Introduction to Zepbound and NAION: 1 Permanently Impairing Side Effect
Welcome to the Zebound and NAION authoritative analysis. Zepbound (tirzepatide) is reshaping how clinicians and patients approach chronic weight management. It is also reshaping expectations. When a medication reliably produces meaningful weight loss, it becomes easy to focus on the primary outcome and overlook low-frequency but high-severity harms.
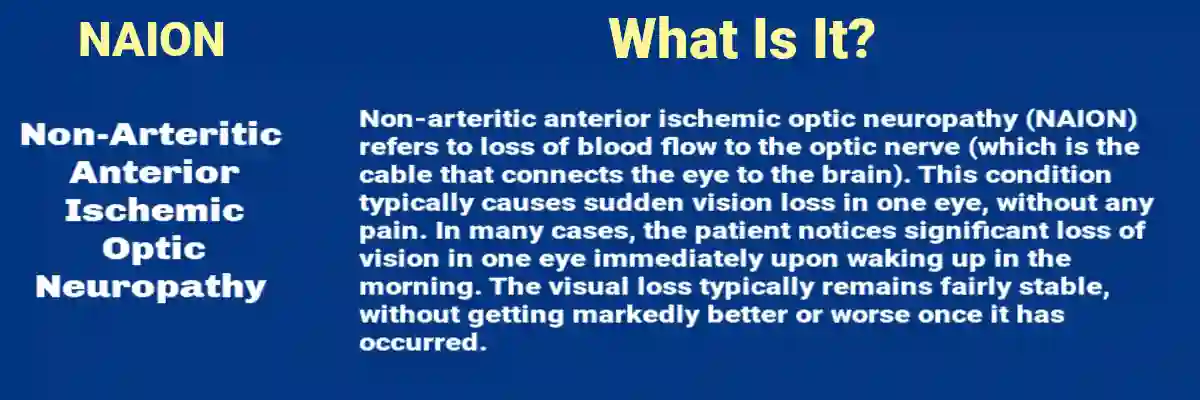
One potential harm that demands a more disciplined discussion is NAION, short for non-arteritic anterior ischemic optic neuropathy. NAION is not a cosmetic issue. It is not a “temporary eye symptom.” It is a form of optic nerve injury that can lead to permanent vision loss, often without warning.
This article explains what NAION is, why it matters in the context of Zepbound, what risk factors are known, what symptoms warrant urgent action, and how proactive governance in prescribing and monitoring can reduce preventable harm.
What Zepbound Is, and Why Safety Conversations Must Evolve With Its Adoption
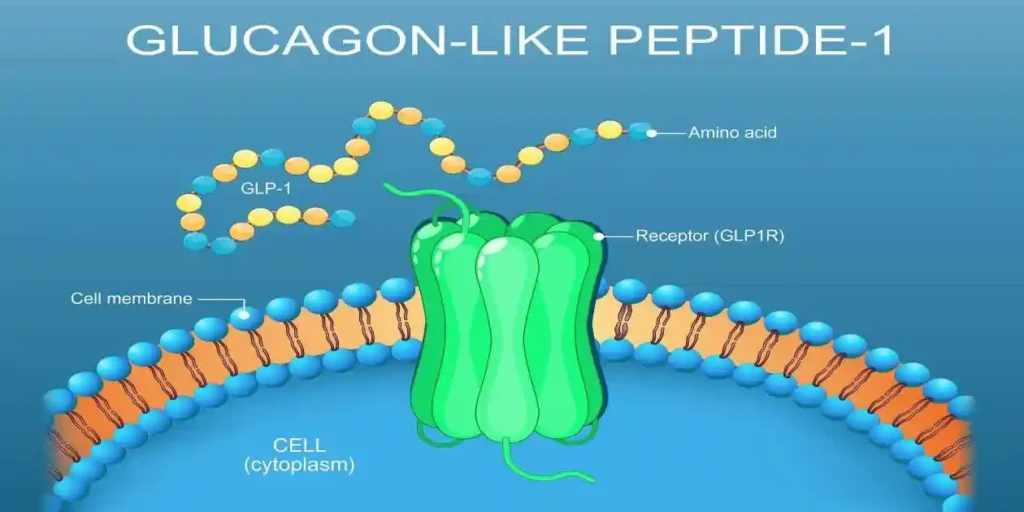
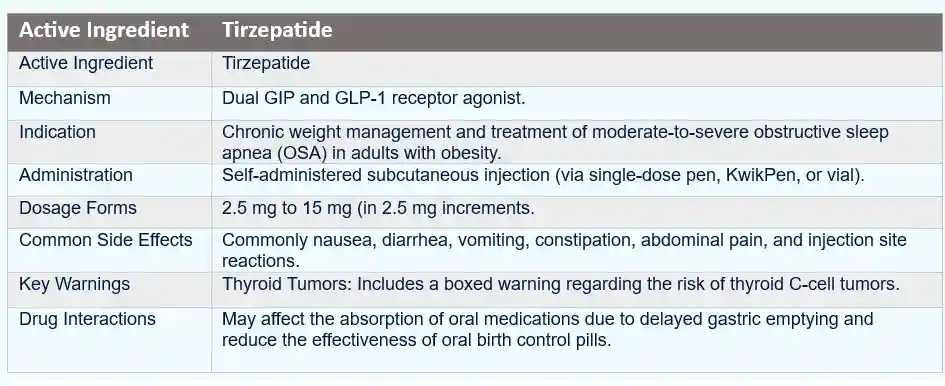
Zepbound is a branded formulation of tirzepatide, a medication that acts on GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptors. This dual incretin activity can improve glycemic regulation and promote weight loss through multiple mechanisms, including appetite suppression, slowed gastric emptying, and changes in energy balance.
