Introduction to Zepbound and NAION: 1 Harrowingly Permanently Side Effect
Welcome to Zepbound and NAION. Zepbound (tirzepatide) has quickly transitioned from “newly approved” to “widely discussed” in the realm of obesity medicine. The clinical rationale is straightforward: significant weight reduction can enhance cardiometabolic risk factors, functional status, and long-term health outcomes. However, with the rapid scaling of this therapy, there arises an equally clear governance obligation: the discipline of risk recognition must also scale accordingly.
One adverse event that has surfaced as a major concern in patient forums, clinician discussions, and risk reviews is NAION, or non-arteritic anterior ischemic optic neuropathy. NAION is not a typical “Zepbound eye side effect.” It represents an acute optic nerve injury that can result in sudden, often irreversible vision loss. For any organization involved in prescribing, dispensing, counseling, or covering Zepbound, NAION signifies a high-severity risk signal that necessitates proactive controls.
This article analyzes what NAION is, why describing it as a “permanent side effect” is not an exaggeration, what is currently understood and unknown about its potential relationship with Zepbound, and what responsible risk management entails in practice.
If you were prescribed Zepbound and took it as directed and suffered Zepbound vision loss or other Zepbound eye problems, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].

A Precise Definition: What NAION Actually Is
Non-arteritic anterior ischemic optic neuropathy (NAION) stands as the most prevalent acute optic neuropathy in older adults. The term “non-arteritic” differentiates it from arteritic ischemic optic neuropathy, which is usually linked with giant cell arteritis, a medical emergency that demands immediate treatment.
NAION occurs when blood flow to the anterior portion of the optic nerve becomes inadequate, resulting in ischemic injury. The hallmark features include:
- Sudden, painless vision loss in one eye (often noticed upon waking)
- Visual field defects, frequently inferior altitudinal loss
- Optic disc swelling on examination
- Variable recovery, but many patients have persistent deficits
The key governance point here is severity: NAION is not primarily about irritation, dryness, or transient blurring. It is a vascular-neurologic event affecting the optic nerve, and the damage can be permanent.
Given these serious implications, it’s crucial for those considering Zepbound to be aware of potential vision loss associated with its use. There have been instances where users have experienced irreversible vision loss, leading to legal action against the manufacturers. This underscores the importance of understanding NAION and its potential link to Zepbound, as well as similar risks associated with other medications like Trulicity which have also been reported to cause NAION.
The “Harrowingly Permanent” Reality: Why NAION Is Feared
NAION is feared because it can change a patient’s life within hours. While some patients experience partial improvement, a significant proportion have ongoing impairment. Clinical outcomes vary, but the risk profile is defined by several characteristics:
- Abrupt onset: patients can go from normal vision to a major deficit without warning.
- Functional impact: visual field loss affects driving, reading, work performance, and independence.
- Limited restorative therapy: unlike some ocular conditions, there is no universally accepted treatment that reliably reverses NAION.
- Risk to the fellow eye: NAION can occur in the other eye over time, increasing cumulative disability risk.
When stakeholders describe NAION as “harrowing,” they are describing the lived reality of sudden loss, uncertainty, and limited clinical options.
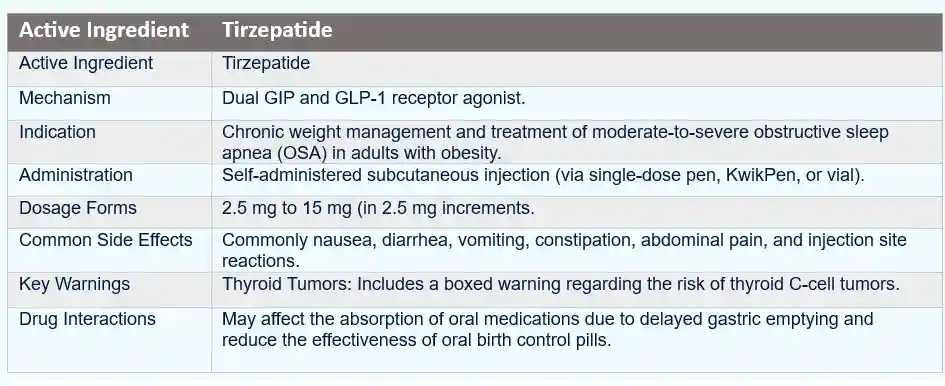
Zepbound in Context: What It Is and Why It Is Used
Zepbound is tirzepatide, a dual incretin agonist that targets GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide) receptors. It is prescribed for chronic weight management in appropriate patients, typically alongside nutrition, physical activity, and behavioral interventions.
