Introduction to Zepbound and NAION
Welcome to this authoritative ananlysis on Zepbound and NAION. Zepbound (tirzepatide) has rapidly become a centerpiece of modern obesity medicine. It is clinically effective, widely discussed, and increasingly prescribed. However, it serves as a reminder of a basic rule in pharmacotherapy: when a medication meaningfully alters metabolic physiology, clinicians and patients must remain vigilant for rare but severe adverse events.
One such event that has drawn renewed attention across the broader GLP-1 receptor agonist category is NAION, short for non-arteritic anterior ischemic optic neuropathy. NAION is not a routine side effect. It is a potentially vision-devitalizing condition that can permanently change a person’s independence, employability, and quality of life in a single morning.
This article explains what NAION is, why the Zepbound conversation exists, what is known and not known, and how to approach risk in a way that is medically responsible, legally prudent, and operationally practical.
If you were prescribed Zepbound and took it as directed and suffered Zepbound and NAION, Zepbound vision loss or other Zepbound Vision Side Effects, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].

What Zepbound Is, and Why It Matters
Zepbound is the trade name for tirzepatide, a once-weekly injectable medication approved for chronic weight management in adults who meet clinical criteria. Tirzepatide is a dual incretin agonist, meaning it activates:
- GLP-1 (glucagon-like peptide-1) receptors, and
- GIP (glucose-dependent insulinotropic polypeptide) receptors
This dual mechanism can improve insulin sensitivity, reduce appetite, slow gastric emptying, and promote meaningful weight loss. For many patients, it also improves cardiometabolic risk factors such as blood pressure, triglycerides, hepatic steatosis markers, and glycemic parameters.
These benefits are real. They are also precisely why the safety conversation must be equally real. High-impact medicines create high-impact risk management obligations, even when severe outcomes are rare.
Recent reports have linked Zepbound to various vision-related issues, including NAION, which is alarming given its potential to cause permanent vision loss. Other reported side effects include eye floaters and other forms of vision loss. These serious side effects have led to discussions about Zepbound vision loss lawsuits, highlighting the importance of understanding these risks thoroughly before starting treatment with this medication.
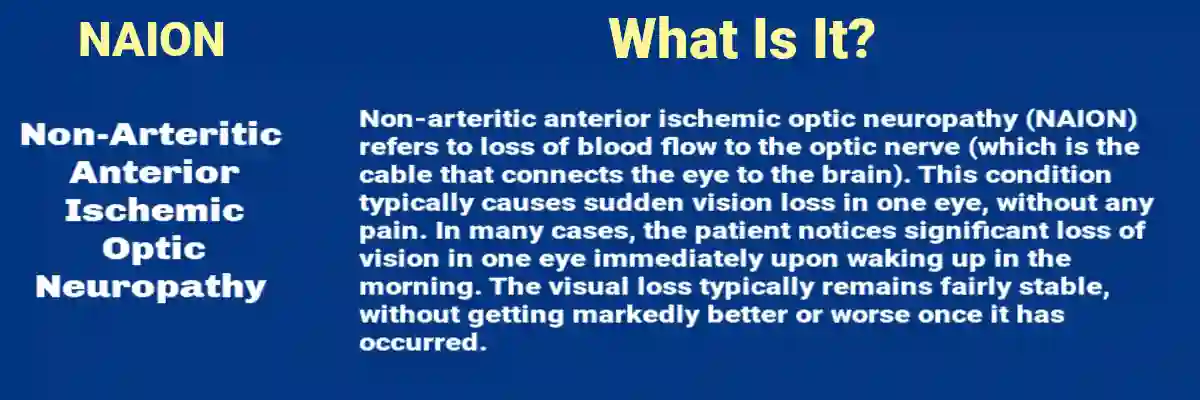
NAION Defined in Plain, Clinical Terms
Non-arteritic anterior ischemic optic neuropathy (NAION) is an acute ischemic event affecting the optic nerve head (the front portion of the optic nerve). It is “non-arteritic” because it is not caused by the inflammatory large-vessel process seen in giant cell arteritis (GCA), which is a different emergency.
NAION usually presents as:
- Sudden, painless vision loss in one eye, often noticed upon waking
- A visual field defect, classically an “altitudinal” loss (upper or lower half of vision)
- Decreased visual acuity and/or contrast sensitivity
- Sometimes reduced color vision
On exam by an eye professional, typical findings include optic disc swelling (edema), often with small hemorrhages. The condition can stabilize, partially improve, or remain permanently impaired. For a meaningful subset of patients, the loss is functionally significant and persistent.
NAION is not a minor inconvenience. It is an event with the potential to impair driving, reading, depth perception, and occupational performance. It is also associated with a risk of involvement of the other eye over time.

