Introduction to Dexcom Recall Update
Welcome this this authoritative Dexcom Recall Update. If you use a Dexcom CGM or you are the parent or partner of someone who does, the word “recall” can hit like a brick. Not because you assume something is automatically dangerous. Sometimes a recall is just a labeling fix or a tiny manufacturing issue.
But with diabetes tech, even small issues feel big, because the whole point is trust. You are making dosing decisions, sleep decisions, driving decisions, and you are leaning on that number.
This is a consumer-focused update. It aims to clarify what recalls usually mean in plain English, how to check if you’re affected, what to do right now if you think you are, and how to protect yourself in the event of a from a Dexcom Device Malfunction while Dexcom Device Malfunctionand regulators sort out the details.
Quick note before we start: recalls change. Batches expand, notices get updated, and rumors move faster than facts. For anything urgent, always verify using Dexcom’s official recall page, your Dexcom account notifications, and the FDA recall database if you’re in the US. This article is meant to make you calmer and more prepared, not to replace official instructions.
If you or a loved one used a defective Dexcom device and suffered harm when your Dexcom device malfunctioned, contact Dexcom Recall Lawsuit Lawyer Timothy L. Miles for a free case evaluation today as you may be eligible for a Dexcom Dev

What a “Dexcom recall” actually means (and what it doesn’t)
A recall is basically a formal process to correct a problem with a product already out in the world that violates U.S. laws and regulations. In medical devices, it can be initiated by the manufacturer, requested by the regulator, or coordinated between both.
And here’s the part people miss.
A recall does not always mean:
- the device will injure you
- the device is “bad” as a whole
- you must stop using it immediately
However, if you’re facing challenges due to a recent recall, it is important to understand that there may be legal avenues available for compensation. For instance, compensation in a Dexcom recall lawsuit could potentially provide some relief if you are eligible for a Dexcom Device Recall Lawsuit.
A recall often means:
- a specific lot number or serial range might have an issue
- the issue might show up only under certain conditions
- the fix might be replacement, software update, extra instructions, or return of affected stock
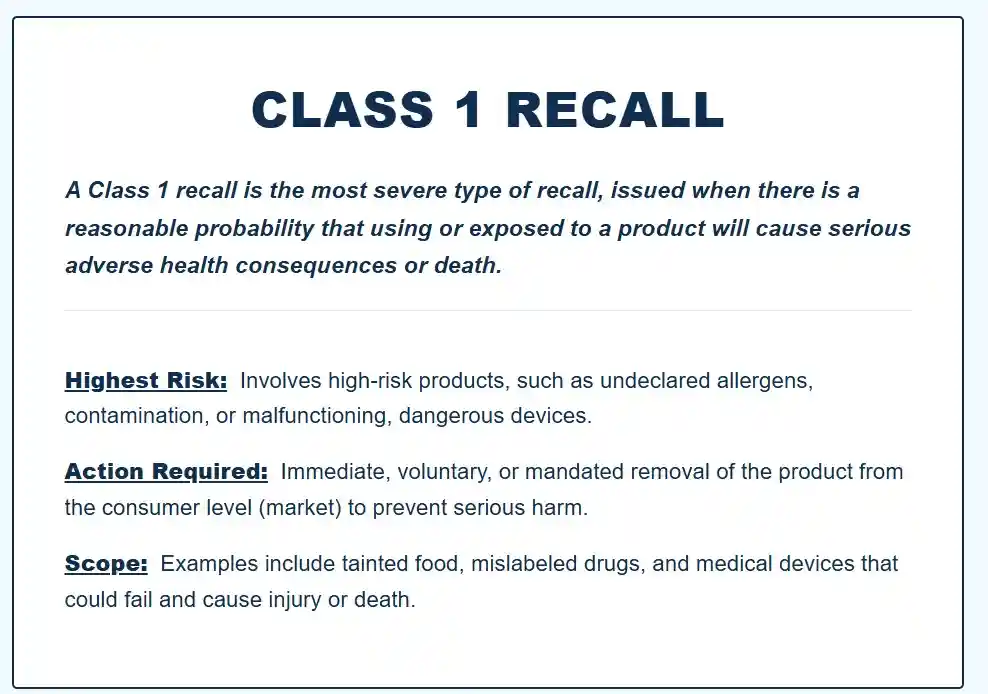
In the US, you’ll sometimes see recall classifications (Class I, II, III). Those classes aren’t about how many units are affected. They’re about the potential severity if the issue happens. Class I is the most serious category, but even then the practical action can vary. Sometimes it’s “stop using this lot now.” Other times it’s “be aware of false low readings and confirm with a fingerstick if symptoms don’t match.”