The benefit case is strong for many patients. However, high-benefit therapies demand high-integrity safety processes. Robust corporate governance in healthcare means:
- Identify plausible high-severity adverse events early.
- Communicate risks without distortion.
- Create escalation pathways for red-flag symptoms.
- Document decision-making and informed consent with discipline.
NAION due to its potential permanence, fits the profile of a risk that must be handled with exceptional care.
Unfortunately, there are reports suggesting a possible link between Zepbound and vision loss, including serious conditions like NAION. Such potential side effects necessitate thorough discussions about the risks involved in using this medication. Moreover, there have been instances where patients have experienced eye floaters after starting Zepbound treatment. These experiences underline the importance of monitoring and managing any adverse effects promptly.
In light of these concerns, it’s crucial for both healthcare providers and patients to remain vigilant about any Zepbound-related eye problems. If such issues arise, they should be documented meticulously as part of the informed consent process.
Furthermore, if a patient suffers from significant vision loss due to Zepbound, they may have grounds for legal recourse. It’s essential to understand one’s rights and seek appropriate guidance in such situations.
Zepbound and NAION: What Is Known, What Is Suspected, and What Is Not Proven
A responsible analysis separates three categories: established facts, biological plausibility, and causality.
1) Established facts
- NAION is a known clinical entity with established risk factors and a baseline incidence in the general population.
- Tirzepatide is a potent metabolic drug that can produce substantial weight loss and improvements in glycemic parameters.
- Patients using GLP-1–based therapies report ocular symptoms for many reasons, ranging from benign to serious, including changes related to diabetes and blood pressure.
2) Biological plausibility (a “could it make sense?” lens)
NAION is associated with vascular perfusion and optic nerve head anatomy. Conditions that influence perfusion, oxygen delivery, and microvascular stability can be relevant. In theory, several pathways could intersect with NAION risk:
- Blood pressure dynamics, particularly nocturnal hypotension
- Rapid metabolic shifts (glycemic changes, fluid balance changes)
- Dehydration or volume depletion, especially during periods of nausea, vomiting, or reduced intake
- Sleep apnea relationships, given sleep apnea is a known NAION risk factor and is also common in obesity
Plausibility does not mean proof. It means the risk is not scientifically absurd and therefore should be treated as a signal worthy of structured monitoring.
3) Causality (the “did the drug cause it?” standard)
Causality requires evidence that exceeds anecdote. That includes pharmacoepidemiologic studies, controlled analyses, adjudicated case series, and reproducible patterns. Even then, causality can be complex due to confounding factors such as diabetes, hypertension, dyslipidemia, and obstructive sleep apnea, which are common in the same population prescribed Zepbound.
Recent updates have raised concerns about potential side effects associated with Zepbound, including instances of blindness linked to its usage. This has led to ongoing lawsuits against the drug manufacturer. As these legal proceedings unfold, they may provide further insights into the safety profile of Zepbound.
At present, patient safety decisions should be made under uncertainty. Governance maturity means you do not wait for perfect certainty before implementing sensible precautions for a high-severity outcome.
If you were prescribed Zepbound and took it as directed and suffere Zepbound vision loss or other Zepbound eye problems, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
Why This Risk Conversation Is Hard: Confounding Factors Are Everywhere
Patients eligible for Zepbound frequently have comorbidities that overlap with NAION risk, including:
- Type 2 diabetes or prediabetes
- Hypertension
- Hyperlipidemia
- Obstructive sleep apnea
- Cardiovascular disease
- Smoking history
- Older age
- Use of certain medications associated in some literature with NAION risk, such as PDE-5 inhibitors (the evidence and interpretation vary, but they are commonly discussed in NAION evaluations)
This overlap creates a governance challenge: if NAION occurs after starting Zepbound, correlation may be strong in a patient narrative but uncertain in a scientific model. The correct organizational response is not denial or alarmism. It is structured clinical vigilance.
The Single Side Effect That Changes the Risk Category: Sudden Vision Loss
Many adverse effects associated with weight loss medications, including incretin therapies, are common and manageable: nausea, constipation, diarrhea, fatigue, reflux, reduced appetite. NAION is different because it changes the risk category from “tolerability management” to “potentially irreversible disability.”
