision problems
- Evidence of functional limitations resulting from vision loss
Consultation with Specialized Legal Counsel
- Contact an Experienced Wegovy Blindness Lawyer: Engaging attorneys experienced in pharmaceutical litigation provides essential guidance through complex legal proceedings. Law firms, such as the Law Offices of Timothy L. Miles practicing in defective drug cases evaluate the strength of individual claims by examining medical evidence, assessing damages, and determining liability theories.
- Contingency Fee Agreements: The Law Offices of Timothy L. Miles takes all cases on a contingency fee basis so it does not cost anything to hire a Wegovy Blindness Lawyer if you are eligible for a Wegovy Blindness Lawsuit. Call today for a free case evaluation. (855) 846–6529 or [email protected].
- Individual Lawsuits Versus Multidistrict Litigation
- Wegovy Blindness Lawsuit: Patients may pursue individual claims or participate in consolidated legal actions. Individual lawsuits allow for personalized case development addressing specific circumstances and damages.
- Multidistrict litigation (MDL): consolidates similar claims for pretrial proceedings, streamlining discovery and potentially expediting resolution while preserving individual trial rights.
Potential Compensation Categories
Successful claims may recover damages for:
- Medical expenses including emergency care, ongoing treatment, and vision rehabilitation
- Lost wages and diminished earning capacity
- Pain and suffering associated with permanent vision impairment
- Loss of quality of life and inability to perform daily activities
- Punitive damages if evidence demonstrates willful misconduct by manufacturers
Conclusion
- Wegovy Blindness Lawsuit: Highlights the critical need for comprehensive Wegovy eye damage awareness among patients and healthcare providers. As evidence continues to emerge regarding Wegovy vision side effects and potential Wegovy eye damage, vigilance remains paramount for all individuals using semaglutide-based medications.
- Remain Proactive: Patients must maintain proactive communication with their ophthalmologists and prescribing physicians, reporting any visual disturbances immediately. The connection between GLP-1 receptor agonists and NAION demands heightened scrutiny from both medical professionals and regulatory agencies. Staying informed about evolving research, FDA updates, and legal developments empowers patients to make educated decisions about their treatment options.
- Lack of Transparency: The pharmaceutical industry bears responsibility for transparent disclosure of all potential adverse effects, including rare but serious complications affecting vision. As litigation progresses and scientific understanding deepens, the medical community must prioritize patient safety through rigorous monitoring protocols and comprehensive informed consent processes. Knowledge serves as the most effective tool for protecting vision health while pursuing weight management goals with these medications.
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com