Introduction to Wegovy and Vision Loss
- Wegovy and Vision Loss: Recent reports have raised serious concerns about Wegovy vision side effects, particularly cases of sudden vision loss and possible blindness among users.
- Wegovy: Is a prescription medication that contains semaglutide as its active ingredient. It has become widely used for managing chronic weight issues in adults with obesity or weight-related health conditions. The U.S Food and Drug Administration (FDA) approved it in 2021, and it works by activating the glucagon-like peptide-1 (GLP-1) receptor to promote significant weight loss, as shown in clinical studies.
- Failure to Warn: Patients who are experiencing Wegovy vision problems have started taking legal action against the manufacturer Novo Nordisk through Wegovy blindness lawsuits. They allege that the company did not provide adequate warnings about severe eye complications. These lawsuits focus on claims that the pharmaceutical company knew about the risks associated with non-arteritic anterior ischemic optic neuropathy (NAION) but failed to inform healthcare providers and patients properly.
- Linking Wegovy to Vision Loss: The connection between Wegovy and vision loss has led some patients who have been affected to take legal action against Novo Nordisk, claiming that the company did not provide proper warnings about the risks to their eyes. This guide offers an in-depth look at the criteria for eligibility, how compensation works, recent regulatory changes, and steps individuals can take to protect themselves if they are currently using or thinking about using GLP-1 receptor agonist therapy. By understanding these various factors, patients can make better choices about their medical treatment and explore potential legal options.
Understanding Wegovy and How It Works
- Glucagon-Like Peptide-1 (GLP-1) Receptor Agonist: Wegovy contains an active ingredient called semaglutide, which is a type of medication known as a glucagon-like peptide-1 (GLP-1) receptor agonist.
- Mimics GLP-1: This means that semaglutide works by mimicking the action of a natural hormone called GLP-1 that is produced in the intestines.
How Semaglutide Works
- Mechanism of Action: When semaglutide is taken, it binds to specific receptors in the body and triggers a series of metabolic responses. The main actions of semaglutide include:
-
- Stimulating Insulin Production: Semaglutide enhances the function of pancreatic beta cells, which are responsible for producing insulin. This leads to increased insulin secretion when blood sugar levels rise.
-
- Suppressing Glucagon Secretion: Glucagon is another hormone produced by the pancreas that raises blood sugar levels. Semaglutide inhibits the release of glucagon from alpha cells, helping to lower blood sugar levels.
-
- Improving Glucose Uptake: By amplifying the body’s natural incretin response, semaglutide promotes better uptake of glucose by peripheral tissues (such as muscles) and liver cells.
-
- Regulating Gastric Emptying: Semaglutide slows down the emptying of food from the stomach, which can lead to prolonged feelings of fullness and reduced appetite.
Benefits for Diabetic Patients
For individuals with diabetes, semaglutide offers several benefits:
- Glucose Control: The medication helps lower fasting blood sugar levels and improves HbA1c measurements (a long-term indicator of blood sugar controll).
- Reduced Risk of Hypoglycemia: Unlike some traditional diabetes medications that can cause low blood sugar episodes, semaglutide’s effects are glucose-dependent. This means it only activates when blood sugar levels are high, reducing the risk of hypoglycemia.
- Weight Management: Semaglutide has been shown to promote weight loss in individuals with obesity or overweight conditions. This can be particularly beneficial for those with type 2 diabetes who often struggle with weight management.
Potential Effects on Vision Health
Interestingly, GLP-1 receptors are also found in various tissues throughout the body, including the eyes. This suggests that semaglutide may have potential effects on vision health as well:
- Microvascular Blood Flow: The presence of GLP-1 receptors in vascular endothelium (the inner lining of blood vessels) indicates that semaglutide might influence blood flow to small vessels in the eyes.
- Inflammatory Responses: GLP-1 receptors have been implicated in modulating inflammation, which could play a role in ocular conditions associated with diabetes.
