Introduction to the Dupixent Lawsuit Update and How Multidistrict Litigation Works
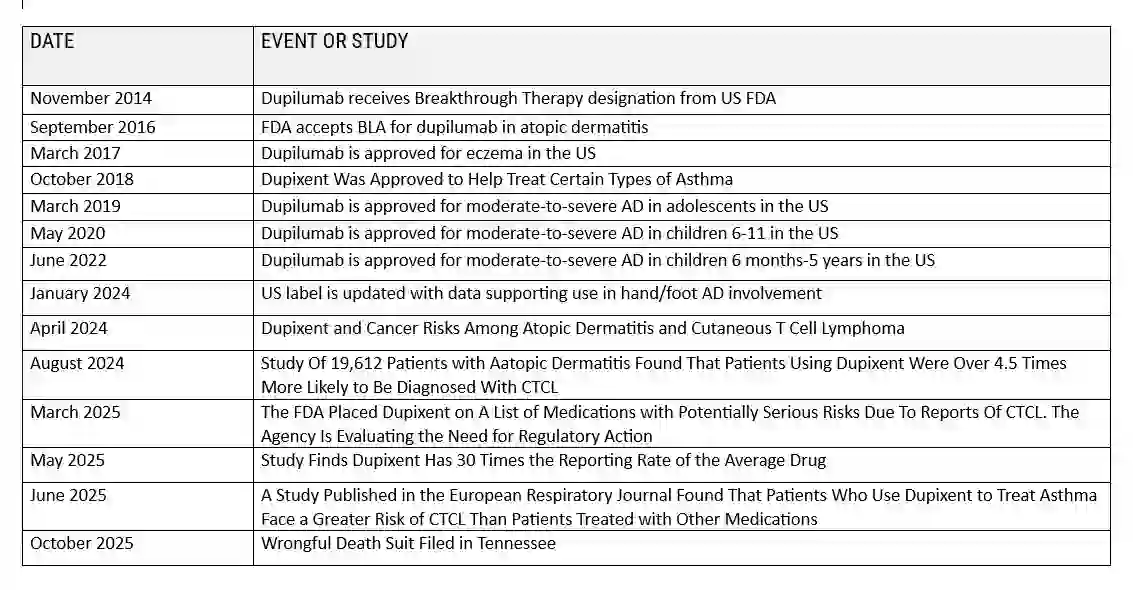
Dupixent and cancer lawsuits are in the early stages, with individual lawsuits being filed across the United States. There is currently no Dupixent class action lawsuit or multidistrict litigation (MDL) for Dupixent cases, as the damages are considered too severe and unique to each patient for a single class action. However, consolidation into an MDL is possible as more cases are filed.
Recent scientific investigations have raised concerns about a potential link between Dupixent and cancer development, particularly cutaneous T-cell lymphoma (CTCL). Multiple peer-reviewed studies published between 2024 and 2025 have documented cases suggesting an increased cancer risk among certain patient populations receiving dupilumab treatment. These findings have led to regulatory scrutiny and legal action against the drug’s manufacturers, Sanofi and Regeneron.
Dupixent (dupilumab): Is an injectable biologic medication that is used to treat particular types of inflammatory diseases. The medicine is currently the subject of a growing number of Dupixent Cancer Lawsuits alleging it causes or accelerates the development of cutaneous T-cell lymphoma (CTCL), a rare form of cancer. As of November 2025, the litigation is in early stages, with no settlements or jury verdicts yet, nor has a Dupixent class action lawsuit been filed or MDL proceedings initiated.
The MDL Process
The MDL Process: An MDL process combines numerous similar federal lawsuits, involving one or more common questions of fact, into a single court for coordinated pretrial proceedings to improve efficiency. The Judicial Panel on Multidistrict Litigation (JPML) decides if cases should be consolidated and designates a single judge to manage the coordinated pretrial process for all involved parties. Specifically, the statute, 28 U.S.C. § 1407(a), provides that “[w]hen civil actions involving one or more common questions of fact are pending in different districts, such actions may be transferred to any district for coordinated or consolidated pretrial proceedings.” (emphasis added).