Why People Are Discussing Zepbound and NAION
The Zepbound and NAION discussion exists because:
- Zepbound belongs to a class of potent metabolic agents that can produce rapid physiologic change, including shifts in blood pressure, glycemic patterns, hydration status, and vascular tone.
- NAION is a vascular optic neuropathy with known associations to systemic vascular risk factors, many of which overlap with populations treated for obesity and metabolic disease.
- Public attention has increased regarding optic nerve events in relation to incretin-based therapies broadly, leading patients to ask direct questions about whether tirzepatide specifically could be implicated.
At this time, the most defensible position is careful, evidence-aligned neutrality:
- While a rare severe event like NAION may be biologically plausible in some circumstances following Zepbound treatment,
- A definitive causal link between tirzepatide and NAION cannot be asserted without robust data.
- The clinical duty to recognize symptoms early remains absolute regardless of causality.
In risk governance terms, this is a classic scenario: low frequency, high severity, high reputational sensitivity, and high patient anxiety. The correct response is not panic but preparedness.
Furthermore, there have been discussions surrounding the potential link between Trulicity and NAION as well as concerns about GLP-1 medications causing vision loss, which adds complexity to this issue. Reports of blurry vision associated with Zepbound have also surfaced alongside claims of vision loss linked to its use.
What NAION Risk Looks Like in the Real World
NAION is classically associated with a combination of anatomical predisposition and systemic risk factors. Established or frequently cited risk factors include:
- Age over 50
- Hypertension
- Diabetes mellitus
- Hyperlipidemia
- Obstructive sleep apnea (OSA)
- Smoking
- Nocturnal hypotension (blood pressure dipping too low overnight, sometimes influenced by timing of antihypertensive medications)
- A “crowded disc” anatomy (small optic cup), sometimes called a “disc at risk”
- Prior NAION in the other eye (significant risk factor for recurrence in the fellow eye)
Notably, several of these are common in patients eligible for pharmacologic weight management. That does not prove medication causality. It does mean the baseline risk context is not trivial.
If you were prescribed Zepbound and took it as directed and suffered Zepbound and NAION, Zepbound vision loss or other Zepbound Vision Side Effects, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
A Forward-Looking Clinical Question: Could Rapid Metabolic Change Matter?
Even without claiming direct causation, it is reasonable to ask how rapid physiologic shifts might intersect with optic nerve perfusion in susceptible individuals.
Potential pathways clinicians may consider, conceptually, include:
- Blood pressure changes, including reductions during weight loss or medication titration, with possible contribution to nocturnal hypotension in predisposed patients.
- Glycemic shifts, particularly in patients with diabetes who experience changes in glucose variability or medication regimens.
- Dehydration or reduced intravascular volume, especially if gastrointestinal side effects reduce intake or increase losses.
- Sleep apnea dynamics, since weight loss can improve OSA over time, yet untreated OSA remains a known NAION risk factor in the interim.
- Vascular dysregulation in patients with existing small-vessel disease.
These are not accusations. They are clinical reasoning points. They reinforce the importance of structured monitoring and patient education during initiation and dose escalation.
It’s also worth noting that certain medications, such as Zepbound, have been linked to various vision-related issues including dry eye syndrome. Patients experiencing these side effects may be eligible for a Zepbound vision loss lawsuit.
The “1 Devitalizing Hideous Side Effect”: Why NAION Is Different From Typical Adverse Events
Most adverse events discussed with Zepbound are familiar and manageable: nausea, constipation, diarrhea, reflux, appetite suppression, fatigue, and transient gastrointestinal discomfort. These can be unpleasant, but they are generally reversible and treatable.
NAION is different because it is:
- Sudden
- Often irreversible
- Functionally disabling
- Time-sensitive for evaluation, because urgent conditions must be excluded
In a patient’s lived experience, sudden vision loss is existential. It changes the risk-benefit narrative instantly. Even if the probability is low, the impact is high. In medical decision-making, that combination demands a specific kind of counseling: clear, calm, specific, and actionable.
What Patients on Zepbound Should Watch For
If a patient is using Zepbound and experiences any of the following symptoms related to NAION, the appropriate response is to treat it as urgent:
- Sudden vision loss or dimming in one eye
- A new dark “curtain,” missing area, or shadow in the visual field
- Sudden loss of peripheral vision
- Marked blurring that is not explained by dry eye or routine refractive change
- Noticeable loss of color intensity in one eye
It’s important to note that NAION is typically painless. The absence of pain should not reassure anyone.
Because multiple eye and neurologic emergencies can mimic each other, the correct next step is not online self-diagnosis. The correct next step is urgent clinical evaluation, often the same day.
For those who have experienced severe vision-related side effects while on Zepbound, such as sudden vision loss, it may be advisable to seek legal counsel. Current updates regarding ongoing lawsuits related to these cases can be found in this Zepbound vision loss lawsuit update.
