
What Is Dupixent?
- Dupixent (dupilumab): Is an injectable biologic medication that is used to treat particular types of inflammatory diseases.
- Mechanism of Action: It works by treating the underlying cause of inflammation and is commonly prescribed for people with atopic dermatitis, eczema, asthma, chronic sinusitis, and eosinophilic esophagitis.
- Recent studies on Dupixent and Cancer: Have shown that Dupixent use may increase the risk of certain types of cancers known as Cutaneous T-cell Lymphomas (CTCL), including its subtypes Mycosis fungoides and Sezary syndrome, by as much as 300% or more in people who used the medication.
- Potential Compensation: People who used Dupixent and were later diagnosed with CTCL may be eligible to file a Dupixent Cancer Lawsuit and potentially entitled to substantial compensation. If you suffered Dupixent and Cancer call Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation. (855)-846-6529 or [email protected].
The Dupixent Common Side Effects Profile
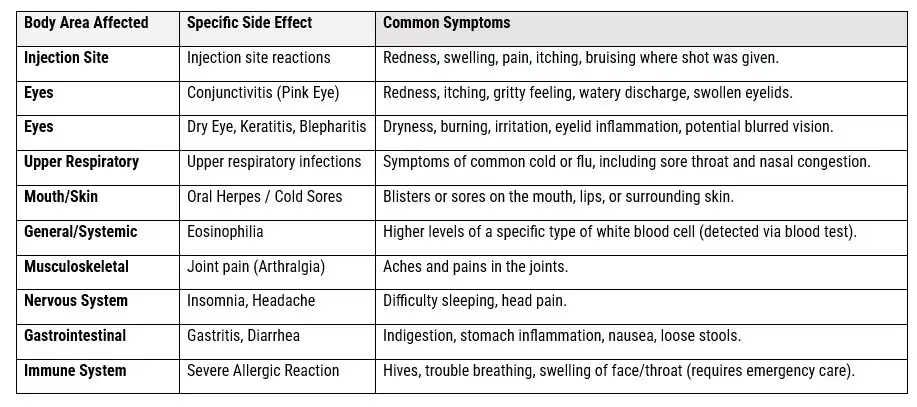
Safety And Efficacy Data of Dupixent Side Effects from Clinical Trials
- Dupixent Side Effects: Were studied in 2 clinical trials with 1,874 adult patients in total with inadequately controlled COPD and a high number of blood eosinophils.
- Clinical Trials: Safety and efficacy data of Dupixent were evaluated in 2 clinical studies involving 1,874 adult patients in total.
- Common Dupixent Side Effects: Both of the results from the trials are illustrated in the table below. Side effects occurred in at least 2% of patients and more frequently with Dupixentthan with placebo,

Safety Information and Indication
Do Not Use: If you are allergic to dupilumab or to any of the ingredients
Before Use: Tell Your Doctor about all your medical conditions, including if you:
- have eye problems.
- have a parasitic (helminth) infection.
- are scheduled to receive any vaccinations. You should not receive a “live vaccine” right before and during treatment with DUPIXENT.
- are pregnant or plan to become pregnant. It is not known whether DUPIXENT will harm your unborn baby.
- A pregnancy registry for women who take DUPIXENT during pregnancy collects information about the health of you and your baby. To enroll or get more information call 1-877-311-8972 or go to https://mothertobaby.org/ongoing-study/dupixent/.
- are breastfeeding or plan to breastfeed. It is not known whether DUPIXENT passes into your breast milk.
Tell Your Doctor: About all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Especially Tell Your Healthcare Provider: If you are taking oral, topical, or inhaled corticosteroid medicines or have COPD and also have asthma. Do not change or stop your other medicines, including corticosteroid medicine or other asthma medicine, without talking to your healthcare provider. This may cause other symptoms that were controlled by those medicines to come back.

Serious Dupixent Side Effects
Serious Dupixent Side Effects: The medication can cause serious side effects, including:
- Allergic reactions. Dupixent can cause allergic reactions, including skin reactions, that can sometimes be severe. Stop using DUPIXENT and tell your healthcare provider or get emergency help right away if you get any of the following signs or symptoms: breathing problems or wheezing, swelling of the face, lips, mouth, tongue or throat, fainting, dizziness, feeling lightheaded, fast pulse, fever, hives, skin rash, including rash that looks like a bullseye or painful red or blue bumps under the skin, general ill feeling, itching, swollen lymph nodes, nausea or vomiting, joint pain, or cramps in your stomach area.
- Eye problems. Tell your healthcare provider if you have any new or worsening eye problems, including eye pain or changes in vision, such as blurred vision. Your healthcare provider may send you to an ophthalmologist for an exam if needed.
- Joint aches and pain. Some people who use Dupixent have had trouble walking or moving due to their joint symptoms, and in some cases needed to be hospitalized. Tell your healthcare provider about any new or worsening joint symptoms. Your healthcare provider may stop Dupixent if you develop joint symptoms.

