When the body experiences a change in sugar level, it can affect the shape of the eye’s lens. This change is what causes blurry vision, a side effect of semaglutide.
American Academy of Ophthalmology, Vered Hazanchuk, Can Ozempic Affect Eye Health? Here’s What Ophthalmologists Want You to Know. Jul. 11, 2024.
Introduction to Demystifying Mounjaro and Blurry Vision
- Mounjaro and Blurry Vision: Another of the Mounjaro vision side effects that has recently surges in that Mounjaro can cause blurry vision, most often due to rapid changes in blood sugar levels that temporarily affect the shape of the eye’s lens. This is a common side effect that may improve as your body adjusts, but it could also be a sign of underlying conditions like diabetic retinopathy or, rarely, a more serious issue. You should contact your doctor, especially if the blurry vision is sudden, persistent, or accompanied by other symptoms like vision loss or pain.
- Mounjaro (tirzepatide): Is a significant advancement in the treatment of type 2 diabetes and obesity. It works by activating two receptors in the body that help regulate blood sugar levels and promote weight loss. This medication has shown great effectiveness in controlling blood sugar and reducing weight, leading to its widespread use among patients and healthcare providers.
- Mounjaro Vision Problems: New evidence has raised serious concerns about vision-related complications linked to Mounjaro use. Clinical research and reports of sudden vision loss, blindness, and a condition called nonarteritic anterior ischemic optic neuropathy (NAION) have led thousands of patients filing an against the manufacturer. These complications are potentially life-changing injuries that go beyond the usual risks associated with diabetes medications.
- Mounjaro Vision Loss Lawsuit Landscape: Has evolved a wave of litigation involving federal and state courts across multiple jurisdictions. Affected patients allege that Eli Lilly and Company failed to provide adequate warnings about the severe Mounjaro vision problems associated with tirzepatide therapy.
THE MOUNJARO COMMON AND SERIOUS SIDE EFFECTS PROFILE
Recent Concerns: Mounjaro and Ocular Complications
- Emerging Research on Monjaro and Visual Loss: Recent clinical observations and emerging research have identified a concerning association between Mounjaro and vision loss, specifically Nonarteritic Anterior Ischemic Optic Neuropathy (NAION). This condition causes sudden, painless vision loss due to insufficient blood flow to the optic nerve head.
- Permanent Vision Impairment or Blindness: The implications of this connection go beyond routine monitoring of side effects, as NAION can lead to permanent vision impairment or blindness.
Why Understanding This Relationship Matters
- Link Between Mounjaro and Vision Loss: Understanding the relationship between Mounjaro vision problems and NAION becomes crucial for:
-
- Informed patient consent and treatment decision-making
- Early recognition of warning signs requiring immediate medical intervention
- Risk assessment among vulnerable patient populations
- Development of preventive strategies to minimize eye complications
- Scrutinize Mounjaro’s Risk Profile: The identification of this potential risk profile requires careful examination of available evidence, clinical manifestations, and appropriate management protocols. This is essential to ensure patient safety while still providing access to effective treatments for diabetes and obesity.
Mechanism of Action
- Treating Metabolic Disorders: Mounjaro, also known as tirzepatide, is a groundbreaking medication developed by Eli Lilly for the treatment of metabolic disorders.
- Dual-Agonist Mechanism: Unlike previous diabetes medications, Mounjaro uses a unique approach called dual-agonist mechanism to target multiple pathways in the body.
How Mounjaro Works: Activating Two Hormone Receptors
- How Mounraro Works: Tirzepatide works by activating two specific hormone receptors in the body:
-
- GLP-1 receptor: This receptor is responsible for stimulating insulin secretion, reducing glucagon release, slowing down stomach emptying, and promoting feelings of fullness.
-
- GIP receptor: The activation of this receptor enhances insulin secretion and may have an impact on fat metabolism and energy expenditure.
- Dual Mechanism of Action: By targeting both the GLP-1 and GIP receptors simultaneously, Mounjaro aims to achieve better results in managing blood sugar levels and promoting weight loss compared to medications that only focus on one pathway.