- Cellular Metabolism: Semaglutide’s impact on cellular metabolism may extend beyond metabolic organs and affect retinal cells or optic nerve function.
While further research is needed to fully understand these potential benefits, they highlight the systemic nature of semaglutide’s actions beyond its primary uses for diabetes management and weight loss.
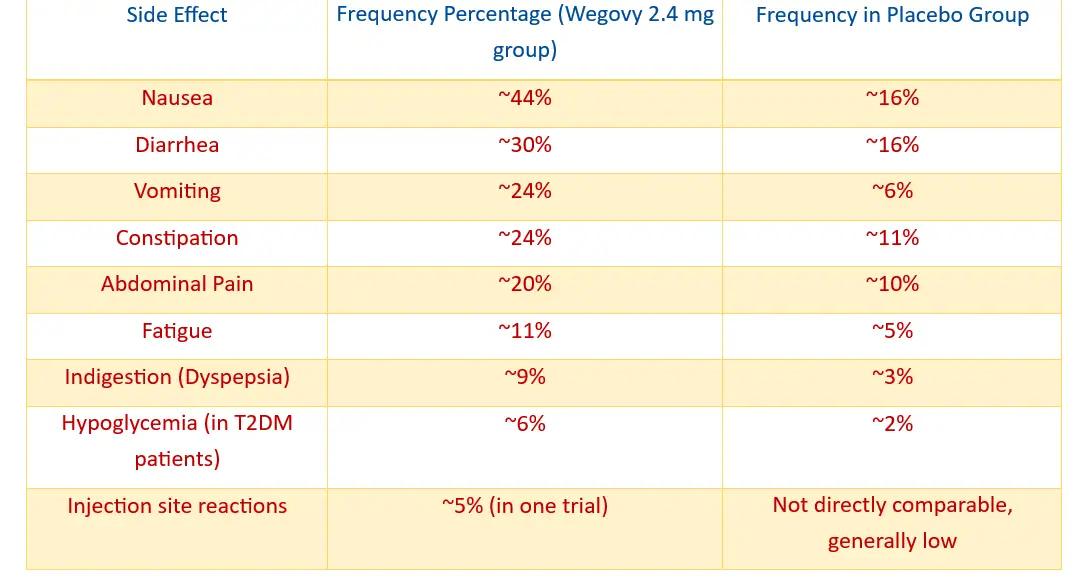
Common Vision Side Effects Associated with Wegovy
- Wegovy Vision Side Effects: Patients undergoing Wegovy treatment report various Wegovy vision side effects that range from mild to moderate in severity. Clinical documentation and patient testimonials reveal a spectrum of ocular manifestations requiring careful monitoring throughout the therapeutic course.
- Transient Symptoms: Understanding these manifestations enables patients to distinguish between transient symptoms and conditions warranting immediate medical consultation.
Blurry Vision and Blood Sugar Fluctuations
- Blurry vision represents the most frequently documented visual complaint among Wegovy users. This phenomenon correlates directly with rapid alterations in blood glucose concentrations, particularly during the initial titration phase. The crystalline lens within the eye responds to osmotic changes by absorbing or releasing fluid, temporarily affecting its refractive properties.
- Patients experiencing significant weight loss or improved glycemic control may notice fluctuating visual acuity as their metabolic parameters stabilize. These episodes typically resolve within two to four weeks as the body adapts to new glucose homeostasis levels.
Dry Eye Syndrome and Ocular Discomfort
The incidence of dry eye syndrome among semaglutide users reflects the medication’s systemic effects on fluid regulation and metabolic processes. Patients describe sensations of grittiness, burning, or foreign body presence in the eyes. The condition stems from altered tear film composition or reduced tear production, potentially exacerbated by the medication’s influence on autonomic nervous system function. Eye twitching, medically termed myokymia, occurs in a subset of users and may relate to electrolyte imbalances or stress responses during metabolic adjustment periods.