While understanding these classifications can help in managing expectations during such stressful times, it’s equally crucial to stay informed about any ongoing legal proceedings related to these recalls. For example, Dexcom device recall lawsuit updates can provide valuable insights into how such situations are being handled legally.
So the action is everything. Always read the “what to do” part. That’s where the truth is.
FDA RECALL GUIDANCE
|
FDA Guidance |
What to Do You Believe You Have a Recalled Product in Their Possession? |
|
Follow recall notification instructions |
Read the recall notice carefully and verify the product description such as brand name, packaging size, and codes such as expiration or best by dates to determine whether your product has been recalled. Be sure to follow any product-specific instructions. Additionally, contact the company for further information. |
Which Dexcom products can be involved in recalls?
Dexcom’s ecosystem is bigger than people think. When a recall happens, it could involve one piece and not the others.
A Dexcom setup can include:
- Sensors (the disposable part you wear)
- Transmitters (on some models, reusable for a period)
- Receivers (dedicated handheld device, for some users)
- Mobile app (iOS or Android)
- Overlays and applicators (the insertion mechanism and adhesive system)
- Software/firmware (updates that can affect alerts, connectivity, display)
A recall might target something like:
- sensors from specific manufacturing lots
- a receiver model with a battery issue
- a labeling or instructions issue that could lead to misuse
- an app version that causes missing alerts in a specific OS version
So step one is always identifying exactly what product you have and which batch.
If you or a loved one used a defective Dexcom device and suffered harm when your Dexcom device malfunctioned, contact Dexcom Recall Lawsuit Lawyer Timothy L. Miles for a free case evaluation today as you may be eligible for a Dexcom Device Recall Lawsuit and potentially be entitled to substantial compensation. (855) 846–6529 or [email protected].
How to tell if your Dexcom is affected (without guessing)
This is the practical checklist I’d use if I were doing this at my kitchen table.
1) Find your product and lot information
Depending on what you use, the identifiers live in different places:
- On the sensor box (often has lot number and expiration)
- On the transmitter box (and sometimes on the transmitter itself)
- On the receiver packaging (serial numbers, model numbers)
- In the Dexcom app settings (device info may list transmitter ID, sensor session info, app version)
If you still have the box, use the box. It’s the easiest and least error prone.
If you don not have packaging, don not panic. Look in the app for transmitter ID and app version, and check your pharmacy or DME supplier invoice for lot info. Many suppliers can look up what they shipped you.
2) Check Dexcom’s official recall communication channels
In a real recall, affected users usually learn about it through at least one of these:
- Dexcom email notification (the one tied to your account)
- In app messages
- Dexcom customer support outreach (varies by recall)
- Notices to pharmacies, clinics, and suppliers
You want to read Dexcom’s exact language about:
- affected lot numbers or serial ranges
- whether you should stop using immediately
- what symptoms or failures to watch for
- replacement process
3) Cross check with the FDA database (US) or your local regulator
If you are in the US, the FDA has recall postings and enforcement reports. Other countries have their own systems. This is useful because it often includes:
- reason for recall
- distribution dates
- scope and units affected
- what the firm is doing to correct it
If you can’t find it there yet, that doesn’t automatically mean it’s fake. Timing varies. But if it’s real and large, it typically shows up.
4) Avoid “my friend said all Dexcom is recalled” posts
This is where the panic spiral starts. Most recalls are limited to certain lots or configurations. The internet tends to flatten nuance. “Dexcom recalled” becomes “Dexcom is unsafe.”
Your job is narrower: Is my specific device in the affected group? That’s it.