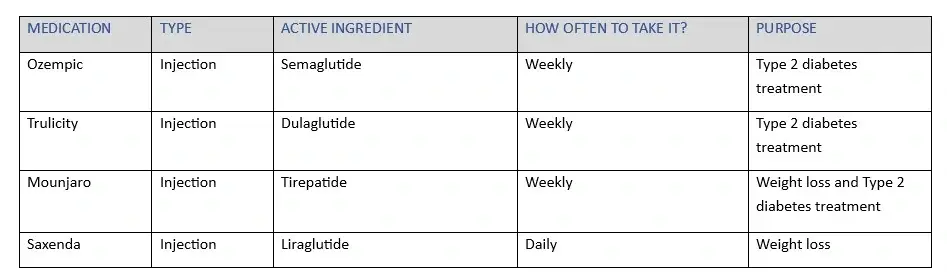
MOUNJARO GLP-1 DRUG COMPARISON CHART
FDA Approval and What It’s Used For
- FDA Approval: In May 2022, the FDA approved Mounjaro for use in adults with type 2 diabetes. The approval was based on extensive clinical trials showing that tirzepatide was more effective than existing treatments in lowering hemoglobin A1c levels (a measure of long-term blood sugar control) and helping patients lose weight.
- Once-a-Week-Injection: Mounjaro is given as an injection once a week, with specific guidelines on how to increase the dosage gradually to minimize side effects and maximize its effectiveness. It is recommended to be used alongside dietary changes and exercise programs as part of a comprehensive approach to managing metabolic diseases.
Mounjaro and Blurry Vision
- Temporary Blurry Vision: As Mounjaro helps lower blood sugar, it can change the shape of the lens in your eyes, causing temporary blurry vision. This is especially common when first starting the medication. Older patients are more likely to experience blurred vision when beginning the medication, because the eye’s lens becomes less flexible as we age, so vision takes longer to stabilize when the body experiences these changes in blood sugar levels.
What to Do if You Experience Mounjaro and Blurry Vision
-
Diabetic retinopathy: For those with diabetes, rapid blood sugar improvement can sometimes temporarily worsen diabetic retinopathy (a condition affecting the retina), which can lead to vision changes.
- Contact your doctor: It is crucial to inform your doctor about any vision changes.
- Seek urgent care: If you experience sudden vision loss, a dark shadow in your vision, severe eye pain, or vision loss with headache and nausea, seek urgent medical care.
-
See an eye doctor: Schedule an appointment with an eye specialist within 24–48 hours if you have persistent new blurring that doesn’t settle after a few days.
-
Get a baseline eye exam: It is recommended to have a baseline eye exam before starting Mounjaro, especially if you have diabetes or other eye conditions.
-
Monitor your eyes:Your eye doctor will guide you on how often you need to be checked. It is important to keep your eye doctor, primary care doctor, and endocrinologist informed about your Mounjaro use.
Other Types of Vision Problems Reported with Mounjaro Use
Diabetic Retinopathy
- Diabetic retinopathy is a major concern for patients using GLP-1 receptor agonists like Mounjaro. It involves gradual harm to the blood vessels in the retina, which can be seen through signs such as microaneurysms, bleeding, and new blood vessel growth. Interestingly, while Mounjaro treatment aims to control blood sugar levels quickly, it may unintentionally worsen retinopathy in susceptible individuals, especially those with existing microvascular issues. This is because a sudden drop in blood glucose levels can disturb the retinal blood vessels, causing them to become more permeable and leading to insufficient blood supply.
Macular Edema
- Another complication associated with Mounjaro is macular edema, characterized by fluid buildup in the macula—the central part of the retina responsible for sharp vision. As a result, patients may experience blurred central vision, difficulty reading, and altered color perception. This condition occurs when the barrier between the blood vessels and retinal tissue is disrupted, allowing fluid leakage into the macula and compromising visual clarity.
Dry Eye Syndrome
- Mounjaro Vision Side Effects and Dry Eyes: Multiple case reports have documented instances of dry eye syndrome among users of GLP-1 receptor agonists. Patients may feel discomfort on the surface of their eyes, experience burning sensations, and have intermittent blurry vision due to insufficient tear production or excessive evaporation of tears. The underlying mechanism could involve effects on the autonomic nervous system or inflammatory responses triggered by the medication.
Myodesopsias (Floaters)
- Another phenomenon reported by some patients is myodesopsias, commonly known as floaters. These individuals perceive dark spots or cobweb-like shapes moving across their field of vision—an occurrence stemming from changes in the vitreous humor or disturbances within the retina.