Dupixent and Cancer
- Dupixent and its Intended Use: Dupixent is a prescription injection manufactured by Sanofi and Regeneron and first approved by the FDA in 2017. It is commonly prescribed for patients with:
-
- Moderate-to-severe eczema (atopic dermatitis)
- Asthma
- Cchronic sinusitis with nasal polyps
- eosinophilic esophagitis
- Dupixent And Cancer: In recent years, however, Dupixent® has been linked to cutaneous T-cell lymphoma (CTCL), a rare and serious form of non-Hodgkin lymphoma. Studies published in 2024 and 2025 suggest that Dupixent® patients may face a 300% higher risk of developing CTCL yet manufacturers never warned healthcare providers or patients about this risk.
- Contact a Dupixent Cancer Lawyer: If you took Dupixent and subsequently developed CTCL or other serious Dupixent side effects or just have general questions, call Dupixent Cancer Lawyer Timothy L. Miles today, free of charge, for a free case evaluation as you may be eligible for a Dupixent Cancer Lawsuit and possibly entitled to substantial compensation. (855) 846-6529 or [email protected].

Symptoms of CTCL
Symptoms of CTCL may include:
- Itching
- Red, scaly, or thickened patches of skin
- Peeling skin
- Discolored skin
- Raised bumps or nodules
- Skin sores or ulcers
- Hair loss
- Enlarged lymph nodes
Several medical reports and studies including the Nation Library of Medicine have suggested that Dupixent may be tied to an increased risk of CTCL.
-
- A study published on April 6, 2024 (Hasan et al., 2024), looked at whether people with atopic dermatitis (AD) who were treated with the drug dupilumab had a higher risk of developing cutaneous T-cell lymphoma (CTCL) compared to those who didn’t take the drug. In their first model, which adjusted for age only, they found that people who took dupilumab had a 300% higher risk of getting CTCL compared to those who didn’t (OR 4.10). Even after they adjusted for more factors—like sex, ethnicity, and race, and removed people who had taken certain other immune-suppressing drugs—the risk was still more than two times higher (OR 3.20).
-
- A second study using TriNetX data (Mandel et al., 2024) was published in August 2024 and followed a method similar to the first study by Hasan et al. The researchers excluded patients who had other inflammatory diseases or had taken biologic drugs that might be linked to lymphoma.After matching patients by age, race, and sex, they found that people with atopic dermatitis (AD) who were treated with dupilumab had a 350% higher risk of developing cutaneous T-cell lymphoma (CTCL) compared to those who weren’t treated with the drug (RR 4.59).
-
- In September 2025, a study led by Sheng-Kai Ma and his team looked at the patients in the United States with asthma who initiated dupilumab or the active comparator (combination therapy with inhaled corticosteroids (ICS) plus long-acting β-agonists (LABA), or ICS/LABA), between 2018 and 2024. After propensity score matching, dupilumab-treated patients were found to have a higher risk of lymphoma (54 versus 43 cases, hazard ratio (HR) 1.79, 95% CI 1.19-2.71), especially T-cell and natural killer (NK)-cell lymphomas (19 versus ≤10 cases, HR 4.58, 95% CI 1.82-11.53).
After adjusting for things like age, gender, income, other health problems, and medicine use, the risk of CTCL was more than 4.5 times higher in the dupilumab group. Dupilumab was also linked to an overall increased risk of any type of lymphoma. When the researchers looked only at patients who had taken their medications for at least 16 weeks, the risk numbers went up even more, especially for combined mature T and NK cell lymphomas, a broader group of cancers including CTCL and other subtypes, which was over 14 times higher for dupilumab users.
- Acceleration of cutaneous T-cell lymphoma following dupilumab administration
-
- A 2019 study published in the Nation Library of Medicine, Acceleration of cutaneous T-cell lymphoma following dupilumab administration, showed multiple patients saw CTCL symptoms accelerate while using dupilumab.
-
- Case reports warning that Dupixent may “unmask” or worsen hidden lymphomas mistaken for eczema
These findings have pushed doctors to re-evaluate how and when to prescribe Dupixent — especially when patients show unusual skin reactions that don’t improve.
Patient Reports of Cancer Linked to Dupixent