That is why it deserves explicit framing as the one harrowingly permanent side effect patients and clinicians must understand.
A governance-forward organization treats sudden vision change as a time-sensitive safety event, similar in seriousness to chest pain, stroke symptoms, or severe allergic reactions.
Practical Symptom Recognition: What Patients Should Not Ignore
NAION often presents without pain. That fact increases risk because patients may delay care.
Red-flag symptoms that should trigger urgent medical evaluation include:
- Sudden blurred vision in one eye
- A new dark area or “shadow” in the visual field
- Loss of a portion of vision, often described as a “curtain” or a missing segment
- Marked reduction in contrast or brightness in one eye
- Any abrupt vision change, especially upon waking
In a high-integrity safety program, these symptoms are not handled with reassurance scripts. They are escalated, triaged, and documented.

What To Do If Symptoms Occur While Taking Zepbound
This section is operational by design. In risk management, clarity beats ambiguity.
- Treat sudden vision changes as urgent. The appropriate next step is urgent evaluation by an eye professional or emergency services, depending on severity and access. Sudden changes in vision could be indicative of serious side effects associated with Zepbound, such as vision loss or blurry vision.
- Do not “wait and see.” NAION and other acute ocular events are time-sensitive to diagnose, and delays can limit options.
- Document medication timing and associated symptoms. Include dose changes, dehydration episodes, vomiting, blood pressure changes, or new medications.
- Clinicians should coordinate care. Primary care, obesity medicine, endocrinology, and ophthalmology coordination should be explicit, not assumed.
This is not a substitute for medical advice. It is a governance-aligned escalation model that prioritizes patient safety when a potentially permanent outcome is on the table.
Informed Consent That Reflects Modern Standards
Informed consent is not a form. It is a process. And when a rare but severe risk exists, the consent process must be proportionate.
A forward-looking informed consent conversation for Zepbound should include:
- Common adverse effects and how they are managed.
- Serious adverse events such as sudden vision loss and what symptoms require urgent action.
- A plain-language explanation that sudden vision loss can be a medical emergency.
- A clear instruction: any sudden visual change should prompt immediate evaluation.
From a corporate governance perspective, this reduces operational risk in three ways:
- It improves patient outcomes through earlier presentation.
- It reduces misinformation and mistrust.
- It creates defensible documentation that appropriate risk disclosure occurred.
Repetition matters here. Repeat the message. Repeat the threshold. Repeat the escalation pathway.
If patients experience severe side effects like dry eye syndrome while taking Zepbound, it is essential to seek immediate medical attention.
Clinical Risk Factors That Merit Proactive Screening and Documentation
If you prescribe or manage Zepbound, NAION risk management begins before the first dose. A disciplined intake can identify patients who may require heightened counseling or monitoring.
Key elements to review and document include:
- History of NAION in either eye
- Known optic disc anatomy concerns if previously documented (many patients will not know this)
- Obstructive sleep apnea diagnosis, symptoms, or CPAP adherence
- Blood pressure patterns and antihypertensive timing, with attention to nocturnal hypotension risk
- Diabetes status, recent A1c trends, and whether rapid glycemic improvement is anticipated
- Current medications that may affect perfusion or vascular risk
- Hydration status risk factors, including prior intolerance to similar therapies
None of these steps prove causality. They demonstrate governance maturity: identify vulnerabilities and reduce preventable harm.
However, it’s crucial to be aware of potential side effects from the prescribed medication. For example, Zepbound, Trulicity, and Saxenda have been associated with severe vision-related side effects.
Operational Controls: What Good Governance Looks Like in Practice
Healthcare organizations, telehealth platforms, pharmacies, and insurers can implement practical controls without overreacting.
Standardized escalation pathways
Create a standard triage script for Zepbound vision symptoms that leads to urgent evaluation, not delayed messaging.
Training and role clarity
Ensure nurses, pharmacists, and support staff know that “painless sudden vision loss” is not a routine side effect discussion related to Zepbound or similar medications.
Documentation discipline
Record adverse event reports with date, dose, symptom onset, and concurrent risk factors. High-quality data supports pharmacovigilance and reduces institutional blind spots.
Pharmacovigilance and reporting
Where appropriate, encourage formal adverse event reporting through established channels. A safety culture depends on structured feedback loops.