Visual Disturbances and Floaters
- Ocular Report Pandemoniums: Of eye floaters and other visual disturbances require clinical differentiation between benign vitreous changes and pathological conditions. Floaters appear as dark spots, threads, or cobweb-like structures drifting across the visual field. While often representing normal age-related vitreous degeneration, their sudden onset or increase in number during Wegovy treatment necessitates ophthalmologic evaluation to exclude retinal complications.
- Record Symptom Peculiarities: Patients experiencing these Wegovy vision problems should document symptom characteristics, frequency, and associated factors for discussion with healthcare providers or consultation with a Wegovy Vision Lawyer if complications arise.
WEGOVY SIDE EFFECTS
Serious Eye Conditions Linked to Semaglutide Use
- Non-arteritic anterior ischemic optic neuropathy (NAION): Represents the most concerning ocular complication associated with semaglutide therapy. This condition manifests as acute, painless vision loss resulting from insufficient blood supply to the optic nerve head. Research published in JAMA Ophthalmology identified a statistically significant correlation between semaglutide use and NAION development, with diabetic patients prescribed the medication demonstrating more than four times the risk compared to those using alternative treatments.
- The pathophysiology of NAION involves compromised perfusion to the anterior portion of the optic nerve, creating an ischemic event that damages nerve tissue irreversibly. The optic nerve requires continuous oxygen and nutrient delivery through small blood vessels; when this supply diminishes below critical thresholds, cellular death occurs within hours. Semaglutide’s effects on vascular function and metabolic parameters may contribute to this reduced perfusion, particularly in patients with existing microvascular compromise.
Elevated Risk Profiles
- Optic Nerve Specific: patient populations demonstrate heightened susceptibility to serious optic nerve issues during semaglutide treatment:
-
- Type 2 diabetes patients with pre-existing microvascular disease
- Individuals with obesity experiencing rapid metabolic shifts
- Patients with hypertension affecting ocular blood flow
- Those with previous cardiovascular events indicating systemic vascular dysfunction
- Ocular Inflammation: Constitutes another serious complication requiring immediate medical attention. Inflammatory responses within ocular structures can manifest as uveitis, scleritis, or retinal vasculitis, each carrying distinct implications for visual preservation. Case reports document instances of sudden blindness occurring within days of semaglutide initiation, though these events remain rare.
- Wegovy Eye Damage: The relationship between Wegovy and vision loss extends beyond NAION to encompass various inflammatory and ischemic processes affecting different ocular structures. Retinal vascular occlusions, optic disc edema, and choroidal ischemia have emerged in clinical surveillance data, necessitating comprehensive ophthalmologic evaluation protocols for patients initiating or continuing semaglutide therapy.
Risk Factors Increasing the Likelihood of Vision Loss on Wegovy
Certain patient groups are more likely to experience vision problems while using semaglutide. The following factors can increase the risk of vision loss in these individuals:
1. Type 2 Diabetes Mellitus
- Impacts Blood Vessels: People with type 2 diabetes are at the highest risk because their condition already affects blood vessels throughout the body, including those in the eyes.
- Wegovy Vision Side Effects: This can lead to complications such as diabetic retinopathy, which can worsen with rapid improvements in blood sugar levels caused by semaglutide.
2. Overweight or Obese Individuals
- The main group of people prescribed Wegovy are those who are overweight or obese.
- These individuals may have additional risks to their eye health due to factors like inflammation, high blood pressure, and abnormal lipid levels.
3. Duration of Diabetes
- The longer someone has had diabetes, the greater the chance of developing eye problems.
- Specifically, if a person has had diabetes for more than ten years, they should be closely monitored for any changes in their vision.
4. Poor Glycemic Control
- Before starting semaglutide treatment, if a person’s blood sugar levels have been poorly controlled (indicated by an HbA1c level above 9%), they may be at higher risk for vision complications.
5. History of Diabetic Retinopathy or Macular Edema
If someone has previously been diagnosed with proliferative diabetic retinopathy or macular edema, there is a possibility that their condition could worsen with Wegovy therapy.