If you or a loved one used a defective Dexcom device and suffered harm when your Dexcom device malfunctioned, contact Dexcom Recall Lawsuit Lawyer Timothy L. Miles for a free case evaluation today as you may be eligible for a Dexcom Device Recall Lawsuit and potentially be entitled to substantial compensation. (855) 846–6529 or [email protected].
What you should do right now if you think you are affected
If you have reason to believe your sensor, transmitter, or receiver is included in a recall, here’s the safest sequence. It’s boring, but boring is good in a recall.
Step 1: Do not make sudden therapy changes based on fear
If your numbers look normal and the system is working, don not rip it off in a rush unless the recall notice specifically says to stop using immediately.
Removing a CGM suddenly can create a different risk. You lose trend data, you lose alarms, and you may not have a backup plan ready.
Step 2: Read the instructions in the recall notice carefully
Look for phrases like:
- “Stop using and discard”
- “Stop using and contact Dexcom for replacement”
- “Continue use but confirm with blood glucose meter when symptoms do not match”
- “Update your app / receiver software”
- “Return product to supplier”
The recommended action usually depends on what the failure mode is. A sensor that may read falsely low is handled differently than a receiver battery that might overheat. Obvious, but worth saying.
Step 3: Use a blood glucose meter when it matters
Even outside recalls, the standard advice remains: confirm with a fingerstick when:
- you have symptoms that don’t match the CGM reading
- the CGM shows a rapid change you don’t feel
- you are about to correct a severe low or severe high
- the CGM shows “LO” or “HI”
- your device is giving frequent signal loss or calibration issues (for models that calibrate)
During a recall investigation, I’d be a bit more conservative for a few days. Especially overnight lows and before driving.
Step 4: Contact Dexcom support with your lot/serial info ready
This speeds everything up.
Have these ready:
- sensor lot number and expiration
- transmitter ID (if applicable)
- receiver serial number (if applicable)
- what phone model and OS version you’re using (if it’s app related)
- a short description of what you experienced (signal loss, false lows, error codes, failed insertion)
Ask directly:
- “Is my lot affected?”
- “Should I stop using it today?”
- “What’s the replacement process and timeline?”
- “Will you ship expedited?”
- “Do I need a return label?”
Write down the case number. If shipments get delayed, that case number matters.
If you or a loved one used a defective Dexcom device and suffered harm when your Dexcom device malfunctioned, contact Dexcom Recall Lawsuit Lawyer Timothy L. Miles for a free case evaluation today as you may be eligible for a Dexcom Device Recall Lawsuit and potentially be entitled to substantial compensation. (855) 846–6529 or [email protected].
Step 5: Contact your pharmacy or DME supplier too
Sometimes Dexcom handles replacements directly. Sometimes the supplier does. Sometimes it depends on insurance. If you’re running low on sensors, you want all paths open.
If you need an emergency override refill, your prescriber can sometimes help. Especially if you have documentation of a recall replacement in process.
The most common CGM problems that get tangled up with recall fear
Not every scary moment is recall related. A lot of CGM issues are just… CGM life. Adhesive, compression lows, hydration, bad sites, Bluetooth drama.
If you’re worried and trying to figure out what’s real, here are the patterns.
Compression lows (very common, very freaky)
If you sleep on the sensor, it can temporarily show a low that isn not real. Then you roll over, it “magically” fixes itself.
What to do:
- confirm with fingerstick if it’s an urgent low
- consider alternate placement sites approved for your model
- be cautious with correction boluses at night if you don’t have a matching symptom pattern
Compression lows are not a “recall” most of the time. They’re physics.
Sensor warmup weirdness and early failures
Many users notice the first 12 to 24 hours can be noisier. Some sensors settle in. Some fail early.
What to do:
- follow Dexcom guidance on failed sensors and replacement
- document the error code and time
- keep spare supplies when possible
Signal loss and app alerts not coming through
This can be:
- Bluetooth range issues
- phone battery optimization settings
- OS update conflicts
- app permissions getting turned off
- background refresh disabled
This is where app related advisories can look like a recall. If there’s an app issue in the wild, Dexcom may publish guidance like “do not update to OS X until app update Y.” It’s not always called a recall, but from a user perspective it feels the same because missed alerts are serious.
