Introduction to the Dupixent Cancer Lawsuit
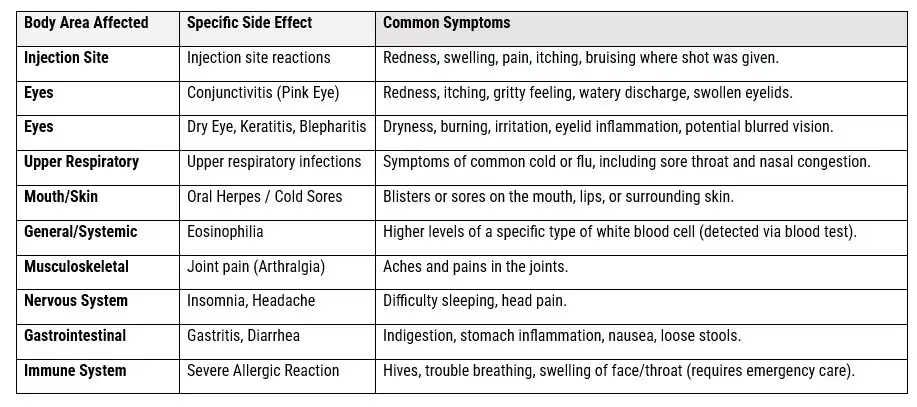
Dupixent (dupilumab) is a prescription medication used for chronic inflammatory conditions such as atopic dermatitis, asthma, and chronic rhinosinusitis with nasal polyps. While it offers significant benefits for many patients, recent legal action has focused on allegations that the manufacturers—Sanofi-Aventis and Regeneron Pharmaceuticals—failed to adequately warn about the risk of developing certain cancers, specifically cutaneous T-cell lymphoma (CTCL), in some users.
The core legal issue at the heart of the Dupixent Cancer Lawsuit is known as “failure to warn.” This doctrine requires drug manufacturers to provide healthcare providers and patients with clear, accurate, and timely information about any substantial risks associated with their products. If a company becomes aware—or reasonably should become aware—of a risk like CTCL but does not disclose it through proper labeling or communications, it can be held legally responsible for any resulting harm.
This article focuses on the failure to warn aspect of the Dupixent cancer lawsuit: what constitutes failure to warn under the law, how this applies to Dupixent’s current litigation, and what affected patients need to know if considering legal action.
What Is “Failure to Warn”?
The Legal Duty
Under U.S. product liability law, pharmaceutical companies have an ongoing duty to:
- Investigate all reasonable safety concerns related to their drugs.
- Promptly update warnings and labeling if new risks are identified.
- Adequately inform prescribers and patients of serious or life-threatening adverse effects—even if those risks are rare.
“Failure to warn” occurs when a manufacturer does not meet these obligations. In the context of medications like Dupixent, this can include:
- Insufficient warning labels about potential cancer risks.
- Omitting information about symptoms that could indicate early cancer development.
- Failing to communicate emerging evidence from post-market surveillance or adverse event reports.
Elements Needed for a Lawsuit
To succeed in a failure-to-warn claim related to Dupixent:
- Duty: The manufacturer had a legal obligation to warn about the risk.
- Breach: The warning provided was inadequate or missing.
- Causation: The lack of an adequate warning led directly to patient harm (e.g., delayed diagnosis or improper monitoring).
- Damages: The patient suffered quantifiable harm—medical costs, lost wages, pain and suffering—as a result.
Failure to Warn in the Dupixent Litigation
Allegations Against Manufacturers
Plaintiffs in current lawsuits allege that Sanofi-Aventis and Regeneron:
- Did not include explicit warnings about cutaneous T-cell lymphoma (CTCL) in prescribing information or patient guides despite accumulating case reports.
- Failed to update labeling or issue “Dear Healthcare Provider” letters after learning of potential links between Dupixent use and increased CTCL incidence.
- Did not ensure that prescribers could recognize early signs of lymphoma potentially masked by improvement in eczema-like symptoms.

Regulatory Oversight
The FDA monitors adverse events reported for all approved medications. If serious risks emerge post-market, manufacturers are expected to:
- Update warnings promptly (including bolded boxed warnings for severe outcomes).
- Communicate new safety information through official channels.
- Proactively conduct additional safety studies if needed.
Failure in any of these areas may constitute negligence under failure-to-warn law.
How Failure to Warn Harms Patients
Without proper warnings:
- Patients may ignore persistent or worsening symptoms that could signal cancer.
- Physicians may misattribute lymphoma-related skin changes as improvements in dermatitis rather than possible malignancy progression.
- Delayed diagnosis can result in more advanced disease at detection, requiring more aggressive treatment with higher risks and worse outcomes.
Compensation in Failure-to-Warn Cases
If a court finds that inadequate warnings contributed to harm, affected patients may be entitled to compensation for:
- Medical expenses (diagnostic tests, treatments)
- Lost income due to illness
- Pain and suffering
- Long-term care needs
- Loss of consortium (for family members)
Final Thoughts on a Failure to Warn
The core focus of current Dupixent lawsuits is whether Sanofi-Aventis and Regeneron failed in their duty to warn about the possible link between Dupixent and certain cancers such as CTCL. If you or someone you know developed lymphoma after using Dupixent and believe there was insufficient warning about this risk, you may have grounds for legal action based on failure-to-warn principles.
Consulting an experienced pharmaceutical litigation attorney can help determine eligibility for compensation—and ensure your rights are protected under evolving drug safety laws.