Nonarteritic Anterior Ischemic Optic Neuropathy
- Breaking down the meaning of the name component-by-component:
-
- Non-Arteritic: The reason it is called “non-arteritic” is because there is reduced blood flow without true inflammation of the blood vessels (as there is in arteritis).
-
- Anterior: It is called “anterior” because the reduced blood flow and injury to the optic nerve happen at the front-most part of the nerve, where the nerve meets the eye.
-
- Ischemic: The reason it is called “ischemic” is because that is the word that describes an injury due to reduced blood flow.
-
- Optic Neuropathy: The reason it is called an “optic neuropathy” because it is an injury to the optic nerve, which disrupts the ability of eye to send information to the brain.
Defining the Characteristics of NAION
- NAION is characterized by sudden, painless, unilateral vision loss upon awakening.
- Is the most common cause of optic nerve swelling and optic neuropathy in adults older than 50.
- NAION and is caused by decreased blood flow to the front part of the optic nerve (optic disc). However, the exact mechanism causing reduced blood flow to the optic nerve is not proven.
- What is known is that NAION occurs more often when a patient has conditions such as diabetes, high blood pressure, and sleep apnea. Additionally, smoking may also elevate the risk of developing NAION.
- Additionally, it happens more often in patients born with small optic discs (the front part of the optic nerve that can be seen within the eye).
- It typically affects one eye, although the other eye sometimes suffers similar loss months or years later (there is about a 15% risk of second eye involvement within 5 years). Rarely, both eyes may be affected at the same time.
- Approximately 15% of people with NAION in one eye will develop NAION in the other eye within 5 years.
- No Effective Treatments at this Time: Unfortunately, there are no treatments for NAION that are proven to be effective. Despite numerous clinical trials studying over a dozen different therapies, none have convincingly improved the visual outcome in patients with NAION. Nevertheless, numerous neuro-ophthalmologists recommend some medical treatments and basic steps for your health:
- Talk with Your Primary Doctor: Screening for diabetes, high cholesterol, high blood pressure, and sleep apnea are important for your overall health.
- Oral Steriod with Caution: Some neuro-ophthalmologists will treat patients with very recent visual loss or worsening visual loss from NAION with oral steroids for a few weeks. Other neuro-ophthalmologists worry that the negative effects from the steroids outweigh the potential benefits.
- Aspirin Regimen: There are many neuro-ophthalmologists who recommend taking daily aspirin with the hope of reducing the risk of the other eye becoming involved, although it is uncertain this helps. Aspirin reduces the risk of heart attacks and strokes, which can also occur in people with NAION.
- Stop Smoking: If you smoke, talk with your primary care provider to stop smoking.
- Medications Associated with NAOIN: If you take medications for erectile dysfunction or amiodarone, please let your doctor know, as these agents can sometimes be associated with NAION.
- Talk with Your Doctor About a Sleep Apnea Test: Because there is some evidence that the risk of second eye involvement may be higher in patients with untreated sleep apnea.
The Link Between Mounjaro and NAION: Evidence and Research Findings
- Recent Clinical Investigations: Have identified a concerning association between GLP-1 receptor agonists, including Mounjaro (tirzepatide), and elevated rates of nonarteritic anterior ischemic optic neuropathy.
- Mounjaro Vision Loss Lawsuit: A landmark study published in 2024 examined medical records from a large ophthalmology practice, revealing statistically significant increases in NAION incidence among patients prescribed these medications for type 2 diabetes and obesity management. Another study established a concerning connection between GLP-1 receptor agonists—including both Mounjaro (tirzepatide) and Ozempic (semaglutide)—and increased incidence rates of NAION. Clinical observations have documented elevated occurrences of this condition among patients prescribed these medications compared to individuals using alternative diabetes treatments.
Epidemiological Evidence of Increased NAION Risk
- NAION Occurrence Rates: Clinical Results: The research documented NAION occurrence rates across distinct patient populations:
-
- Type 2 diabetes patients: Those prescribed semaglutide or tirzepatide demonstrated a 4.28-fold increased risk of developing NAION compared to patients using alternative diabetes medications
- Obesity treatment cohorts: Individuals receiving GLP-1 drugs for weight management exhibited a 7.64-fold elevated risk relative to those prescribed other weight-loss interventions
- Absolute incidence rates: The cumulative three-year probability of NAION development reached 8.9% among GLP-1 drug users versus 1.8% in comparison groups
- Linking Mounrjaro to Optic Nerve Problems: These findings represent the first substantial epidemiological evidence linking Mounjaro vision problems to this severe optic nerve disorder. The magnitude of risk elevation has prompted calls for enhanced surveillance protocols and patient counseling regarding potential ocular complications.