6. Concurrent Cardiovascular Disease or Chronic Kidney Disease
Having existing cardiovascular disease or chronic kidney disease can also pose risks to retinal health. It is important to consider these conditions when evaluating a patient’s suitability for semaglutide treatment.
7. Uncontrolled Hypertension
High blood pressure that affects the blood vessels in the retina can further increase the likelihood of vision loss during Wegovy therapy. Managing hypertension is crucial in protecting eye health.
Patients who exhibit multiple risk factors should undergo thorough eye examinations before starting Wegovy treatment. If advanced diabetic retinopathy is present and worsening, alternative approaches to weight management should be carefully considered as semaglutide’s effects on metabolism may lead to irreversible vision impairment in vulnerable populations.
Recognizing Symptoms of Vision Problems Related to Wegovy Use
- Patients starting Wegovy treatment should be on the lookout for specific eye-related signs that could indicate potential complications. If you experience any symptoms of vision loss associated with Wegovy, seek medical attention immediately to prevent permanent damage to your eyes.
Key Visual Signs That Need Immediate Attention
- Wegovy Eye Damage: Blurred vision is one of the most commonly reported symptoms. It may manifest as difficulty focusing on objects at different distances or experiencing persistent haziness in your visual fields. This symptom could suggest fluctuations in blood sugar levels affecting the lens of your eye or a more serious underlying condition.
- Double vision (diplopia) is another significant warning sign. It may indicate nerve damage or muscular dysfunction affecting the coordination of your eye movements. If you experience this symptom, it’s crucial to stop any activities that require precise vision and seek urgent evaluation from an eye specialist.
- Additional Ocular Symptoms: Other symptoms that require clinical investigation include:
-
- Sudden appearance of floaters or dark spots obstructing your visual fields
- Flashing lights or photopsia occurring without any external triggers
- Loss of peripheral vision creating a tunnel-like perception
- Eye pain accompanied by changes in your vision or headaches
- Sudden loss of vision in one or both eyes, regardless of how long it lasts
- Alterations in your color vision or difficulty distinguishing between different hues
- Light sensitivity (photophobia) interfering with your daily activities
- Track Symptomatic Irregularities: The timing of when these symptoms start in relation to when you begin taking Wegovy can provide important diagnostic information. It’s helpful for patients to keep track of the exact timing, duration, and severity of any visual disturbances they experience to assist their healthcare provider in making an accurate assessment.
Medical Advice and Preventive Measures for Patients Using Wegovy
- Wegovy Vision Loss Lawsuit: Patients starting Wegovy treatment need careful monitoring to reduce the risks associated with Wegovy and Vision Loss. Before beginning treatment, it’s important to have an eye exam to establish a baseline. This exam will serve as a reference point for identifying any future changes in eye health.
- Baseline Assessment: During this initial assessment, key factors such as visual acuity (sharpness of vision), intraocular pressure (pressure inside the eye), and fundoscopic findings (examination of the back of the eye) should be documented.
Importance of Seeking Medical Advice for Wegovy-Related Vision Problems
- Systematic Observation Plan: When implementing a structured monitoring plan, it is crucial to seek medical advice for any vision problems related to Wegovy. Patients should schedule comprehensive eye examinations at specific intervals:
-
- Month 3: Initial follow-up assessment to identify early changes in retinal blood vessels or optic nerve appearance
- Month 6: Mid-treatment evaluation focusing on markers of diabetic retinopathy progression
- Month 12: Annual comprehensive examination including optical coherence tomography when necessary
- As needed: Immediate evaluation upon experiencing any visual symptoms
Intensified Monitoring Protocols for Diabetic Patients
- Monitoring Patterns: Diabetic patients require more frequent monitoring protocols, with examinations occurring every three to four months during the initial treatment phase. It is important to document any fluctuations in blood glucose levels alongside visual assessments.
- Wegovy Vision Loss Lawsuit: This information will help clinicians identify any connections between metabolic control and eye complications.