Convenience for all Involved: The JPML is empowered to transfer civil action if it makes the “determination that transfers for such proceedings will be for the convenience of parties and witnesses and will promote the just and efficient conduct of such actions.” Furthermore, “coordinated or consolidated pretrial proceedings shall be conducted by a judge or judges to whom such actions are assigned by the judicial panel on multidistrict litigation.”
Dexident Cancer Lawsuit MDL Coming Soon: As it is becoming clear as more and more cases are filed in the Dexident Cancer Lawsuit, when case like these are pending in different federal courts across the country, a party can file a transfer for coordinated or consolidated pretrial proceedings or the JPML may transfer the cases on its own motion. Eventually, the defendants will file a motion with the JPML to have each Dexident Cancer Lawsuit transferred to a single court before a single judge.
The Purpose of Multidistrict Litigation
- Judicial efficiency: It prevents duplicative discovery and inconsistent rulings that would occur if each Dexident Cancer Lawsuit was handled separately.
- Consistency: It ensures that cases with similar facts are treated consistently.
- Convenience: It is more convenient for the parties, witnesses, and the judiciary.
Key Steps in the MDL Process
The multidistrict litigation (MDL) process is a federal legal procedure designed to manage numerous civil cases with common questions of fact filed in different U.S. district courts. The primary goal is judicial efficiency by centralizing pretrial activities, avoiding duplicative discovery, and ensuring consistent rulings.
The MDL Process Steps
The MDL process generally follows these key stages:
- Case Filing: Multiple lawsuits involving similar issues (e.g., defective products, dangerous drugs, mass disasters) are filed across various federal courts.
- JPML Review and Transfer: A party (plaintiff or defendant) or the court itself can petition the Judicial Panel on Multidistrict Litigation (JPML) to consolidate the cases. The JPML, a panel of seven federal judges, decides if consolidation is appropriate and selects a single “transferee” court and judge to oversee all the cases for pretrial proceedings.
- Coordinated Pretrial Proceedings: The assigned MDL judge manages all common pretrial matters for every case in the MDL. This often involves:
-
- Leadership Appointment: The judge appoints plaintiffs’ and defense steering committees to coordinate strategy and speak for their respective sides.
-
- Discovery: Both sides engage in extensive evidence gathering, document exchange, and depositions (sworn testimony), pooling resources to handle massive amounts of information efficiently.
-
- Motions: The judge rules on various legal motions, such as motions to dismiss or motions regarding the admissibility of evidence.
- Bellwether Trials: If cases do not settle during the pretrial phase, the judge may select a few representative cases for “bellwether” trials. The outcomes of these test trials help all parties assess the strengths and weaknesses of their claims and defenses, which in turn guides potential settlement negotiations for the remaining cases.
-
- Settlement: Most MDLs result in a global settlement, where defendants create a fund to compensate most or all plaintiffs who agree to the terms.
-
- Remand: If a case is not resolved through settlement or dismissal, it is sent back (“remanded”) to its original federal district court for an individual trial.
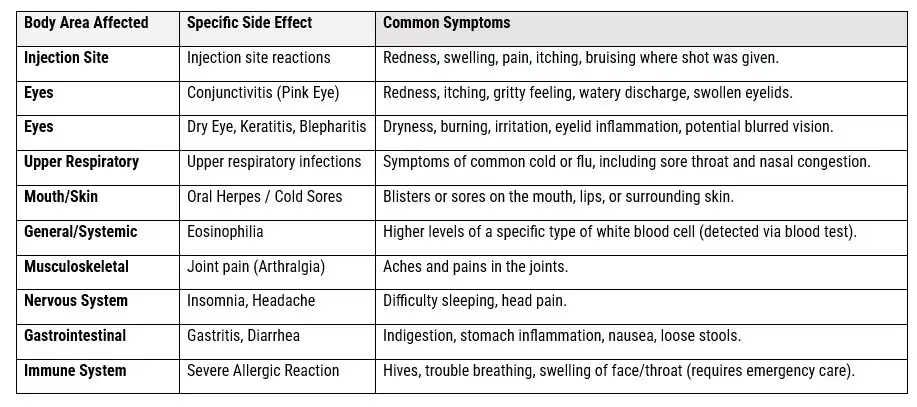
THE COMMON DUPIXENT SIDE EFFECTS PROFILE
What is a Mass Tort?