What Clinicians Should Do if NAION Is Suspected
When NAION is suspected, the first responsibility is not to debate medication causality. The first responsibility is to protect the patient from missed emergencies.
A typical urgent approach includes:
- Immediate ophthalmic evaluation, ideally neuro-ophthalmology or ophthalmology with optic nerve expertise.
- Rule out arteritic AION (giant cell arteritis) in appropriate patients, especially those over 50 with systemic symptoms such as scalp tenderness, jaw claudication, fever, weight loss, or polymyalgia rheumatica symptoms. This often triggers urgent ESR/CRP testing and potential steroid initiation per clinician judgment.
- Document symptom onset, risk factors, and a medication timeline including tirzepatide dose changes, hydration status, blood pressure medications, and recent illnesses.
- Consider systemic evaluation for vascular risks, including blood pressure patterns, diabetes control, lipid management, smoking status, and sleep apnea screening.
From a governance standpoint, documentation matters. From a patient standpoint, speed matters.
If you were prescribed Zepbound and took it as directed and suffered Zepbound and NAION, Zepbound vision loss or other Zepbound Vision Side Effects, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
Should Zepbound Be Stopped if NAION Occurs?
A medication decision after suspected NAION should be individualized and clinician-led. However, a practical framework is:
- Do not inject another dose until urgently evaluated, unless a treating clinician explicitly advises otherwise.
- If NAION is confirmed and the patient has been using Zepbound, clinicians commonly reassess the entire risk-benefit profile of the medication. This includes considering the Zepbound blindness lawsuit which highlights potential severe side effects like blindness associated with its use.
- It’s also crucial to stay updated on any ongoing Zepbound lawsuit that may provide further insights into the medication’s safety profile.
A strong, proactive stance is to treat optic nerve ischemia as a “high-severity safety signal” for that individual patient, even when broader population-level causality remains uncertain.
How to Reduce Risk Responsibly Without Fear-Based Medicine
The goal is not to frighten patients away from effective treatment. The goal is to practice modern medicine properly: proactive, structured, and transparent.
1) Establish baseline risk before starting
Patients with multiple NAION risk factors benefit from a deliberate conversation that includes:
- History of optic nerve disease or unexplained vision events
- Diabetes, hypertension, hyperlipidemia
- Smoking status
- Sleep apnea symptoms (snoring, witnessed apneas, daytime sleepiness)
- Timing of blood pressure medications and history of lightheadedness on standing
For higher-risk individuals, some clinicians may consider baseline eye evaluation, especially if there is prior optic neuropathy history. This is not a universal requirement, but it can be a reasonable governance decision.
2) Manage dehydration and nutritional intake during titration
Gastrointestinal effects are common early in therapy. Practical risk reduction includes:
- Maintaining adequate fluid intake
- Avoiding aggressive caloric restriction that compromises hydration and electrolytes
- Prompt management of persistent vomiting or severe diarrhea
- Clear escalation pathways: when to call, when to be seen, when to go to urgent care
3) Monitor blood pressure thoughtfully
Weight loss and improved metabolic status can lower blood pressure, sometimes quickly. Clinicians should:
- Monitor blood pressure trends
- Reassess antihypertensive dosing over time
- Be attentive to symptoms of hypotension, particularly morning dizziness
- Consider the concept of nocturnal hypotension in susceptible patients, as clinically appropriate
4) Identify and treat obstructive sleep apnea
OSA is both common and underdiagnosed. It is also modifiable. A forward-looking obesity management plan should treat sleep apnea screening and treatment as part of whole-person cardiometabolic care, not as a side project.
5) Repeat the message that matters
In safety counseling, repetition is a feature, not a bug:
- Sudden vision change is urgent.
- Sudden vision change is urgent.
- Sudden vision change is urgent.
Patients remember what is repeated.
What Is Known, What Is Not Known, and Why That Distinction Matters
In pharmacovigilance, there is a critical difference between:
- Signal detection (a pattern that deserves attention),
- Association (a correlation in a dataset), and
- Causation (a proven relationship with plausible mechanism, consistent evidence, and exclusion of confounding).
For Zepbound and NAION, most public discourse tends to skip these definitions. A professional and accurate approach requires stating clearly:
- NAION occurs in the general population, especially in patients with vascular risk factors.
- Patients using weight management medications often have overlapping vascular risk profiles.
- Post-marketing safety surveillance can identify rare events, but rare events are statistically challenging to interpret.
- A definitive causal claim requires high-quality evidence, careful control of confounders, and consistent replication.
That said, clinicians do not need causal certainty to act on symptoms. In medicine, you treat the patient in front of you, not the statistical debate. It’s important to note that Zepbound has been associated with some severe vision side effects like NAION which necessitate immediate medical attention.
Corporate Governance, Clinical Governance, and Why This Topic Will Not Go Away
Zepbound sits at the intersection of high demand, high visibility, and high expectation. In that environment, governance is not optional.