What to do:
- ensure notifications are enabled
- disable aggressive battery optimization for the Dexcom app
- keep the phone on the same side of the body as the sensor when possible
- consider using a dedicated receiver if you’ve had repeated phone alert failures and you can get one covered
Insertion problems and bleeding
Sometimes insertion hurts. Sometimes you hit a blood vessel. Sometimes a sensor fails after a bleeder.
What to do:
- follow Dexcom instructions, especially around bleeding and sensor performance
- if the sensor is inaccurate or fails, report it and seek replacement
- if bleeding is heavy or pain is severe, remove and treat as you would any skin injury and contact a clinician if needed
Again, not usually recall related. But a recall might involve an applicator defect or insertion mechanism problem. That’s why lot numbers matter.
If you or a loved one used a defective Dexcom device and suffered harm when your Dexcom device malfunctioned, contact Dexcom Recall Lawsuit Lawyer Timothy L. Miles for a free case evaluation today as you may be eligible for a Dexcom Device Recall Lawsuit and potentially be entitled to substantial compensation. (855) 846–6529 or [email protected].

If you’re waiting for replacements and running low, here’s a realistic plan
This is the part that actually stresses people out. Not the paperwork. The gap.
1) Count what you have, then add a buffer for failures
If you have 2 sensors left, you don’t really have 20 days of coverage. You have 20 days minus the chance one fails early. Be conservative.
2) Ask Dexcom about expedited shipping
If the recall notice involves replacement, ask if they can ship faster due to medical necessity. Some users get expedited replacements depending on the situation.
3) Ask your prescriber for an emergency fill or backup
Depending on your insurance and country, a prescriber can:
- submit a prior auth note
- document recall impacted supply
- prescribe a short term alternate CGM (not always possible)
- ensure you have test strips and lancets for backup
If you don’t have a meter right now, get one. Even a cheap one. It’s your parachute.
4) Consider temporary alarm strategies
If you must go without CGM for a bit:
- set phone alarms to check glucose at night if you’re prone to nocturnal lows
- check before driving, before exercise, and before bed
- keep fast acting carbs nearby
- let a family member know if you’re in a higher risk period
It’s not fun. But it’s manageable for a short window if you plan it.
What to ask your doctor or diabetes educator (because they can help more than you think)
If you are unsure whether to keep using a potentially affected device while waiting on replacements, this is a great time to talk to your clinician. Not because they have inside recall info, but because they can tailor risk based on you.
Questions that get useful answers:
- “Given my hypo history, do you want me to confirm CGM lows with fingersticks for a week?”
- “Should I temporarily adjust low alert thresholds?”
- “If I lose CGM for a few days, what’s the safest checking schedule?”
- “Do I need a prescription for extra strips right now?”
- “Can you document medical necessity for expedited replacements or overrides?”
Clinicians can also file adverse event reports if something truly went wrong, which helps regulators see patterns.
How to report a serious issue (and why it matters)
If you experience an event that could cause harm, like a dangerously inaccurate reading leading to incorrect dosing, repeated missed urgent low alerts, device overheating, burns, things like that, report it.
Where to report depends on your location:
- To Dexcom: customer support, with device identifiers and event details
- To regulators: in the US, MedWatch (FDA). In other countries, your national reporting system.
It’s annoying to fill out forms when you’re tired. I get it. But these reports are often what turns “isolated complaints” into “ok, this is a pattern, we need action.”
How to spot misinformation during a recall wave
A few red flags that a post or screenshot isn’t reliable:
- no lot numbers listed, just “all units”
- no link to Dexcom or a regulator notice
- claims that don’t match how medical recalls work, like “lawsuit confirmed device is defective for everyone” – which might hint at a defective Dexcom device lawsuit
- blurry screenshots with no source
- instructions to “switch brands immediately” with affiliate links
There’s also the opposite problem. People downplay it and say “recalls don’t matter.” They do matter. But you need the specifics so your response matches the real risk.
A simple consumer checklist (print this mentally)
If you want the short version to follow when you’re stressed, here:
- Identify your device and lot/serial numbers.
- Read Dexcom’s official recall instructions for that exact product.