Scientific Studies Investigating the Correlation Between Mounjaro and Vision Loss Incidents
- Investigating the Link to GLP-1 Receptors: The medical community has initiated rigorous investigations into the potential association between GLP-1 receptor agonists and adverse ocular outcomes. Research published in JAMA Ophthalmology has provided critical insights into the relationship between these medications and vision-threatening conditions, particularly nonarter
Insights on Mechanisms Behind Adverse Effects
- Mounjaro Eye Problems Due to Reduced Blood Flow to the Optic Nerve: Research emphasizes the role of rapid glycemic fluctuations in compromising optic nerve perfusion. The sudden normalization of blood glucose levels, while therapeutically beneficial for metabolic control, may paradoxically create conditions that reduce blood flow to the optic nerve head.
- Normalization of Ichemia (Oxygen Supply to Blood): This phenomenon, termed “normalization ischemia,” represents a critical pathway through which GLP-1 medications may precipitate vision loss.
Clinical Monitoring Protocols
- Establishing a Bassline Assessment: Physicians recommend establishing baseline ophthalmologic assessments before initiating Mounjaro therapy. This initial evaluation creates a reference point for detecting subsequent changes in ocular health. Patients should receive clear instructions to report any visual disturbances immediately, including:
-
- Sudden onset of blurred vision or visual field defects
- Unexplained eye pain or pressure
- Changes in color perception
- Appearance of floaters or flashing lights
- Difficulty with night vision or contrast sensitivity
- FDA Warnings: The FDA warnings on GLP-1 drugs underscore the necessity for heightened vigilance during the first several months of treatment, when metabolic changes occur most rapidly. Healthcare teams must educate patients about the distinction between temporary visual fluctuations related to blood sugar stabilization and potentially serious complications requiring urgent evaluation.
Frequently Reported Ocular Symptoms
- Mounjaro Eye Side Effects: Clinical observations and patient reports have documented several visual disturbances associated with tirzepatide therapy:
-
- Eye twitching (myokymia): Involuntary eyelid spasms affecting one or both eyes, typically presenting as benign fasciculations of the orbicularis oculi muscle
- Dry eye syndrome: Reduced tear production or increased tear evaporation leading to ocular surface irritation, burning sensations, and foreign body sensation
- Eye inflammation: Conjunctival hyperemia, periorbital edema, or anterior uveitis manifesting as redness, pain, and photophobia
- Eye floaters: Perception of spots, threads, or cobweb-like structures in the visual field resulting from vitreous changes or posterior vitreous detachment
- Blurred vision: Decreased visual acuity affecting near or distance vision, with varying degrees of severity and duration
Blood Sugar Fluctuations and Visual Acuity
- Metabolic Changes: The pharmacodynamic properties of Mounjaro produce rapid glycemic control in many patients, creating substantial shifts in blood glucose levels. These metabolic changes directly affect the osmotic balance within ocular structures, particularly the crystalline lens. Glucose concentration alterations modify the refractive index of the lens, resulting in temporary blurred vision that may persist for several weeks during dose titration phases.
- Normalization of Glucose Levels: Patients experiencing rapid hemoglobin A1c reduction face heightened risk for refractive changes. The lens accommodates to chronic hyperglycemic states; sudden normalization of glucose levels disrupts this adaptation, producing transient myopia or hyperopia. This phenomenon typically resolves within four to eight weeks as ocular tissues equilibrate to improved metabolic conditions.
Proposed Pathophysiological Mechanisms
Understanding the causes of vision loss from Mounjaro requires examination of multiple biological pathways. Researchers have proposed several mechanisms through which GLP-1 drugs and NAION risk may be connected:
- Hemodynamic alterations induced by rapid glucose normalization may compromise perfusion pressure within the optic nerve head. The sudden metabolic shifts associated with tirzepatide therapy can destabilize autoregulatory mechanisms that maintain adequate blood flow to vulnerable neural tissues.
- Inflammatory mediator modulation represents another hypothesized pathway. GLP-1 receptor activation influences cytokine profiles and vascular endothelial function, potentially creating conditions conducive to microvascular occlusion in anatomically predisposed optic discs.