- Patient and Healthcare Reports on FAERS: In addition to the clinical research and studies, the U.S. Federal and Drug Administration’s (FDA), Adverse Event Reporting System (FAERS) included numerous patient reports and reports from healthcare providers linking Dupixent (dupilumab) to cancer, specifically to multiple types of T-cell lymphoma. FAERS is a vital database that regulators and researchers use to recognize potential drug risks requiring further investigation.
- Reports Confirm Clinical Research: As you will note by visiting FAERS from the button below, the current dashboard shows close to 300 reports of T-cell lymphoma cases among Dupixent patients, which includes confirmed reports of cutaneous T-cell lymphoma (CTCL) and other related and serious conditions. These patient and healthcare reports to the FDA continue to fuel concerns that was addressed in clinical studies, strengthening the need for doctors to closely monitoring patients taking Dupixent.
- The database shows the following type of cases have been reported by patients taking Dupixent or reported by healthcare providers:
-
- Cutaneous T-Cell Lymphoma
- Adult T-Cell Lymphoma/Leukemia
- T-Cell Lymphoma (general)
- CTCL, Stage IV
- CTCL, Stage III
- Angioimmunoblastic T-Cell Lymphoma
- CTCL, Stage I
- Recurrent T-Cell Lymphoma
- Lymphoma (unspecified)
- Anaplastic Large Cell Lymphoma (T- and null-cell types)
- Unclassifiable T-Cell Lymphoma
Volume of Reports: The vast volume of these reports only provides further evidence that Dupixent may be contributing to serious cancer risks in certain patients.
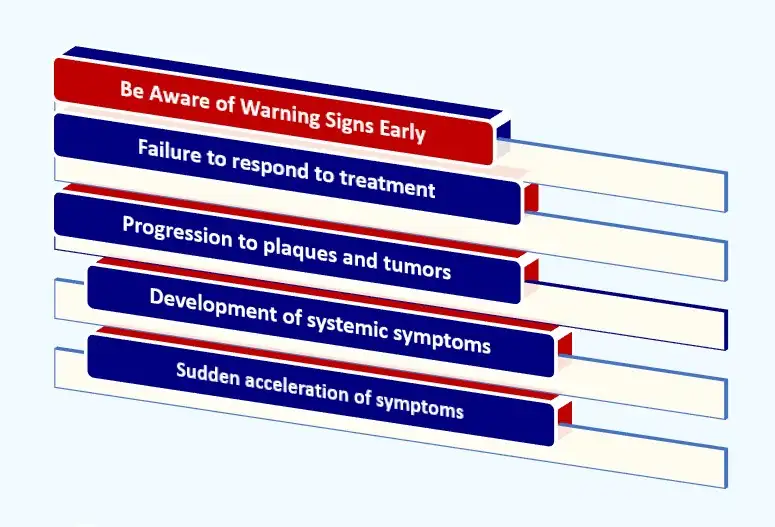
Key Warning Signs for Dupixent and Cancer
Severe Eczema Patients Be Vigilant: For individuals using Dupixent for severe eczema, the diagnostic challenge is compounded because the drug’s anti-inflammatory action may temporarily mask or alter the appearance of early CTCL symptoms.
- Be Aware of Warning Signs Early: Warning signs that may differentiate CTCL from typical eczema in Dupixent users include:
-
- Failure to respond to treatment: Rashes or lesions that persist, worsen, or spread despite ongoing Dupixent use should raise suspicion of an underlying condition.
-
- Progression to plaques and tumors: The development of thicker, raised lesions (plaques) or lumps/nodules (tumors) that can ulcerate is a sign of advancing CTCL not typically seen in standard eczema.
-
- Development of systemic symptoms: Advanced CTCL can lead to symptoms not associated with eczema, such as persistently swollen lymph nodes (in the neck, armpits, or groin), unexplained fatigue, or weight loss.
-
- Sudden acceleration of symptoms: In some case reports, patients experienced a rapid worsening of skin changes shortly after starting Dupixent, suggesting the drug may accelerate the progression of an undetected lymphoma.
Skin Changes and Seeking Immediate Medical Help: If you notice persistent or unusual skin changes while on Dupixent, it is crucial to seek a medical evaluation, including a skin biopsy reviewed by a specialist familiar with cutaneous lymphomas
Dupixent Lawsuit Status (as of November 2025)
- Dupixent Lawsuit: Individual lawsuits have been filed across the U.S., but they have not yet been consolidated into a formal multidistrict litigation (MDL).
- First Lawsuit: The first known Dupixent Lawsuit for wrongful death lawsuit was filed in a federal court in Tennessee in October 2025. The lawsuit alleges the drug caused a woman’s fatal T-cell lymphoma and that the manufacturers failed to provide adequate warnings.
- No MDL or Class Action: There is currently no formal multidistrict litigation (MDL) or class-action lawsuit. However, legal experts anticipate that cases may be consolidated into an MDL in late 2025 or early 2026 to streamline the legal process if more cases emerge.
- Allegations: Lawsuits primarily allege that the manufacturers, Sanofi and Regeneron, failed to adequately warn patients and healthcare providers about Dupixent and Cancer risks associated with the drug.
- FDA Investigation: The U.S. Federal and Drug Administration (FDA) is formally investigating the potential link and evaluating whether new cancer warnings should be added to the Dupixent label. The current label does not mention a CTCL risk.
- Settlements: No settlements have been reached in any Dupixent Lawsuit, and there are no jury verdicts, making it difficult to estimate potential settlement amounts.