Patient education artifacts
Provide a brief handout or portal message that highlights red flags, including Zepbound vision changes, and explains what to do.
This is proactive governance: not fear, not denial, but controlled preparedness.
Avoiding Two Common Failures: Alarmism and Dismissal
Risk communication fails in predictable ways.
- Alarmism exaggerates frequency or certainty and can lead patients to discontinue beneficial therapy without guidance.
- Dismissal minimizes severity and can delay urgent evaluation for a potentially irreversible event.
The correct posture is neutral, precise, and action-oriented:
- NAION is serious.
- It may be rare.
- Causality may be uncertain.
- The response to symptoms should be urgent and standardized.
That is how integrity is protected and how trust is preserved.
A Clear Bottom Line for Patients and Clinicians
Zepbound can be an effective tool for weight management, and many patients will use it safely. However, NAION represents a uniquely consequential potential outcome because it can be sudden and permanent such as Zepbound and blurry vision and Zepbound and eye floaters.
The forward-thinking approach is simple and disciplined:
- Know the red flags.
- Escalate immediately if they occur.
- Document risk factors and counsel proactively.
- Maintain a safety culture grounded in clarity, accuracy, and repetition.
Robust clinical governance does not wait for perfect certainty. It builds systems that protect patients now, improves decision quality over time, and reinforces integrity at every step. This aligns with the principles of quality risk management which emphasize systematic processes to identify, assess, control, and review risks throughout the patient care continuum.
If you were prescribed Zepbound and took it as directed and suffereds Zepbound and blurry vision, Zepbound and NAION, Zepbound vision loss or other Zepbound eye problems, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
Frequently Asked Questions About Zepbound and NAION
What is NAION and why is it a concern with Zepbound (tirzepatide) use?
Non-arteritic anterior ischemic optic neuropathy (NAION) is an acute optic nerve injury characterized by sudden, painless vision loss, often irreversible. It occurs due to inadequate blood flow to the optic nerve. NAION has emerged as a significant risk associated with Zepbound (tirzepatide), making it a high-severity adverse event that requires careful Zepbound and NAION risk management.
How does NAION affect vision and daily functioning?
NAION causes sudden vision loss, typically in one eye, leading to visual field defects such as inferior altitudinal loss. This can impair essential activities like driving, reading, and working, severely impacting a patient’s independence. The damage from Zepbound and NAION may be permanent due to limited restorative treatment options making it one of the most atrotious Zepbound vision side effects.
What is Zepbound (tirzepatide) and its clinical use?
Zepbound is the brand name for tirzepatide, a dual incretin agonist targeting GLP-1 and GIP receptors. It is prescribed for chronic weight management alongside lifestyle interventions like nutrition and physical activity. While effective for weight reduction and cardiometabolic benefits, it carries risks that require vigilant safety governance.
Why must healthcare providers exercise caution when prescribing Zepbound regarding NAION?
Because NAION represents a severe, potentially permanent vision loss risk linked to Zepbound use, healthcare providers must identify this adverse event early, communicate risks clearly to patients, monitor for symptoms vigilantly, and document informed consent thoroughly to ensure responsible risk management.
Are there reports of other eye-related side effects with Zepbound besides NAION?
Yes, some patients have reported experiencing Zepbound and eye floaters after starting Zepbound treatment. Although less severe than NAION, these symptoms underscore the need for ongoing monitoring and prompt management of any ocular Zepbound Eye Side Effects, during therapy.
What steps should organizations take to manage the risk of Zepbound and NAION?
Organizations involved in prescribing or dispensing Zepbound should implement proactive controls including early identification of high-severity Zepbound vision problems like NAION, clear patient counseling about potential vision loss, establishing escalation pathways for red-flag symptoms such as sudden vision changes, and maintaining disciplined documentation of all decision-making processes related to therapy risks.

If You Suffered from Zepbound and NAION or Other Zepboound Vision Problems, Contact Zepbound Vision Loss Lawyer Timothy L. Miles Today
If you were prescribed Zepbound and took it as directed and suffere Zepbound vision loss or other Zepbound eye problems, contact Zepbound Vision Loss Lawyer Timothy L. Miles today. You could be eligible for a Zepbound vision loss lawsuit and potentially entitled to substantial compensation. (855) 846–6529 or [email protected].
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com
Facebook Linkedin Pinterest youtube