- Structural crowding at the optic nerve head, particularly in patients with small cup-to-disc ratios, may interact synergistically with medication-induced physiological changes. The risk factors for NAION with Mounjaro appear amplified in individuals possessing this anatomical vulnerability, where compromised circulation through an already crowded space precipitates ischemic injury.
Quality of Life Implications
- Mounjaro Vision Damage: The cumulative burden of visual disturbances significantly affects patient adherence and daily functioning. Individuals experiencing persistent dry eye syndrome report difficulty with computer work, reading, and driving, particularly during evening hours. Eye floaters create anxiety and distraction, interfering with concentration and task completion. Unpredictable episodes of blurred vision compromise occupational performance and recreational activities requiring precise visual acuity.
- Document Symptoms: Documentation of symptom severity, frequency, and temporal relationship to medication administration provides essential data for clinical decision-making. Patients maintaining detailed symptom diaries facilitate accurate assessment of whether vision side effects represent benign adaptation phenomena or herald more serious pathological processes requiring intervention.
Proposed Pathophysiological Mechanisms
Understanding the causes of vision loss from Mounjaro requires examination of multiple biological pathways. Researchers have proposed several mechanisms through which GLP-1 drugs and NAION risk may be connected:
- Hemodynamic alterations induced by rapid glucose normalization may compromise perfusion pressure within the optic nerve head. The sudden metabolic shifts associated with tirzepatide therapy can destabilize autoregulatory mechanisms that maintain adequate blood flow to vulnerable neural tissues.
- Inflammatory mediator modulation represents another hypothesized pathway. GLP-1 receptor activation influences cytokine profiles and vascular endothelial function, potentially creating conditions conducive to microvascular occlusion in anatomically predisposed optic discs.
- Structural crowding at the optic nerve head, particularly in patients with small cup-to-disc ratios, may interact synergistically with medication-induced physiological changes. The risk factors for NAION with Mounjaro appear amplified in individuals possessing this anatomical vulnerability, where compromised circulation through an already crowded space precipitates ischemic injury.
Differentiating Between Mild and Serious Manifestations
Mild Mounjaro vision problems include:
- Intermittent eye twitching or lid spasms
- Temporary blurred vision coinciding with blood sugar changes
Serious symptoms that require immediate evaluation include:
- New visual field defects
- Ongoing changes in vision unrelated to glucose levels
The timing of when symptoms start in relation to when medication is initiated or increased can provide valuable diagnostic information. Keeping a record of visual disturbances through systematic self-monitoring helps healthcare providers differentiate between medication-related effects and unrelated eye problems, allowing for appropriate intervention strategies.
Who is at Risk for Vision Loss When Using Mounjaro?
- Higher Susceptibility to Mounjaro Vision Damage: Certain groups of patients are more likely to experience Mounjaro Vision Problems. It is important to identify these risks before starting tirzepatide treatment.
- Proper Minorizing Protocol: By understanding these factors, healthcare providers can set up proper monitoring and protection strategies.
High-Risk Patient Categories
- Elderly Patients: Are particularly vulnerable to Mounjaro eye issues. Age-related changes in blood vessels can affect the supply of blood to the optic nerve, increasing the risk of ischemic events. Patients over 60 years old have less ability to compensate for changes in blood flow or energy needs of the optic nerve.
- Pre-Existing Diabetic Eye Disease: Face additional risks when using Mounjaro. Those diagnosed with diabetic retinopathy, macular edema, or previous episodes of vision impairment already have weakened eye structures due to long-term high blood sugar levels. The introduction of a powerful medication that lowers glucose levels may disrupt the delicate balance in these eyes.
- Established Vascular Disease—such as coronary artery disease, peripheral arterial disease, or conditions affecting blood vessels in the brain—have problems with circulation throughout their bodies, including the supply of blood to their eyes. These individuals have difficulty regulating blood flow on their own, which limits the eye’s ability to maintain adequate oxygen and nutrient delivery during metabolic changes.
Metabolic Fluctuations as Risk Amplifiers
- Sudden Drop in Glucose: Mounjaro can cause rapid changes in blood sugar levels, which can be especially dangerous for certain individuals. When glucose levels drop suddenly, it can disrupt the balance of fluids in the tissues of the eye and potentially reduce blood flow to the optic nerve head.
- Aggressively Control Blood Sugar: Patients who are undergoing aggressive control of their blood sugar levels—especially those transitioning from poorly managed diabetes—are at greater risk during the early stages of treatment.