Mass Torts: A mass tort refers to a legal action that involves a large number of plaintiffs who have suffered similar injuries or damages as a result of the actions or negligence of a single defendant or group of defendants. Unlike a class action lawsuit, where a large group of people are represented by one or a few individuals, mass tort cases treat each plaintiff as an individual case.
Each Plaintiff Retains their Separate Case: This means that each plaintiff has their own separate claim, but the cases are consolidated for purposes of efficiency and convenience. Mass torts are often filed against pharmaceutical companies, as in the case of the Dexident Cancer Lawsuit, manufacturers of defective products, or entities responsible for environmental disasters.
The Dexident Cancer Lawsuit: One key characteristic of a mass tort is that the injuries or damages suffered by the plaintiffs are similar. For example, in a mass tort against a pharmaceutical company, as in the Dexident Cancer Lawsuit, the plaintiffs may have all taken the same medication and experienced severe side effects or complications. The similarities in their injuries allow their cases to be consolidated into a mass tort, rather than each filing a separate lawsuit. This consolidation allows for greater efficiency in the legal process and can also result in more consistent outcomes for the plaintiffs.
Involve Large Number of Plaintiffs: Another important aspect of mass torts is that they often involve a large number of plaintiffs. This could range from hundreds to thousands or even more individuals who have been affected by the same actions or negligence of the defendant. The sheer number of plaintiffs can make these cases complex and challenging to manage. However, by consolidating the cases into a mass tort, the legal process can be streamlined and resources can be effectively utilized.
Similar Injuries: A mass tort is a legal action that involves a large number of plaintiffs who have suffered similar injuries or damages as a result of the actions or negligence of one or more defendants as is alleged in the Dexident Cancer Lawsuit, where each plaintiff took the same drug, but allege, among other things, the defendant did not warn patients or healcare providers about certain Dupixent side effects, most notably Dupixent and cancer risks.
Conclusion: These cases are consolidated for efficiency and convenience, allowing for streamlined management and potentially more consistent outcomes. Mass torts often arise in cases involving pharmaceutical companies, manufacturers of defective products, or entities responsible for environmental disasters. The consolidation of cases can be achieved through approaches such as multidistrict litigation or coordination of state court actions.
The Key Differences from a Class Action
Here are the key differences between MDL (Multidistrict Litigation) and Class Action lawsuits:
- Nature of Lawsuit: A class action is a single lawsuit filed by a group (class) of people, while MDL involves multiple individual lawsuits that share common facts but remain separate cases.
- Plaintiffs: In a class action, class representatives act on behalf of all members of the class. In MDL, each plaintiff maintains their individual lawsuit, but they are coordinated for pre-trial proceedings.
- Procedure: MDL is used for efficiency in handling complex cases by centralizing them in one court, whereas a class action consolidates claims into one single case.
- Outcomes: The outcomes in a class action affect all class members, while in MDL, each case can have different outcomes based on individual circumstances.
- These distinctions help clarify the legal processes involved in each type of lawsuit.
Bellwether Trials
- Bellwether Trials: A bellwether trial is a test case selected from a pool of similar lawsuits, typically within a larger Multidistrict Litigation (MDL), to help guide settlement discussions and resolve the entire group of cases. While the outcome of a bellwether trial does not directly bind other cases, it provides valuable information about jury reactions, evidence strength, and legal issues, which helps both parties evaluate the strengths and weaknesses of their claims and can lead to faster, more efficient settlements.
How Bellwether trials work
-
Case selection:A pool of representative cases is first selected from the larger MDL. These cases are then advanced for discovery.
-
Trial:One or more cases are chosen from the pool to go to trial. These are the “bellwether” cases, named after the practice of a bellwether sheep leading a flock.
-
Outcome:The verdict from a bellwether trial provides a preview of how other similar cases might fare in court. It offers insights into how a jury might respond to the evidence and arguments, the effectiveness of expert testimony, and the overall strengths and weaknesses of the claims.
-
Resolution:The results of the bellwether trials can lead to significant settlement discussions and agreements for the rest of the cases in the MDL, sometimes resolving hundreds or thousands of claims at once.