As utilization expands, safety conversations must evolve in parallel. That is not a critique of innovation. It is the responsible lifecycle of innovation.
For any widely prescribed drug, the central governance question becomes consistent:
- What adverse events are plausible based on mechanism, physiology, and class effects?
- What adverse events are rare but severe enough to justify specific warnings and workflows?
- What monitoring and escalation pathways reduce time-to-diagnosis and improve outcomes?
NAION fits squarely into the “rare but severe” category because the functional impact can be permanent. Given the serious nature of this condition, it’s crucial for patients and healthcare providers to remain vigilant about potential eye side effects associated with Zepbound. Furthermore, understanding the possible legal implications related to NAION as a result of Zepbound usage could provide additional insights into this matter.
In light of these potential risks, patients may wonder about the safety of continuing their use of GLP-1 drugs like Zepbound. It’s important to consult with healthcare professionals regarding such concerns. For instance, the American Academy of Ophthalmology has provided guidance on whether one should stop taking GLP-1 drugs like Ozempic, which could offer valuable insights for Zepbound users as well.
NAION, Defined: A Sudden Optic Nerve Stroke-Like Event
Non-arteritic anterior ischemic optic neuropathy (NAION) is the most common acute optic neuropathy in older adults. It is often described as a “stroke of the optic nerve,” though the pathophysiology is more specific: NAION occurs when blood flow to the anterior portion of the optic nerve head becomes insufficient, leading to ischemic injury.
Key characteristics clinicians commonly emphasize include:
- Sudden, painless vision loss, typically in one eye
- Altitudinal visual field defect (loss in the upper or lower half of vision)
- Optic disc swelling on exam in the acute stage
- Variable recovery, with many patients left with persistent deficits
There is no guaranteed reversal. Some individuals experience partial improvement, but many have permanent impairment, particularly in visual field function. Importantly, the fellow eye can be affected later, meaning a first event can place a patient in a higher stakes risk category moving forward.
NAION is also clinically distinct from other optic nerve conditions:
- It is non-arteritic, meaning it is not caused by giant cell arteritis (which requires immediate steroid therapy).
- It is anterior, meaning it involves the optic nerve head (visible swelling early on).
- It is ischemic, meaning blood supply compromise is central to injury.
The outcome burden is not theoretical. NAION can impair reading, driving, occupational function, and independent living.
In recent years, there have been emerging concerns about certain medications potentially triggering NAION. For instance, Saxenda, a popular weight loss drug, has been linked to cases of NAION. Similarly, Mounjaro, another weight management medication, has also raised alarms due to its association with this serious eye condition.
Moreover, if you or someone you know has experienced vision loss following the use of Zepbound, it’s crucial to consult with a Zepbound vision loss lawyer who can provide guidance and support through this challenging time. There are also resources available for those facing Zepbound-related eye problems, including information on who is eligible for a Zepbound vision loss lawsuit.

Why NAION Is a Different Kind of Side Effect: Low Frequency, High Severity, High Permanence
Many medication side effects are unpleasant but reversible. NAION is different because it can be:
- Acute
- Functionally disabling
- Permanently impairing
- Time-sensitive for evaluation
- Difficult to treat once established
This makes NAION the kind of risk that should be handled with proactive disclosure and structured escalation, not casual reassurance.
From a governance lens, a “rare” event is not automatically a “negligible” event. In safety management, severity and reversibility carry weight equal to frequency. That is why aviation, manufacturing, and healthcare quality systems prioritize “sentinel events,” even when they are uncommon.