Personalized Risk Assessment Protocols
- Complete Risk Assessment: Before starting Mounjaro, it is important to assess the risk of complications related to diabetes. This assessment should include:
-
- A thorough eye examination by an ophthalmologist, including evaluation of the optic nerve
- Screening for vascular health, including checking blood pressure control
- Reviewing any existing microvascular complications
- Analyzing patterns of glycemic variability and current control status
- Assessing any other medications being taken that may affect blood vessel function
Raising Awareness About the Ocular Risks Associated with Mounjaro Use
- Informational Awareness on Mounjaro Eye Problems: Is a critical part of patient safety protocols. Healthcare providers must prioritize comprehensive education initiatives that equip patients with the knowledge necessary to identify early warning signs of ocular complications. Patients need clear, accessible information about Mounjaro vision problems, ranging from minor irritations to severe conditions like NAION. Educational materials should emphasize the difference between temporary symptoms and urgent situations that require immediate eye doctor evaluation.
- Transparency: The medical community has a responsibility to establish strong monitoring systems for adverse ocular events related to tirzepatide therapy. By systematically collecting and analyzing data, researchers can improve risk assessment models and develop preventive strategies based on evidence. Pharmaceutical manufacturers must be more transparent in their safety communications, ensuring that prescribing information reflects the latest understanding of ocular risks. Regular updates to product labeling should include new evidence from post-marketing surveillance and clinical studies.
FREQUENTLY ASKED QUESTIONS ABOUT MOUNJARO VISION PROBLEMS
1. What Are the Mounjaro vision side effects?
- A study published in 2024 revealed alarming statistics about NAION risk among patients using GLP-1 receptor agonists like Mounjaro. The research demonstrated that individuals with type 2 diabetes taking these medications faced up to seven times higher risk of developing NAION compared to those using alternative diabetes treatments.
- While not an exhaustive list, some other Mounjaro vision problems include:
-
- The relationship between diabetes complications and retinopathy risk when on Mounjaro requires careful examination, as the medication’s glycemic effects create a complex interaction with existing retinal pathology. Research published in Diabetes Care journal has documented that rapid improvements in glycemic control—while therapeutically beneficial—may paradoxically accelerate diabetic retinopathy progression in susceptible individuals.
-
- Eye floaters
-
- Eye twitching
-
- Inflammation of the eye
2. Is there a link between Mounjaro and Vision Loss?
- Yes, Nonarteritic Anterior Ischemic Optic Neuropathy (NAION) represents one of the most severe vision-related risks documented in association with tirzepatide therapy.
- This condition manifests through sudden vision loss resulting from compromised blood flow to the optic nerve head, leading to optic nerve damage that may be permanent. NAION symptoms and risks include:
-
- Acute, painless vision loss typically affecting one eye
- Visual field defects, particularly altitudinal field loss
- Optic disc swelling observable during fundoscopic examination
- Irreversible vision impairment in the majority of cases
3. What should I do if I experience Mounjaro vision problems?
- Stop taking the medication and contact your healthcare provider immediately.
- Prompt assessment can help prevent further Mounjaro vision side effects.
4. Am I eligible to file a Mounjaro blindness lawsuit if I suffered Mounjaro vision problems?
- You may be eligible for a Mounjaro blindness lawsuit if you experienced significant eye-related side effects after using Mounjaro and can establish a connection between your injury and the medication.
- To see if you are eligible, contact Mounjaro blindness lawyer Timothy L. Miles for a free case evaluation. (855) 846–6529 or [email protected].
Conclusion
- Mounjaro Vision Side Effects: The emerging evidence surrounding Mounjaro Vision Problems necessitates heightened awareness among patients and healthcare professionals. A comprehensive summary of Mounjaro vision problems reveals potential associations with nonarteritic anterior ischemic optic neuropathy, diabetic retinopathy exacerbation, and other serious ocular complications that demand immediate attention
- Patient Vigilance Remains Paramount: In mitigating these risks. Individuals prescribed Zepbound must maintain proactive communication with healthcare providers, reporting any visual disturbances—no matter how subtle—without delay. Regular ophthalmologic examinations should constitute a non-negotiable component of treatment protocols, particularly for patients presenting with diabetes or pre-existing vascular conditions.
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com
Facebook Linkedin Pinterest youtube