-
Benefits:This process can reduce the time and cost of litigation for everyone involved. It also provides a way to test complex claims that would be too burdensome for individual trials.

The Dupixent Cancer Lawsuit Status
Frequently Asked Questions About the Dupixent Cancer Lawsuit
What Is the Compensation in a Dupixent Cancer Lawsuit?
Individuals who develop cutaneous T-cell lymphoma following Dupixent treatment may pursue compensation in a Dupixent cancer lawsuit to address the substantial financial and personal burdens resulting from their diagnosis. The legal framework governing pharmaceutical litigation recognizes multiple categories of recoverable damages designed to restore plaintiffs to their pre-injury status to the extent monetary compensation permits.
Who Can Qualify for a Dupixent Cancer Lawsuit?
Individuals seeking to qualify for a Dupixent cancer lawsuit must meet specific eligibility criteria established through legal precedent and medical documentation requirements. The primary qualification centers on demonstrating a direct connection between Dupixent usage and subsequent cancer diagnosis.
Primary Eligibility Requirements
To be eligible for a Dupixent lawsuit, claimants must satisfy the following fundamental criteria:
- Confirmed CTCL Diagnosis: Medical records must document a diagnosis of Cutaneous T-Cell Lymphoma, including Mycosis Fungoides or Sézary syndrome, following Dupixent treatment
- Documented Dupixent Use: Prescription records and pharmacy documentation establishing regular use of dupilumab for an FDA-approved condition
- Temporal Relationship: Evidence demonstrating the cancer diagnosis occurred during or after Dupixent treatment, establishing a plausible timeline
- Medical Documentation: Comprehensive medical records including biopsy results, pathology reports, and oncology consultations confirming CTCL diagnosis
Additional Qualifying Factors
Beyond the primary requirements, several supplementary factors strengthen eligibility:
Exclusionary Circumstances
Certain situations may disqualify potential claimants from pursuing legal action:
Individuals who qualify for a Dupixent cancer lawsuit should gather comprehensive medical records, prescription histories, and documentation of all treatment-related expenses. The strength of a potential claim depends substantially on the quality and completeness of medical evidence establishing the connection between Dupixent exposure and subsequent CTCL development.
What Is the Link Between Dupixent and Cancer Risks
Scientific investigations have revealed a statistically significant association between Dupixent use and elevated CTCL risk with Dupixent. Research published in peer-reviewed medical journals demonstrates that patients treated with dupilumab exhibited a substantially higher incidence of cutaneous T-cell lymphoma compared to control populations. The data indicates a risk elevation ranging from 2.5 to 4.59 times greater than baseline rates observed in patients not receiving the medication.
The most compelling evidence emerged from post-marketing surveillance studies and clinical trial analyses conducted after Dupixent received FDA approval in 2017. These investigations tracked thousands of patients across multiple therapeutic indications, including atopic dermatitis, asthma, and chronic rhinosinusitis with nasal polyposis. The link between Dupixent and cancer became apparent when researchers identified clusters of CTCL diagnoses among patients with no prior history of lymphoproliferative disorders.
Key Research Findings:
- Patients with moderate-to-severe atopic dermatitis treated with dupilumab demonstrated a 4.59-fold increased risk of developing CTCL
- The median time from treatment initiation to CTCL diagnosis ranged from several months to multiple years
- Risk appeared dose-dependent and duration-dependent in certain patient populations
- Younger patients without traditional risk factors for lymphoma developed aggressive forms of CTCL
The mechanism underlying Dupixent and cancer risks remains under active investigation. Dupilumab functions as an interleukin-4 receptor alpha antagonist, blocking signaling pathways involved in type 2 inflammatory responses. Researchers hypothesize that sustained immunomodulation may create conditions favorable for malignant T-cell proliferation in susceptible individuals. Alternative theories suggest the medication may unmask pre-existing subclinical lymphoproliferative conditions or directly influence T-cell differentiation pathways.
These findings prompted regulatory agencies to scrutinize safety data and manufacturers to update prescribing information, though questions persist regarding the adequacy and timing of risk communication to healthcare providers and patients.