Scientific Evidence and Regulatory Action
- FDA Investigation: The FDA is formally investigating the potential safety signal for CTCL linked to Dupixent use. This investigation began in March 2025.
-
- Cause of investigation: The FDA’s investigation was prompted by a significant number of adverse event reports linking Dupixent to cutaneous T-cell lymphoma (CTCL).
-
- Allegations: A Dupixent Lawsuit against the manufacturers claim they failed to adequately warn patients and doctors about this potential risk.
-
- Status: The FDA has not recalled Dupixent, but the investigation is formal and follows a pattern of regulatory actions that may include label revisions.
- Adverse Event Reports: The FDA’s Adverse Event Reporting System (FAERS) has received hundreds of reports of lymphoma among Dupixent users.
- Increased Risk Studies: Several peer-reviewed studies suggest an increased risk of Dupixent and Cancer.
Who is Eligible for a Dupixent Cancer Lawsuit?
Dupixent Cancer Lawsuit: You may be eligible if you:
- Used Dupixent (dupilumab) as prescribed
- Were later diagnosed with T-Cell Lymphoma, especially Cutaneous T-Cell Lymphoma
- Experienced serious Dupixent side effects or worsening symptoms
- Are within your state’s statute of limitations

Compensation in a Dupixent Cancer Lawsuit
If you are eligible for a Dupixent Cancer Lawsuit, a successful plaintiff is entitled to compensatory damages which would include compensation for things such as:
- past and future medical expenses;
- pain and suffering including emotional distress; and
- lost wages and the reduction in your ability to earn an income.
If your case proceeds to trial it is also possible the court could impose punitive damages, intended to punish the defendant for their actions. Contact Dupixent Cancer Lawyer Timothy L. Miles who can tell you if you are eligible for a Dupixent Cancer Lawsuit and can explain more about the type of damages you are entitled to receive. (855) 846–6529 or [email protected].

If I Was Diagnosed with Dupixent and Cancer, How Do I Get Started?
- Get a Free Case Evaluation: If you or a loved on developed Dupixent and Cancer after taking Dupixent, the most important thing to do now is get a free case evaluation from a Dupixent Cancer Lawyer to see if you qualify for a Dupixent Cancer Lawsuit. These are not typical cases any lawyer can litigate. Contact Dupixent Cancer Lawyer Timothy L. Miles who can tell you if you are eligible for a Dupixent Cancer Lawsuit and can explain more about what will happen next. (855) 846–6529 or [email protected].
- Medical Records Review: The earlier your records are reviewed, the stronger the foundation becomes, especially in cases where the biopsy history is limited or where the initial diagnosis was “eczema” that simply never responded the way it should have. A lawyer experienced in drug litigation can help secure the complete medical file, obtain an independent pathology review if necessary, and establish a clear timeline that courts and juries can follow.
- Time Is of the Essence: If you have a valid case, then it is vital to preserve your rights under the statute of limitations in your state. These deadlines are different in different states, but it normally starts running from the time of diagnosis or from the point when a reasonable person should have realized the connection between the diagnosis and the drug. Time is of the essence so give Dupixent Cancer Lawyer Timothy L. Miles a call today.

Contingency Fee Agreements: No Cost to Hire a Lawyer
- No Fee: It does not cost anything to hire a lawyer if you are eligible for a Dupixent Cancer Lawsuit. We take all cases on a contingency basis which means we do not get paid unless we win or settle your case.
- Talk with a Lawyer Free of Charge: A lawyer can explain the process of a Dupixent Cancer Lawsuit and answer any questions you may have free of charge. The call is free and so is the fee unless we will or settle your case, so call Dupixent Cancer Lawsuit Timothy L. Miles today to see if you may be entitled to significant compensation.

Contact Timothy L. Miles Today About a Dupixent Cancer Lawsuit
If you believe you qualify for a Dupixent Cancer Lawsuit, contact Dupixent Cancer Lawyer Timothy L. Miles today for a free case evaluation to see if you are eligible for a Dupixent Cancer Lawsuit and possible entitled to substantial compensation. 855/846-6529 or via e-mail at [email protected]. (24/7/365).
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com
Facebook Linkedin Pinterest youtube