NAION belongs in that mindset.
Is NAION Proven to Be Caused by Zepbound?
The careful, accurate answer is that a definitive causal relationship cannot be asserted from public discussion alone. Individual case reports, pharmacovigilance signals, and post-marketing observations can raise concern about the worst vision side effects of Zepbound, but causality requires structured evaluation, including:
- Baseline incidence rates in relevant populations
- Confounder adjustment (diabetes, hypertension, sleep apnea, dyslipidemia)
- Temporal association and dechallenge patterns
- Mechanistic plausibility
- Consistency across datasets
However, clinical prudence does not require perfect certainty before acting on a high severity signal. Prudent practice requires that clinicians and patients understand the condition, recognize symptoms early, and treat sudden vision loss as urgent, regardless of whether a specific drug is the ultimate cause.
In other words, the best immediate objective is not winning a causality debate. The best immediate objective is preventing delayed diagnosis and reducing avoidable harm.
Mechanistic Plausibility: How Could a Weight Loss Medication Intersect With Optic Nerve Perfusion?
NAION is fundamentally an optic nerve perfusion problem. Anything that meaningfully shifts perfusion pressure, blood viscosity, vascular regulation, or systemic hemodynamics could, in theory, intersect with risk in susceptible individuals.
Potential pathways clinicians consider in similar contexts include:
- Blood pressure changes, including nocturnal hypotension
- NAION has been associated in some literature with low nighttime blood pressure. If a patient experiences lower baseline pressures or takes antihypertensives at night, optic nerve perfusion may drop during sleep.
- Volume depletion and dehydration
- Gastrointestinal adverse effects such as nausea, vomiting, or reduced intake can contribute to dehydration. Dehydration can reduce circulating volume and potentially affect perfusion, particularly in those with vascular disease.
- Rapid metabolic shifts and vascular tone changes
- Significant weight loss and improved glycemic control can change autonomic tone and cardiovascular parameters. These changes are often beneficial overall, but they alter the physiologic baseline.
- Underlying risk clustering in the treated population
- Zepbound is used in populations where NAION risk factors are already more prevalent, including obesity, insulin resistance, hypertension, dyslipidemia, and sleep apnea.
None of these points prove causation. They explain why the question is clinically legitimate and why clinicians should treat NAION education as a safety essential rather than an optional aside.
Who Is at Higher Risk for NAION?
NAION risk is multifactorial. Many cases occur in individuals with overlapping vascular and anatomical risk factors.
Commonly cited risk factors include:
- Age over 50
- Hypertension
- Diabetes mellitus
- Hyperlipidemia
- Obstructive sleep apnea
- Smoking
- Nocturnal hypotension
- “Crowded” optic disc anatomy (often described as a “disc at risk,” meaning a small cup-to-disc ratio)
This is where proactive screening becomes practical. Many Zepbound candidates already have one or more of these risk factors. That does not mean they should not use the medication. It means risk should be contextualized, documented, and monitored with the seriousness proportional to potential harm.
TIRZEPATIDE SAFETY DRUG PROFILE
The Symptom Pattern That Requires Immediate Action
If there is one operational takeaway for patients, it is this:
Any sudden change in vision should be treated as an emergency.
NAION typically presents as:
- Sudden vision loss in one eye, often noticed on waking
- A “shadow,” “curtain,” or missing portion of the visual field
- Blurred or dim vision
- Reduced color saturation in the affected eye
- Usually painless
Patients may underestimate it because it can be partial. They may still see “well enough” to work around it. That delay is dangerous because the differential diagnosis includes conditions that can require urgent treatment, including:
- Giant cell arteritis (vision- and life-threatening)
- Retinal detachment
- Central retinal artery occlusion
- Optic neuritis
- Acute angle-closure glaucoma
- Stroke-related visual pathway events
The correct pathway is not “wait and see.” The correct pathway is urgent evaluation by emergency services or immediate ophthalmology or neuro-ophthalmology assessment.
In some instances, Zepbound has been linked to serious vision problems, including sudden vision loss which requires immediate medical attention. If you are experiencing such symptoms after starting Zepbound, it’s crucial to seek help right away. Keep in mind that there are ongoing legal discussions regarding Zepbound and its associated vision loss, so staying informed about your health and any potential side effects of medication is key.
What to Do If You Are on Zepbound and Notice Sudden Vision Changes
If you are currently using Zepbound (or any incretin-based therapy) and you experience sudden visual symptoms, such as eye floaters, it’s crucial to act swiftly:
- Seek urgent medical care immediately.