The Role of Symptom Diaries and Communication in Eye Care
- Document All Ocular Manifestations: Patients can play an active role in their eye care by maintaining detailed symptom diaries. These diaries should record the timing, duration, and characteristics of any visual disturbances experienced. This information will be valuable during ophthalmologic consultations as it can assist in differentiating between benign fluctuations and pathological processes that require intervention.
- Symmetrical Care Delivery: Effective communication between prescribing physicians and ophthalmologists is essential for coordinated care delivery. This is particularly important when dose adjustments or treatment discontinuation becomes necessary based on emerging findings related to the eyes.
Legal Aspects: Understanding the Wegovy Vision Loss Lawsuit Landscape
- Wegovy Vision Side Effects: The emergence of vision-related complications associated with Wegovy has led to an increase in lawsuits against Novo Nordisk, the pharmaceutical manufacturer.
- Multiple Wegovy vision loss lawsuits have been filed in different regions, claiming that the company did not adequately inform patients and healthcare providers about the potential risks of serious eye injuries, particularly non-arteritic anterior ischemic optic neuropathy (NAION).
Key Allegations in Wegovy Vision Loss Lawsuits
Failure to Warn: Plaintiffs in these cases argue that Novo Nordisk knew about the link between semaglutide and Wegovy eye damage but failed to provide proper warnings in product labeling or marketing materials. The legal claims usually involve several arguments:
- Failure to warn: Allegations that the manufacturer did not adequately communicate known risks to prescribing physicians and patients
- Design defect: Arguments that the medication’s formulation presents unreasonable dangers that outweigh its therapeutic benefits for certain patient populations
- Negligence: Claims that the company failed to conduct adequate post-market surveillance or respond appropriately to emerging safety signals
Impact of Research Studies on Legal Proceedings
Wegovy Vision Loss Lawsuit: Legal actions have increased following the release of research studies showing higher rates of NAION among semaglutide users. Lawyers representing affected patients are combining individual cases to establish patterns of injury and prove the manufacturer’s alleged awareness of these risks.
Litigation Landscape: The litigation landscape is continuously changing as more patients report vision problems and seek legal action for medical expenses, lost wages, and decreased quality of life resulting from permanent visual impairment.
Current Research and Future Outlook on Semaglutide’s Impact on Eye Health
Continuing Clinical Research: The medical community continues to investigate the relationship between Wegovy and vision loss through rigorous clinical research protocols. The FOCUS study semaglutide eye health research represents a crucial investigation designed to evaluate the eye safety profile of GLP-1 receptor agonists in diverse patient populations.
Tracking Vascular Abnormalities: This prospective clinical trial examines specific endpoints related to retinal complications, optic nerve function, and vascular changes within the eye structure. Researchers track patients receiving semaglutide therapy over extended periods to establish definitive causal relationships between medication exposure and vision-related adverse events.
Key research objectives include:
- Quantifying the incidence rate of NAION among semaglutide users compared to control populations
- Identifying biomarkers that predict increased susceptibility to ocular complications
- Establishing standardized monitoring protocols for patients initiating GLP-1 therapy
- Evaluating dose-dependent relationships between semaglutide concentration and vision changes
Continuing Wegovy Eye Damage Reseaech: Preliminary data from ongoing investigations suggest the necessity for enhanced screening protocols in high-risk patient categories. The scientific community anticipates publication of comprehensive findings within the next 24-36 months, which will inform updated clinical guidelines for ophthalmologic surveillance during semaglutide treatment.
Regulatory Scrutiny: Regulatory agencies continue to monitor post-marketing surveillance data to refine safety recommendations and patient education materials regarding potential ocular risks associated with Wegovy therapy.
Timothy L. Miles, Esq.
Law Offices of Timothy L. Miles
Tapestry at Brentwood Town Center
300 Centerview Dr. #247
Mailbox #1091
Brentwood,TN 37027
Phone: (855) Tim-MLaw (855-846-6529)
Email: [email protected]
Website: www.classactionlawyertn.com