For health systems, clinics, telehealth platforms, and prescribing programs, the operational obligations are straightforward:
- Standardize informed consent language for severe, vision-related symptoms that warrant urgent evaluation.
- Create escalation protocols that route visual complaints immediately to clinician review.
- Train front-line staff to treat “sudden vision loss” as an emergency phrase, not as a routine ticket.
- Maintain clear documentation of counseling, patient questions, and clinical decisions.
- Review adverse event reports with a structured safety committee mindset, not an ad hoc reaction.
This is forward-thinking risk management. It protects patients. It protects clinicians. It protects organizations.
Most importantly, it preserves trust, which is the most fragile asset in modern care delivery.
Practical Patient Script: What to Ask Before and During Zepbound
Patients do not need a lecture. They need clarity. These questions are appropriate and useful:
- “Given my blood pressure and diabetes history, am I at higher baseline risk for optic nerve problems?”
- “If I notice sudden vision change due to Zepbound, who do I call first, and how fast should I be seen?”
- “Should we monitor my blood pressure more closely during dose increases?”
- “Do you suspect sleep apnea, and should I be tested?”
- “If severe nausea reduces my intake, what hydration plan do you want me to follow?”
These questions create a partnership model. They replace internet anxiety with a clinical plan.
However, if patients experience severe complications such as sudden vision loss, or even blindness, it’s crucial to have established protocols in place. Such scenarios underline the importance of having well-defined governance structures within health systems dealing with Zepbound prescriptions.
The legal landscape surrounding these issues is also evolving. Recent lawsuit updates highlight the need for accountability and transparency in the use of Zepbound within healthcare settings.
The Bottom Line
Zepbound is an effective medication that can transform metabolic health. NAION is a rare but potentially devastating optic nerve event that can transform a life in the opposite direction. The correct approach is not denial and not alarmism.
The correct approach is structured vigilance:
- Know the symptoms.
- Treat sudden vision change as urgent.
- Manage vascular risk factors proactively.
- Document, monitor, and escalate appropriately.
That is how modern medicine earns its results and keeps its integrity.
If you were prescribed Zepbound and took it as directed and suffered Zepbound and NAION, Zepbound vision loss or other Zepbound Vision Side Effects, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
Frequently Asked Questions about Zepbound and NAION
What is Zepbound (tirzepatide) and how does it work for weight management?
Zepbound is the trade name for tirzepatide, a once-weekly injectable medication approved for chronic weight management in adults meeting clinical criteria. It is a dual incretin agonist that activates GLP-1 and GIP receptors, improving insulin sensitivity, reducing appetite, slowing gastric emptying, and promoting meaningful weight loss along with improving cardiometabolic risk factors.
What is non-arteritic anterior ischemic optic neuropathy (NAION) and why is it significant?
NAION is an acute ischemic event affecting the optic nerve head, characterized by sudden, painless vision loss in one eye, typically noticed upon waking. It can cause permanent vision impairment affecting independence, employability, and quality of life. NAION presents with visual field defects and optic disc swelling and may lead to persistent functional vision loss.
Why has Zepbound and NAION become a concern in discussions about Zepbound?
NAION has drawn attention because Zepbound induces rapid metabolic changes affecting vascular tone and glycemic patterns. Since NAION is associated with systemic vascular risk factors common in patients treated for obesity and metabolic disease, there is biological plausibility that tirzepatide could be linked to rare cases of NAION, although definitive causal evidence is lacking.
Are Zepbound vision side effects common with Zepbound treatment?
Vision-related side effects such as eye floaters and vision loss including NAION are reported but considered rare. Despite their rarity, these serious adverse events are significant due to their potential to cause permanent vision damage. Patients experiencing any visual symptoms while on Zepbound should seek urgent evaluation by a qualified clinician.
What precautions should patients and clinicians take regarding Zepbound and NAION risk when using Zepbound?
Given the severity of NAION despite its rarity, clinicians and patients must remain vigilant for sudden visual changes during treatment. Early recognition of symptoms like painless vision loss or visual field defects is critical. Risk assessment should be medically responsible, legally prudent, and operationally practical to balance treatment benefits against potential adverse events.
Is there definitive evidence linking Zepbound to NAION?
Currently, no robust data definitively establishes a causal link between tirzepatide (Zepbound) and NAION. The relationship remains biologically plausible but unproven. Ongoing vigilance and research are necessary while maintaining a neutral evidence-aligned stance regarding this potential risk.
If You Suffered from Zepbound and NAION or Other Zepboound Vision Problems, Contact Zepbound Vision Loss Lawyer Timothy L. Miles Today
If you were prescribed Zepbound and took it as directed and suffered Zepbound and NAION, Zepbound vision loss or other Zepbound Vision Side Effects, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com