- Do not wait for a routine appointment. Do not self-diagnose.
- Tell the evaluating clinician what medication(s) you are taking, including Zepbound, dose, start date, and any recent dose changes. This is important as there have been reported cases linking Zepbound to serious eye problems.
- Report associated factors that influence perfusion and risk, such as: recent vomiting, poor intake, dehydration, lightheadedness or fainting, blood pressure readings (especially if low), new or intensified antihypertensive therapy, and sleep apnea symptoms or CPAP nonadherence.
- Do not stop critical medications on your own, but do communicate urgency. Medication decisions should be made with a clinician who can weigh systemic risks and benefits.
This is not about panic. It is about time-sensitive triage.

Clinical Evaluation: What Typically Happens Next
In suspected NAION (Non-Arteritic Anterior Ischemic Optic Neuropathy), clinicians typically focus on confirming diagnosis and excluding arteritic causes. Evaluation often includes:
- Comprehensive ophthalmologic exam with optic disc assessment
- Visual field testing
- Optical coherence tomography (OCT) to assess optic nerve head and retinal nerve fiber layer
- Blood pressure assessment and cardiovascular risk review
- Laboratory testing if giant cell arteritis is suspected (commonly ESR, CRP, platelet count), and possible temporal artery evaluation based on clinical presentation
Treatment options for NAION are limited, and practice varies. The most important immediate objective is accurate diagnosis and risk control to reduce the chance of additional injury, including involvement of the other eye.
It’s also worth noting that there have been ongoing discussions about potential lawsuits related to Zepbound due to these serious side effects. If you experience significant vision problems after starting Zepbound, you might want to consider seeking legal advice regarding the Zepbound blindness lawsuit which could provide further insights into your situation.
The Governance Gap: Why Many Patients Never Hear About NAION Until After It Happens
In real-world care, many adverse event conversations are shaped by time constraints, patient cognitive load, and the desire to avoid alarming people. Those pressures are understandable. They are also exactly how high severity events become “surprises.”
Robust medication governance requires:
- Consistency in disclosure
- Clarity in written and verbal education
- Defined escalation instructions
- Documentation of counseling
- Feedback loops when adverse events occur
In other words, a serious but uncommon risk needs a standardized workflow. Without that workflow, disclosure becomes dependent on individual clinician habits, and outcomes become dependent on patient luck.
If an organization wants to demonstrate integrity, it does not hide behind rarity. It operationalizes readiness.
Practical Risk Reduction While Using Zepbound
Risk reduction of Zepbound eye issues should be framed as reasonable controls, not fear-based restrictions. Many actions are aligned with standard cardiovascular and metabolic best practices.
1) Prioritize hydration and illness planning
If nausea or reduced intake is significant, dehydration becomes more likely. Patients should have a clear plan for:
- Maintaining fluid intake
- Recognizing dehydration (dry mouth, dizziness, reduced urination)
- Knowing when to seek care for Zepbound and persistent vomiting or other seriousZepbound vision problems
2) Monitor blood pressure, especially if also on antihypertensives
Weight loss and metabolic improvement can lower blood pressure over time. That is usually beneficial. It also means medication regimens may need reassessment.
Patients using antihypertensives should ask a targeted question:
- “Should we adjust my blood pressure medication as my weight decreases?”
3) Treat sleep apnea as a serious comorbidity associated with Zepbound and NAION, not a side note
Obstructive sleep apnea is a known NAION risk factor. If a patient has suspected or diagnosed sleep apnea:
- Screening should be current
- CPAP adherence should be reinforced
- Symptoms should be re-evaluated as weight changes
4) Reduce modifiable vascular risks associate with Zepbound and NAION
Smoking cessation, lipid control, and diabetes management are not generic advice. They are optic nerve protection strategies in susceptible individuals.
5) Establish an eye symptom escalation plan for Zepbound vision problems
Patients should know exactly what to do and where to go if vision changes occur, especially in light of potential side effects from medications like Zepbound. This should be written, not improvised.
Questions Patients Should About Zepbound Vision Side Effects Prior to Taking Medication
Patients do not need a medical degree to participate in risk management. They need permission and a structured script.
Here are practical questions that clarify decision quality:
- “What are the most serious adverse events I should watch for, even if rare?” Zepbound has been associated with serious vision problems, which patients should be aware of.
- “If I have sudden vision changes, where should I go immediately?” It’s crucial to have a plan as sudden vision changes can occur.
- “Do I have NAION risk factors such as sleep apnea, hypertension, diabetes, or smoking?”