- Confirm highs/lows with a meter when symptoms don’t match, and in high stakes moments.
- Contact Dexcom support for confirmation and replacements, get a case number.
- Loop in your pharmacy or DME supplier if supply is tight.
- Build a short term backup plan if you may run out.
- Report serious adverse events to Dexcom and your regulator.
That’s it. No doom scrolling required.

What happens next, usually
Most recall processes follow a predictable arc:
- Dexcom identifies the issue and affected lots.
- Notices go out to suppliers and users.
- Replacement or correction begins.
- Regulators publish or update postings.
- The scope may expand if new lots are included.
- Over time, the issue becomes less noisy because affected stock is pulled out of circulation.
So if you’re reading this because you just heard about it today, you’re not late. You’re just in the loud part of the cycle.
Understanding the Legal Implications
In some cases, users may experience severe issues due to a defective device leading to potential legal action. If you’re wondering whether you qualify for a Dexcom lawsuit, it’s crucial to consult with legal professionals who specialize in such matters.
Final thoughts
Dexcom recalls, when they happen, tend to be specific. Lot based, version based, situation based. That’s both reassuring and frustrating. Reassuring because it’s usually not “everything.” Frustrating because it means you have homework.
But you can do the homework in ten minutes.
Find the lot number. Check the official notice. Confirm critical readings with a meter for a bit. Get your replacement process started, and get a case number. Then stop refreshing social media like it’s going to give you a better answer than the people who actually have the list of affected lots.
If you want, paste the exact product you’re using (G6, G7, receiver vs phone, and your country) and what you’ve heard about the recall, and I can help you narrow down the right place to verify it and what info to gather before you call support.
If you or a loved one used a defective Dexcom device and suffered harm when your Dexcom device malfunctioned, contact Dexcom Recall Lawsuit Lawyer Timothy L. Miles for a free case evaluation today as you may be eligible for a Dexcom Device Recall Lawsuit and potentially be entitled to substantial compensation. (855) 846–6529 or [email protected].
Frequently Asked Questions about theRecalled Dexcom Device
What does a Dexcom recall mean and should I stop using my device immediately?
A Dexcom recall is a formal process to correct an issue with a product already in use. It doesn’t always mean the device will injure you or is bad as a whole, and you may not need to stop using it immediately. Often, recalls target specific lot numbers or serial ranges and might require actions like replacement, software updates, or extra instructions. Always read the official ‘what to do’ instructions from Dexcom.
Which Dexcom products can be involved in recalls?
Dexcom’s ecosystem includes sensors, transmitters, receivers, mobile apps, overlays/applicators, and software/firmware. A recall can affect any of these components individually or in combination, such as sensors from specific lots, a receiver model with battery issues, labeling problems, or app versions causing missing alerts.
How can I check if Defective Dexcom Device affected by a recall?
First, identify your product and lot information from sensor boxes, transmitter packaging, receiver packaging, or the Dexcom app settings. Then check Dexcom’s official recall communications via email notifications tied to your account, in-app messages, customer support outreach, or notices from pharmacies and clinics. Lastly, cross-check with the FDA recall database (if in the US) or your local regulator’s system for official details.
What are the different classes of medical device recalls and what do they mean for Dexcom users?
In the US, recalls are classified as Class I (most serious), Class II, or Class III based on potential severity if the issue occurs—not on how many units are affected. For example, Class I might require immediate stop of use for certain lots or heightened awareness of false readings. Understanding these classes helps manage expectations but always follow the specific instructions provided by Dexcom.
How should I protect myself from common CGM failure modes during a Dexcom recall?
While Dexcom and regulators work out recall details, stay informed by verifying official sources regularly. Follow all recommended precautions such as confirming suspicious readings with fingerstick tests if advised. Keep your devices updated with the latest software/firmware and adhere strictly to any additional instructions provided by Dexcom to ensure safe use.
Are there legal options available if I face challenges due to a recent Dexcom recall?
Yes. If you experience issues related to a recent Dexcom recall, there may be legal avenues for compensation through lawsuits related to the recall. Staying informed about ongoing legal proceedings via reliable sources can provide insights into potential relief options available for affected users.