- “Should we monitor my blood pressure more closely as I lose weight?”
- “If I develop severe nausea or poor intake, what is the plan to prevent dehydration?”
These questions do not undermine treatment. They strengthen treatment.

What Clinicians and Practices Should Standardize Now Regarding Zepbound Eye Issues
For prescribers and healthcare organizations, the forward-looking goal is to reduce variability. When the drug is common, the process must be industrialized in the best sense of the term: consistent, auditable, patient-centered, and resilient.
A pragmatic standard could include:
- A written adverse event sheet that explicitly includes sudden Zepbound and vision loss as an emergency symptom
- Counseling documentation embedded in the EHR
- Baseline assessment of risk factors for Zepbound and NAION such as sleep apnea and hypertension control
- A plan for blood pressure reassessment after meaningful weight loss
- Clear referral pathways to ophthalmology or emergency evaluation for acute Zepbound eye issues
- Internal review of Zepbound eye problems and near misses to improve educational materials
Repetition for emphasis is appropriate here: rare does not mean negligible. rare does not mean ignorable. rare does not mean unmanageable.
In addition to these considerations, it’s important to note that a new risk factor has been identified for potentially blinding conditions. This underscores the necessity of comprehensive risk assessment and management in clinical practice.
The Bottom Line: Zepbound and NAION
Zepbound can be an effective tool for chronic weight management, and its benefits can be substantial. At the same time, Zepbound and NAION is a potentially permanently impairing optic nerve event that warrants explicit education and an immediate-action plan, as do other serious Zepbound eye issues.
The most responsible posture is proactive:
- Define the Zepbound and NAION risk clearly.
- Identify higher-risk patients thoughtfully.
- Monitor blood pressure and hydration realistically.
- Treat sudden vision changes as urgent, every time.
- Standardize counseling so safety does not depend on chance.
In modern pharmacotherapy, success is not only measured by pounds lost or biomarkers improved. Success is measured by outcomes achieved without preventable harm, supported by governance that is structured, transparent, and prepared. This includes understanding the potential side effects of medications like Zepbound, which can be found in resources such as this comprehensive guide on NAION.
Frequently Asked Questions about Zepbound and NAION and other Zepbound Vision Side Effects
What is Zepbound (tirzepatide) and how does it work for weight management?
Zepbound is a branded formulation of tirzepatide, a medication that acts on GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptors. This dual incretin activity improves glycemic regulation and promotes weight loss through mechanisms such as appetite suppression, slowed gastric emptying, and changes in energy balance although it comes with a host of Zepbound vision problems.
What is Zepboud and NAION and why is it a concern for patients?
NAION, or non-arteritic anterior ischemic optic neuropathy, is a sudden optic nerve injury caused by insufficient blood flow to the anterior optic nerve head. It can lead to painless, sudden vision loss often without warning and may result in permanent impairment. NAION is a rare but severe potential side effect linked to medications like Zepbound, making vigilance essential.
What are the symptoms of Zepbound and NAION that require urgent medical attention?
Symptoms of NAION include sudden, painless vision loss in one eye, altitudinal visual field defects (loss of upper or lower half of vision), and optic disc swelling during the acute phase. Any sudden change in vision should be treated as a medical emergency requiring immediate evaluation.
Are there known risk factors for developing Zepbound and NAION while taking weight loss drugs?
While specific risk factors vary, patients with pre-existing optic nerve issues or vascular risk factors may be at higher risk. Since NAION can affect the fellow eye later on, a first event increases future risk. Patients should discuss their individual risk profile with healthcare providers before starting or continuing Zepbound.
How should healthcare providers monitor patients on Zepbound to reduce preventable harm from Zepbound and NAION?
Proactive governance includes educating patients about potential eye side effects, promptly investigating any reported vision changes, and establishing clear monitoring and escalation pathways to reduce time-to-diagnosis. Early detection improves outcomes and helps mitigate permanent vision loss risks associated with NAION.
What legal options exist for patients who experience vision loss potentially linked to Zepbound Vision Side Effects?
Patients who suffer vision loss following Zepbound usage may consider consulting attorneys such as Timothy L. Miles who aare experienced in handling cases related to drug-induced eye injuries. Legal guidance can provide insights into rights and possible compensation related to adverse effects such as Zepbound and NAION.

If You Suffered from Zepbound and NAION or Other Zepboound Vision Problems, Contact Zepbound Vision Loss Lawyer Timothy L. Miles Today
If you were prescribed Zepbound and took it as directed and suffered Zepbound vision loss or other Zepbound eye problems, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com
Facebook Linkedin Pinterest youtube